Abstract
Objectives
Antiviral treatments for Bell palsy have been widely used, but there is no definite conclusion of which is the most effective antiviral drug. We conducted a systematic review of randomized controlled trials (RCTs) including network meta-analysis to investigate the comparative effectiveness of antiviral treatments for Bell palsy.
Data
RCTs comparing effectiveness between antiviral treatments and placebo were included. Risk of bias within and across studies was assessed with the Cochrane tool and the GRADE approach, respectively. Random-effects pairwise meta-analyses were conducted, followed by network meta-analysis.
Sources
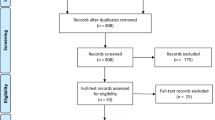
Three electronic databases were searched from inception to May 18, 2020.
Study selection
11 trials and 3393 patients with four arms and eleven contrasts were included.
Results
Significant differences were observed between placebo and famciclovir with respect to overall recovery and no statistically significant differences were found from other comparisons. Treatment ranking based on the evidence network indicated that famciclovir shared the best results, followed by valacyclovir, acyclovir, and finally placebo. Adverse events of famciclovir were too rare and slight to be analyzed. Our confidence in pairwise comparisons was moderate to low, due to study limitations, inconsistency, and imprecision; our confidence in ranking was moderate, due to study limitations. Inconsistency is not deemed to exist by a loop-specific approach and node-splitting procedure. Results of exploring publication bias are satisfying.
Conclusions
According to pairwise and network comparisons, famciclovir could be better than placebo and the effectiveness of other antiviral treatments are similar. For clinical efficacy, famciclovir obtains the best recovery rate of facial function for Bell palsy. Acyclovir has the lowest rate of synkinesis, though, it is not adequately recommended and more superior trails are needed in the future.













Similar content being viewed by others
Change history
01 February 2022
A Correction to this paper has been published: https://doi.org/10.1007/s00415-022-10976-5
References
Pereira LM, Obara K, Dias JM, Menacho MO, Lavado EL, Cardoso JR (2011) Facial exercise therapy for facial palsy: systematic review and meta-analysis. Clin Rehabil 25(7):649–658. https://doi.org/10.1177/0269215510395634
Peitersen E (2002) Bell’s palsy: the spontaneous course of 2,500 peripheral facial nerve palsies of different etiologies. Acta Otolaryngol Suppl 549:4–30
Gilden DH (2004) Clinical practice. Bell’s Palsy N Engl J Med 351(13):1323–1331. https://doi.org/10.1056/NEJMcp041120
Adour KK, Byl FM, Hilsinger RL Jr, Kahn ZM, Sheldon MI (1978) The true nature of Bell’s palsy: analysis of 1000 consecutive patients. Laryngoscope 88(5):787–801. https://doi.org/10.1002/lary.1978.88.5.787
Berg T, Axelsson S, Engström M, Stjernquist-Desatnik A, Pitkäranta A, Kanerva M, Jonsson L (2009) The course of pain in Bell’s palsy: treatment with prednisolone and valacyclovir. Otol Neurotol 30(6):842–846. https://doi.org/10.1097/MAO.0b013e3181ab31c8
Teixeira LJ, Valbuza JS, Prado GF (2011) Physical therapy for Bell’s palsy (idiopathic facial paralysis). Cochrane Database Syst Rev 12:006283. https://doi.org/10.1002/14651858.CD006283.pub3
Peitersen E (1992) Natural history of Bell’s palsy. Acta Otolaryngol Suppl 492:122–124. https://doi.org/10.3109/00016489209136829
Engström M, Berg T, Stjernquist-Desatnik A, Axelsson S, Pitkäranta A, Hultcrantz M, Kanerva M, Hanner P, Jonsson L (2008) Prednisolone and valaciclovir in Bell’s palsy: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet Neurol 7(11):993–1000. https://doi.org/10.1016/s1474-4422(08)70221-7
Lockhart P, Daly F, Pitkethly M, Comerford N, Sullivan F (2009) Antiviral treatment for Bell’s palsy (idiopathic facial paralysis). Cochrane Database Syst Rev 4:cd001869. https://doi.org/10.1002/14651858.CD001869.pub4
Salinas RA, Alvarez G, Daly F, Ferreira J (2010) Corticosteroids for Bell’s palsy (idiopathic facial paralysis). Cochrane Database Syst Rev 3:cd001942. https://doi.org/10.1002/14651858.CD001942.pub4
