Abstract
Background
This study aims to determine the genetic and clinical features of TARDBP-mutated patients in our cohort of Chinese patients with amyotrophic lateral sclerosis (ALS) combined with data in the literature.
Methods
We performed TARDBP mutation screening in 1258 Chinese ALS patients, including 1204 sporadic ALS (sALS) and 54 familial ALS (fALS) patients. A systematic literature review was conducted by searching TARDBP-mutated patients from China in the online databases.
Results
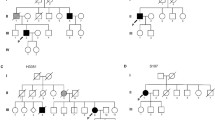
In our cohort, the mutant frequency of TARDBP variants was 0.3% (4/1258), with two recurrent variants (p.G294V, p.G298V) and one novel variant (p.S332G) identified. Combining with data in the literature review, the TARDBP-mutant frequency in the Chinese population was 1.4% (83/5998), with 0.8% (46/5470) in sALS and 7.0% (37/528) in fALS. Most patients had limb onset (63.0%), with an average life expectancy of 4.3 years (range 0.5–13). Disease durations significantly differed (p = 0.002), with p.M337V showing the longest duration (80 months) and p.N378D showing the shortest duration (16.7 months).
Conclusion
Our study found that TARDBP mutation was not rare in Chinese fALS patients. Different TARDBP mutations were associated with specific features in phenotypes.




Similar content being viewed by others
References
Zou ZY, Zhou ZR, Che CH, Liu CY, He RL, Huang HP (2017) Genetic epidemiology of amyotrophic lateral sclerosis: a systematic review and meta-analysis. J Neurol Neurosurg Psychiatry 88:540–549. https://doi.org/10.1136/jnnp-2016-315018
Chadi G, Maximino JR, Jorge FMH, Borba FC, Gilio JM, Callegaro D, Lopes CG, Santos SND, Rebelo GNS (2017) Genetic analysis of patients with familial and sporadic amyotrophic lateral sclerosis in a Brazilian Research Center. Amyotroph Lateral Scler Frontotemporal Degener 18:249–255. https://doi.org/10.1080/21678421.2016.1254245
Saberi S, Stauffer JE, Schulte DJ, Ravits J (2015) Neuropathology of amyotrophic lateral sclerosis and its variants. Neurol Clin 33:855–876. https://doi.org/10.1016/j.ncl.2015.07.012
Blokhuis AM, Groen EJ, Koppers M, van den Berg LH, Pasterkamp RJ (2013) Protein aggregation in amyotrophic lateral sclerosis. Acta Neuropathol 125:777–794. https://doi.org/10.1007/s00401-013-1125-6
Ye CH, Lu XL, Zheng MY, Zhen J, Li ZP, Shi L, Liu ZY, Feng LY, Pei Z, Yao XL (2013) Absence of mutations in exon 6 of the TARDBP gene in 207 Chinese patients with sporadic amyotrohic lateral sclerosis. PLoS ONE 8:e68106. https://doi.org/10.1371/journal.pone.0068106
Hou L, Jiao B, Xiao T, Zhou L, Zhou Z, Du J, Yan X, Wang J, Tang B, Shen L (2016) Screening of SOD1, FUS and TARDBP genes in patients with amyotrophic lateral sclerosis in central-southern China. Sci Rep 6:32478. https://doi.org/10.1038/srep32478
Etherington GJ, Ramirez-Gonzalez RH, MacLean D (2015) bio-samtools 2: a package for analysis and visualization of sequence and alignment data with SAMtools in ruby. Bioinformatics 31:2565–2567. https://doi.org/10.1093/bioinformatics/btv178
Langmead B, Trapnell C, Pop M, Salzberg SL (2009) Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 10:R25. https://doi.org/10.1186/gb-2009-10-3-r25
Schwarz JM, Cooper DN, Schuelke M, Seelow D (2014) MutationTaster2: mutation prediction for the deep-sequencing age. Nat Methods 11:361–362. https://doi.org/10.1038/nmeth.2890
Kumar P, Henikoff S, Ng PC (2009) Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat Protoc 4:1073–1081. https://doi.org/10.1038/nprot.2009.86
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, Kondrashov AS, Sunyaev SR (2010) A method and server for predicting damaging missense mutations. Nat Methods 7:248–249. https://doi.org/10.1038/nmeth0410-248
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, Voelkerding K, Rehm HL, Committee ALQA (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American college of medical genetics and genomics and the association for molecular pathology. Genet Med 17:405–424. https://doi.org/10.1038/gim.2015.30
Piaceri I, Del Mastio M, Tedde A, Bagnoli S, Latorraca S, Massaro F, Paganini M, Corrado A, Sorbi S, Nacmias B (2012) Clinical heterogeneity in Italian patients with amyotrophic lateral sclerosis. Clin Genet 82:83–87. https://doi.org/10.1111/j.1399-0004.2011.01726.x
