Abstract
Background
Persons with multiple sclerosis (PwMS) are at an elevated risk of depression. Decreased Conscientiousness may affect patient outcomes in PwMS. Low Conscientiousness has a strong correlation with depression. Previous work has also reported that white matter (WM) tract disruption in frontal–parietal networks explains reduced Conscientiousness in PwMS.
Objective
We hypothesized that Conscientiousness-associated WM tract disruption predicts new-onset depression over 5 years in PwMS and evaluated this by assessing the predictive power of mean Conscientiousness associated frontal–parietal network (CFPN) disruption in PwMS for clinically diagnosed depression over 5 years.
Methods
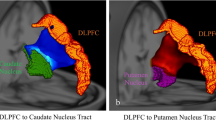
This longitudinal retrospective analysis included 53 PwMS who were not previously diagnosed as depressed. All participants underwent structural MRI. Medical records were reviewed to evaluate diagnosis of depression for these patients over 5 years. WM tract damage between pairs of gray matter regions in the CFPN was measured using diffusion imaging. The relationship between CFPN disruption and depression was analyzed using logistic regression.
Results
Participants with MS had a mean age of 46.0 years (SD = 11.2). 22.6% (n = 12) acquired a diagnosis of clinical depression over the 5-year period. Baseline disruption in the CFPN was a significant predictor (ROC AUC = 61.8%). of new-onset clinical depression, accounting for age, sex, lateral ventricular volume, disease modifying treatment, and lesion volume.
Conclusion
Baseline CFPN disruption is associated with progression to clinical depression over 5 years in PwMS. Development of new WM pathology within this network may be a risk factor for depression.




Similar content being viewed by others
Data availability
Anonymized data will be shared by request from any qualified investigator.
Code availability
All custom code complies with field standards and will be shared by request from any qualified investigator.
References
Kaye J, Morton J, Bowcutt M, Maupin D (2000) Depression: the forgotten diagnosis among hospitalized adults. J Neurosci Nurs 32(1):7
Benedict RH, Wahlig E, Bakshi R, Fishman I, Munschauer F, Zivadinov R, Weinstock-Guttman B (2005) Predicting quality of life in multiple sclerosis: accounting for physical disability, fatigue, cognition, mood disorder, personality, and behavior change. J Neurol Sci 231(1–2):29–34. https://doi.org/10.1016/j.jns.2004.12.009
Siegert RJ, Abernethy DA (2005) Depression in multiple sclerosis: a review. J Neurol Neurosurg Psychiatry 76(4):469–475. https://doi.org/10.1136/jnnp.2004.054635
Benedict RHB, Priore RL, Miller C, Munschauer F, Jacobs L (2001) Personality disorder in multiple sclerosis correlates with cognitive impairment. J Neuropsychiatry Clin Neurosci 13(1):70–76. https://doi.org/10.1176/appi.neuropsych.13.1.70
McCrae RR, John OP (1992) An introduction to the five-factor model and its applications. J Pers 60(2):175–215. https://doi.org/10.1111/j.1467-6494.1992.tb00970.x
Bruce JM, Lynch SG (2011) Personality traits in multiple sclerosis: association with mood and anxiety disorders. J Psychosom Res 70(5):479–485. https://doi.org/10.1016/j.jpsychores.2010.12.010
Klein DN, Kotov R, Bufferd SJ (2011) Personality and depression: explanatory models and review of the evidence. Annu Rev Clin Psychol 7:269–295. https://doi.org/10.1146/annurev-clinpsy-032210-104540
Fuchs TA, Dwyer MG, Kuceyeski A, Choudhery S, Carolus K, Li X, Mallory M, Weinstock-Guttman B, Jakimovski D, Ramasamy D, Zivadinov R, Benedict RHB (2018) White matter tract network disruption explains reduced conscientiousness in multiple sclerosis. Hum Brain Mapp 39(9):3682–3690. https://doi.org/10.1002/hbm.24203
Chow PI, Roberts BW (2014) Examining the relationship between changes in personality and changes in depression. J Res Pers 51:38–46. https://doi.org/10.1016/j.jrp.2014.04.007