Linder TE, Abdelkafy W, Cavero-Vanek S (2010) The management of peripheral facial nerve palsy: “paresis” versus “paralysis” and sources of ambiguity in study designs. Otol Neurotol 31(2):319–327. https://doi.org/10.1097/MAO.0b013e3181cabd90
Heckmann JG, Urban PP, Pitz S, Guntinas-Lichius O, Gágyor I (2019) The Diagnosis and treatment of idiopathic facial paresis (Bell’s Palsy). Dtsch Arztebl Int 116(41):692–702. https://doi.org/10.3238/arztebl.2019.0692
Baugh RF, Basura GJ, Ishii LE, Schwartz SR, Drumheller CM, Burkholder R, Deckard NA, Dawson C, Driscoll C, Gillespie MB, Gurgel RK, Halperin J, Khalid AN, Kumar KA, Micco A, Munsell D, Rosenbaum S, Vaughan W (2013) Clinical practice guideline: Bell’s palsy. Otolaryngol Head Neck Surg 149(3 Suppl):S1-27. https://doi.org/10.1177/0194599813505967
Gagyor I, Madhok VB, Daly F, Somasundara D, Sullivan M, Gammie F, Sullivan F (2015) Antiviral treatment for Bell’s palsy (idiopathic facial paralysis). Cochrane Database Syst Rev 11:Cd001869. https://doi.org/10.1002/14651858.CD001869.pub8
Fu X, Tang L, Wang C, Li M, Wu H, Li J, Ma Q, Yang W (2018) A Network Meta-analysis to compare the efficacy of steroid and antiviral medications for facial paralysis from Bell´s Palsy. Pain Physician 21(6):559–569
Zhang R, Wu T, Wang R, Wang D, Liu Q (2019) Compare the efficacy of acupuncture with drugs in the treatment of Bell’s palsy: a systematic review and meta-analysis of RCTs. Medicine (Baltimore) 98(19):e15566. https://doi.org/10.1097/md.0000000000015566
Agostini F, Mangone M, Santilli V, Paoloni M, Bernetti A, Saggini R, Paolucci T (2020) Idiopathic facial palsy: umbrella review of systematic reviews and meta-analyses. J Biol Regul Homeost Agents 34(4):1245–1255. https://doi.org/10.23812/20-339-a
Kim SJ, Lee HY (2020) Acute peripheral facial palsy: recent guidelines and a systematic review of the literature. J Korean Med Sci 35(30):e245. https://doi.org/10.3346/jkms.2020.35.e245
Shamseer L, Moher D, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P, Stewart LA (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ 350:g7647. https://doi.org/10.1136/bmj.g7647
Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, Thomas J (2019) Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for systematic reviews of interventions. Cochrane Database Syst Rev 10:Ed00042. https://doi.org/10.1002/14651858.Ed000142
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors) (2020) Cochrane Handbook for Systematic Reviews of Interventions version 6.1 (Updated September 2020). Cochrane. www.training.cochrane.org/handbook
Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, Ioannidis JP, Straus S, Thorlund K, Jansen JP, Mulrow C, Catalá-López F, Gøtzsche PC, Dickersin K, Boutron I, Altman DG, Moher D (2015) The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 162(11):777–784. https://doi.org/10.7326/m14-2385
Stegenga B, Dijkstra PU (2007) Principles of evidence-based medicine applied to dentistry. Ned Tijdschr Tandheelkd 114(4):155–160
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. https://doi.org/10.1136/bmj.d5928
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898. https://doi.org/10.1136/bmj.l4898
Higgins JP, Jackson D, Barrett JK, Lu G, Ades AE, White IR (2012) Consistency and inconsistency in network meta-analysis: concepts and models for multi-arm studies. Res Synth Methods 3(2):98–110. https://doi.org/10.1002/jrsm.1044
Lu G, Ades AE (2004) Combination of direct and indirect evidence in mixed treatment comparisons. Stat Med 23(20):3105–3124. https://doi.org/10.1002/sim.1875
Salanti G, Higgins JP, Ades AE, Ioannidis JP (2008) Evaluation of networks of randomized trials. Stat Methods Med Res 17(3):279–301. https://doi.org/10.1177/0962280207080643
Chaimani A, Higgins JP, Mavridis D, Spyridonos P, Salanti G (2013) Graphical tools for network meta-analysis in STATA. PLoS ONE 8(10):e76654. https://doi.org/10.1371/journal.pone.0076654
Salanti G, Ades AE, Ioannidis JP (2011) Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 64(2):163–171. https://doi.org/10.1016/j.jclinepi.2010.03.016
Mavridis D, Giannatsi M, Cipriani A, Salanti G (2015) A primer on network meta-analysis with emphasis on mental health. Evid Based Ment Health 18(2):40–46. https://doi.org/10.1136/eb-2015-102088