Williams KL, Durnall JC, Thoeng AD, Warraich ST, Nicholson GA, Blair IP (2009) A novel TARDBP mutation in an Australian amyotrophic lateral sclerosis kindred. J Neurol Neurosurg Psychiatry 80:1286–1288. https://doi.org/10.1136/jnnp.2008.163261
Kreiter N, Pal A, Lojewski X, Corcia P, Naujock M, Reinhardt P, Sterneckert J, Petri S, Wegner F, Storch A, Hermann A (2018) Age-dependent neurodegeneration and organelle transport deficiencies in mutant TDP43 patient-derived neurons are independent of TDP43 aggregation. Neurobiol Dis 115:167–181. https://doi.org/10.1016/j.nbd.2018.03.010
Lin J, Chen W, Huang P, Xie Y, Zheng M, Yao X (2021) The distinct manifestation of young-onset amyotrophic lateral sclerosis in China. Amyotroph Lateral Scler Frontotemporal Degener 22:30–37. https://doi.org/10.1080/21678421.2020.1797091
Liu ZJ, Lin HX, Liu GL, Tao QQ, Ni W, Xiao BG, Wu ZY (2017) The investigation of genetic and clinical features in Chinese patients with juvenile amyotrophic lateral sclerosis. Clin Genet 92:267–273. https://doi.org/10.1111/cge.13015
Liu ZJ, Lin HX, Wei Q, Zhang QJ, Chen CX, Tao QQ, Liu GL, Ni W, Gitler AD, Li HF, Wu ZY (2019) Genetic spectrum and variability in Chinese patients with amyotrophic lateral sclerosis. Aging Dis 10:1199–1206. https://doi.org/10.4336/AD.2019.0215
Deng J, Wu W, Xie Z, Gang Q, Yu M, Liu J, Wang Q, Lv H, Zhang W, Huang Y, Wang T, Yuan Y, Hong D, Wang Z (2019) Novel and recurrent mutations in a cohort of Chinese patients with young-onset amyotrophic lateral sclerosis. Front Neurosci 13:1289. https://doi.org/10.3389/fnins.2019.01289
Wei Q, Chen X, Zheng Z, Guo X, Huang R, Cao B, Zeng Y, Shang H (2015) The predictors of survival in Chinese amyotrophic lateral sclerosis patients. Amyotroph Lateral Scler Frontotemporal Degener 16:237–244. https://doi.org/10.3109/21678421.2014.993650
Marin B, Logroscino G, Boumediene F, Labrunie A, Couratier P, Babron MC, Leutenegger AL, Preux PM, Beghi E (2016) Clinical and demographic factors and outcome of amyotrophic lateral sclerosis in relation to population ancestral origin. Eur J Epidemiol 31:229–245. https://doi.org/10.1007/s10654-015-0090-x
Xiong HL, Wang JY, Sun YM, Wu JJ, Chen Y, Qiao K, Zheng QJ, Zhao GX, Wu ZY (2010) Association between novel TARDBP mutations and Chinese patients with amyotrophic lateral sclerosis. BMC Med Genet 11:8. https://doi.org/10.1186/1471-2350-11-8
Ju X, Liu W, Li X, Liu N, Zhang N, Liu T, Deng M (2016) Two distinct clinical features and cognitive impairment in amyotrophic lateral sclerosis patients with TARDBP gene mutations in the Chinese population. Neurobiol Aging 38:216-e1-216. https://doi.org/10.1016/j.neurobiolaging.2015.10.032
Xu GR, Hu W, Zhan LL, Wang C, Xu LQ, Lin MT, Chen WJ, Wang N, Zhang QJ (2018) High frequency of the TARDBP pM337 V mutation among south-eastern Chinese patients with familial amyotrophic lateral sclerosis. BMC Neurol 18(1):1–6. https://doi.org/10.1186/s12883-018-1028-1
Gendron TF, Rademakers R, Petrucelli L (2013) TARDBP mutation analysis in TDP-43 proteinopathies and deciphering the toxicity of mutant TDP-43. J Alzheimers Dis 33(Suppl 1):S35-45. https://doi.org/10.3233/JAD-2012-129036
Corcia P, Valdmanis P, Millecamps S, Lionnet C, Blasco H, Mouzat K, Daoud H, Belzil V, Morales R, Pageot N, Danel-Brunaud V, Vandenberghe N, Pradat PF, Couratier P, Salachas F, Lumbroso S, Rouleau GA, Meininger V, Camu W (2012) Phenotype and genotype analysis in amyotrophic lateral sclerosis with TARDBP gene mutations. Neurology 78:1519–1526. https://doi.org/10.1212/WNL.0b013e3182553c88
Rutherford NJ, Zhang YJ, Baker M, Gass JM, Finch NA, Xu YF, Stewart H, Kelley BJ, Kuntz K, Crook RJ, Sreedharan J, Vance C, Sorenson E, Lippa C, Bigio EH, Geschwind DH, Knopman DS, Mitsumoto H, Petersen RC, Cashman NR, Hutton M, Shaw CE, Boylan KB, Boeve B, Graff-Radford NR, Wszolek ZK, Caselli RJ, Dickson DW, Mackenzie IR, Petrucelli L, Rademakers R (2008) Novel mutations in TARDBP (TDP-43) in patients with familial amyotrophic lateral sclerosis. PLoS Genet 4:e1000193. https://doi.org/10.1371/journal.pgen.1000193