Costa PT, Mac Crae RR (1992) Neo personality inventory-revised (NEO PI-R). Psychological Assessment Resources, Odessa
Zivadinov R, Ramasamy DP, Benedict RR, Polak P, Hagemeier J, Magnano C, Dwyer MG, Bergsland N, Bertolino N, Weinstock-Guttman B (2016) Cerebral microbleeds in multiple sclerosis evaluated on susceptibility-weighted images and quantitative susceptibility maps: a case-control study. Radiology 281(3):884–895
Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, Fujihara K, Havrdova E, Hutchinson M, Kappos L, Lublin FD, Montalban X, O'Connor P, Sandberg-Wollheim M, Thompson AJ, Waubant E, Weinshenker B, Wolinsky JS (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69(2):292–302. https://doi.org/10.1002/ana.22366
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33(11):1444–1452. https://doi.org/10.1212/wnl.33.11.1444
Gelineau-Morel R, Tomassini V, Jenkinson M, Johansen-Berg H, Matthews PM, Palace J (2012) The effect of hypointense white matter lesions on automated gray matter segmentation in multiple sclerosis. Hum Brain Mapp 33(12):2802–2814. https://doi.org/10.1002/hbm.21402
Smith SM, Zhang Y, Jenkinson M, Chen J, Matthews PM, Federico A, De Stefano N (2002) Accurate, robust, and automated longitudinal and cross-sectional brain change analysis. Neuroimage 17(1):479–489. https://doi.org/10.1006/nimg.2002.1040
Zivadinov R, Heininen-Brown M, Schirda CV, Poloni GU, Bergsland N, Magnano CR, Durfee J, Kennedy C, Carl E, Hagemeier J, Benedict RH, Weinstock-Guttman B, Dwyer MG (2012) Abnormal subcortical deep-gray matter susceptibility-weighted imaging filtered phase measurements in patients with multiple sclerosis: a case-control study. Neuroimage 59(1):331–339. https://doi.org/10.1016/j.neuroimage.2011.07.045
Avants BB, Tustison NJ, Song G, Cook PA, Klein A, Gee JC (2011) A reproducible evaluation of ANTs similarity metric performance in brain image registration. Neuroimage 54(3):2033–2044. https://doi.org/10.1016/j.neuroimage.2010.09.025
Andersson JL, Sotiropoulos SN (2016) An integrated approach to correction for off-resonance effects and subject movement in diffusion MR imaging. Neuroimage 125:1063–1078
Jenkinson M, Beckmann CF, Behrens TE, Woolrich MW, Smith SM (2012) Fsl. Neuroimage 62(2):782–790. https://doi.org/10.1016/j.neuroimage.2011.09.015
Dale AM, Fischl B, Sereno MI (1999) Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage 9(2):179–194. https://doi.org/10.1006/nimg.1998.0395
Kuceyeski A, Maruta J, Relkin N, Raj A (2013) The network modification (NeMo) tool: elucidating the effect of white matter integrity changes on cortical and subcortical structural connectivity. Brain Connect 3(5):451–463. https://doi.org/10.1089/brain.2013.0147
Fuchs TA, Benedict RH, Wilding G, Wojcik C, Jakimovski D, Bergsland N, Ramasamy DP, Weinstock-Guttman B, Zivadinov R, Dwyer MG (2019) Trait conscientiousness predicts rate of brain atrophy in multiple sclerosis. Mult Scler. https://doi.org/10.1177/1352458519858605
Biver F, Goldman S, Delvenne V, Luxen A, Demaertelaer V, Hubain P, Mendlewicz J, Lotstra F (1994) Frontal and parietal metabolic disturbances in unipolar depression. Biol Psychiatry 36(6):381–388. https://doi.org/10.1016/0006-3223(94)91213-0
Mayberg HS (1994) Frontal lobe dysfunction in secondary depression. J Neuropsychiatry Clin Neurosci 6(4):428–442. https://doi.org/10.1176/jnp.6.4.428
Wen H, Liu Y, Rekik I, Wang S, Zhang J, Zhang Y, Peng Y, He H (2017) Disrupted topological organization of structural networks revealed by probabilistic diffusion tractography in Tourette syndrome children. Hum Brain Mapp 38(8):3988–4008. https://doi.org/10.1002/hbm.23643
Eapen V, Fox-Hiley P, Banerjee S, Robertson M (2004) Clinical features and associated psychopathology in a Tourette syndrome cohort. Acta Neurol Scand 109(4):255–260. https://doi.org/10.1046/j.1600-0404.2003.00228.x
Castellanos FX, Proal E (2012) Large-scale brain systems in ADHD: beyond the prefrontal-striatal model. Trends Cogn Sci 16(1):17–26. https://doi.org/10.1016/j.tics.2011.11.007