Salanti G, Del Giovane C, Chaimani A, Caldwell DM, Higgins JP (2014) Evaluating the quality of evidence from a network meta-analysis. PLoS ONE 9(7):e99682. https://doi.org/10.1371/journal.pone.0099682
Mubareka S, Leung V, Aoki FY, Vinh DC (2010) Famciclovir: a focus on efficacy and safety. Expert Opin Drug Saf 9(4):643–658. https://doi.org/10.1517/14740338.2010.485189
Semaan JR, Parmar M (2020) Famciclovir. StatPearls Publishing, StatPearls
Copyright © 2020, StatPearls Publishing LLC., Treasure Island (FL),
Earnshaw DL, Bacon TH, Darlison SJ, Edmonds K, Perkins RM, Vere Hodge RA (1992) Mode of antiviral action of penciclovir in MRC-5 cells infected with herpes simplex virus type 1 (HSV-1), HSV-2, and varicella-zoster virus. Antimicrob Agents Chemother 36(12):2747–2757. https://doi.org/10.1128/aac.36.12.2747
Hodge RA, Perkins RM (1989) Mode of action of 9-(4-hydroxy-3-hydroxymethylbut-1-yl)guanine (BRL 39123) against herpes simplex virus in MRC-5 cells. Antimicrob Agents Chemother 33(2):223–229. https://doi.org/10.1128/aac.33.2.223
Perry CM, Wagstaff AJ (1995) Famciclovir. A review of its pharmacological properties and therapeutic efficacy in herpesvirus infections. Drugs 50(2):396–415. https://doi.org/10.2165/00003495-199550020-00011
Thackray AM, Field HJ (1996) Differential effects of famciclovir and valaciclovir on the pathogenesis of herpes simplex virus in a murine infection model including reactivation from latency. J Infect Dis 173(2):291–299. https://doi.org/10.1093/infdis/173.2.291
Thackray AM, Field HJ (1998) Famciclovir and valaciclovir differ in the prevention of herpes simplex virus type 1 latency in mice: a quantitative study. Antimicrob Agents Chemother 42(7):1555–1562. https://doi.org/10.1128/aac.42.7.1555
Field HJ, Tewari D, Sutton D, Thackray AM (1995) Comparison of efficacies of famciclovir and valaciclovir against herpes simplex virus type 1 in a murine immunosuppression model. Antimicrob Agents Chemother 39(5):1114–1119. https://doi.org/10.1128/aac.39.5.1114
Loutsch JM, Sainz B Jr, Marquart ME, Zheng X, Kesavan P, Higaki S, Hill JM, Tal-Singer R (2001) Effect of famciclovir on herpes simplex virus type 1 corneal disease and establishment of latency in rabbits. Antimicrob Agents Chemother 45(7):2044–2053. https://doi.org/10.1128/aac.45.7.2044-2053.2001
Pott Junior H, de Oliveira MFB, Gambero S, Amazonas RB (2018) Randomized clinical trial of famciclovir or acyclovir for the treatment of herpes zoster in adults. Int J Infect Dis 72:11–15. https://doi.org/10.1016/j.ijid.2018.04.4324
Yaldiz M, Solak B, Kara RO, Cosansu N, Erdem MT (2018) Comparison of Famciclovir, Valaciclovir, and Brivudine Treatments in Adult Immunocompetent Patients With Herpes Zoster. Am J Ther 25(6):e626–e634. https://doi.org/10.1097/mjt.0000000000000436
Minnerop M, Herbst M, Fimmers R, Kaabar P, Matz B, Klockgether T, Wüllner U (2008) Bell’s palsy: combined treatment of famciclovir and prednisone is superior to prednisone alone. J Neurol 255(11):1726–1730. https://doi.org/10.1007/s00415-008-0008-6
Kawaguchi K, Inamura H, Abe Y, Koshu H, Takashita E, Muraki Y, Matsuzaki Y, Nishimura H, Ishikawa H, Fukao A et al (2007) Reactivation of herpes simplex virus type 1 and varicella-zoster virus and therapeutic effects of combination therapy with prednisolone and valacyclovir in patients with Bell’s palsy. Laryngoscope 117(1):147–156. https://doi.org/10.1097/01.mlg.0000248737.65607.9e
Kang HM, Jung SY, Byun JY, Park MS, Yeo SG (2015) Steroid plus antiviral treatment for Bell’s palsy. J Intern Med 277(5):532–539
Funding
The article was supported by “345 Talent Project of Shengjing Hospital”.
Author information
Authors and Affiliations
Contributions
Jiajie Cao contributed to the majority of the writing and performed the statistics; Xinyue Zhang provided data; Zhiming Wang edited the article.
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
The original online version of this article was revised: The Funding information section was missing.
Rights and permissions
About this article
Cite this article
Cao, J., Zhang, X. & Wang, Z. Effectiveness comparisons of antiviral treatments for Bell palsy: a systematic review and network meta-analysis. J Neurol 269, 1353–1367 (2022). https://doi.org/10.1007/s00415-021-10487-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10487-9