Wang F, Fu S, Lei J, Wu H, Shi S, Chen K, Hu J, Xu X (2020) Identification of novel FUS and TARDBP gene mutations in Chinese amyotrophic lateral sclerosis patients with HRM analysis. Aging 12:22859–22868. https://doi.org/10.8632/aging.103967
Guennoc AM, Heuze-Vourc’h N, Gordon PH, Courty Y, Vourc’h P, Andres CR, Corcia P (2013) Benign lower limb amyotrophy due to TARDBP mutation or post-polio syndrome? Amyotroph Lateral Scler Frontotemporal Degener 14:476–478. https://doi.org/10.3109/21678421.2013.764567
Tunca C, Seker T, Akcimen F, Coskun C, Bayraktar E, Palvadeau R, Zor S, Kocoglu C, Kartal E, Sen NE, Hamzeiy H, Ozoguz Erimis A, Norman U, Karakahya O, Olgun G, Akgun T, Durmus H, Sahin E, Cakar A, Basar Gursoy E, Babacan Yildiz G, Isak B, Uluc K, Hanagasi H, Bilgic B, Turgut N, Aysal F, Ertas M, Boz C, Kotan D, Idrisoglu H, Soysal A, Uzun Adatepe N, Akalin MA, Koc F, Tan E, Oflazer P, Deymeer F, Tastan O, Cicek AE, Kavak E, Parman Y, Basak AN (2020) Revisiting the complex architecture of ALS in Turkey: expanding genotypes, shared phenotypes, molecular networks, and a public variant database. Hum Mutat 41:e7–e45. https://doi.org/10.1002/humu.24055
Theunissen F, Anderton RS, Mastaglia FL, Flynn LL, Winter SJ, James I, Bedlack R, Hodgetts S, Fletcher S, Wilton SD, Laing NG, MacShane M, Needham M, Saunders A, Mackay-Sim A, Melamed Z, Ravits J, Cleveland DW, Akkari PA (2021) Novel STMN2 variant linked to amyotrophic lateral sclerosis risk and clinical phenotype. Front Aging Neurosci 13:658226. https://doi.org/10.3389/fnagi.2021.658226
Ji AL, Zhang X, Chen WW, Huang WJ (2017) Genetics insight into the amyotrophic lateral sclerosis/frontotemporal dementia spectrum. J Med Genet 54:145–154. https://doi.org/10.1136/jmedgenet-2016-104271
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Bruce J, Schuck T, Grossman M, Clark CM, McCluskey LF, Miller BL, Masliah E, Mackenzie IR, Feldman H, Feiden W, Kretzschmar HA, Trojanowski JQ, Lee VM (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314:130–133. https://doi.org/10.1126/science.1134108
Suk TR, Rousseaux MWC (2020) The role of TDP-43 mislocalization in amyotrophic lateral sclerosis. Mol Neurodegener 15:45. https://doi.org/10.1186/s13024-020-00397-1
Kim G, Gautier O, Tassoni-Tsuchida E, Ma XR, Gitler AD (2020) ALS genetics: gains, losses, and implications for future therapies. Neuron 108:822–842. https://doi.org/10.1016/j.neuron.2020.08.022
Sun CS, Wang CY, Chen BP, He RY, Liu GC, Wang CH, Chen W, Chern Y, Huang JJ (2014) The influence of pathological mutations and proline substitutions in TDP-43 glycine-rich peptides on its amyloid properties and cellular toxicity. PLoS ONE 9:e103644. https://doi.org/10.1371/journal.pone.0103644
Acknowledgements
We thank the support of patients and their families. We also thank Miss Chen Wang for her great assistance in sample collecting and management.
Funding
This work was financially supported by the Strategic Priority Research Program of the Chinese Academy of Sciences “Biological basis of aging and therapeutic strategies” (Grant number: XDB39040100), the CAMS Innovation Fund for Medical Sciences (CIFMS) grant (Grant Number: 2016-I2M-1-002, 2021-1-I2M-018, and 2021-1-I2M-034), the National Natural Science Foundation of China (Grant Number: 81971293, 81788101), the National Key Research and Development Program of China (Grant Number 2016 YFC0905100).
Author information
Authors and Affiliations
Contributions
JL: conceptualization, methodology, software, writing—original draft preparation. QL: conceptualization, writing—reviewing and editing. XS: data curation, investigation. KZ: data curation, visualization. SL: data curation, software. ZW: data curation, software. XY: investigation, validation. MSL: supervision, validation. LC: conceptualization, writing—reviewing and editing. XZ: supervision, validation.
Corresponding authors
Ethics declarations
Conflicts of interest
None.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, J., Liu, Q., Sun, X. et al. Genotype–phenotype association of TARDBP mutations in Chinese patients with amyotrophic lateral sclerosis: a single-center study and systematic review of published literature. J Neurol 269, 4204–4212 (2022). https://doi.org/10.1007/s00415-022-11042-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-022-11042-w