Connor DF, Edwards G, Fletcher KE, Baird J, Barkley RA, Steingard RJ (2003) Correlates of comorbid psychopathology in children with ADHD. J Am Acad Child Adolesc Psychiatry 42(2):193–200. https://doi.org/10.1097/00004583-200302000-00013
Lin HY, Perry A, Cocchi L, Roberts JA, Tseng WI, Breakspear M, Gau SS (2019) Development of frontoparietal connectivity predicts longitudinal symptom changes in young people with autism spectrum disorder. Transl Psychiatry 9(1):86. https://doi.org/10.1038/s41398-019-0418-5
Hollocks MJ, Lerh JW, Magiati I, Meiser-Stedman R, Brugha TS (2019) Anxiety and depression in adults with autism spectrum disorder: a systematic review and meta-analysis. Psychol Med 49(4):559–572. https://doi.org/10.1017/S0033291718002283
Eddy CM, Rickards HE, Critchley HD, Cavanna AE (2013) A controlled study of personality and affect in Tourette syndrome. Compr Psychiatry 54(2):105–110. https://doi.org/10.1016/j.comppsych.2012.07.004
Stanton K, Watson D (2016) Adult ADHD: associations with personality and other psychopathology. J Psychopathol Behav Assess 38(2):195–208. https://doi.org/10.1007/s10862-015-9519-5
Lodi-Smith J, Rodgers JD, Cunningham SA, Lopata C, Thomeer ML (2019) Meta-analysis of Big Five personality traits in autism spectrum disorder. Autism 23(3):556–565. https://doi.org/10.1177/1362361318766571
Rao V, Bertrand M, Rosenberg P, Makley M, Schretlen DJ, Brandt J, Mielke MM (2010) Predictors of new-onset depression after mild traumatic brain injury. J Neuropsychiatry Clin Neurosci 22(1):100–104. https://doi.org/10.1176/appi.neuropsych.22.1.100
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
KA, TF, and MGD contributed to the study conception and design. Material preparation and analysis were performed by KA, TF, DJ, NB, DR, CV, BWG and MGD. Data collection was performed by RZ, DJ, BWG, and RHBB. The first draft of the manuscript was written by KA, and all authors contributed to revising and drafting previous versions of this manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
Ralph H. B. Benedict has received research support from Accorda, Genzyme, Biogen, and Mallinckrodt, and is on the speakers’ bureau for EMD Serono, consults for Abbvie, Biogen, Genentech, Roche, Sanofi, Verasci, and Novartis, and receives royalties for Psychological Assessment Resources. Robert Zivadinov received personal compensation from Teva Pharmaceuticals, Biogen Idec, EMD Serono, Genzyme-Sanofi, Claret Medical, IMS Health and Novartis for speaking and consultant fees. He received financial support for research activities from Teva Pharmaceuticals, Genzyme-Sanofi, Novartis, Claret Medical, Intekrin and IMS Health. Michael G Dwyer received personal compensation from Novartis and Claret Medical for speaking and consultant fees. He received financial support for research activities from Novartis. Bianca Weinstock-Guttman received honoraria as a speaker and as a consultant for Biogen Idec, EMD Serono, Novartis and Mallinckrodt. Dr Weinstock-Guttman received research funds from Biogen Idec, Teva Pharmaceuticals, EMD Serono, Genzyme, Sanofi, Novartis. Caila B Vaughn has received personal compensation from Merck/EMD Serono for consultant fees. Tom Fuchs, Kira Ashton, Niels Bergsland, Dejan Jakimovski, Devon Oship, and Deepa Ramasamy have nothing to disclose.
Ethical standard statement
The study protocol was approved by the University and Buffalo Institutional Ethics Review Board, and was therefore performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
All participants’ written informed consent was obtained before participation.
Consent for publication
All participants’ written informed consent was obtained to publish their de-identified data before participation.
Rights and permissions
About this article
Cite this article
Ashton, K., Fuchs, T.A., Oship, D. et al. Diagnosis of depression in multiple sclerosis is predicted by frontal–parietal white matter tract disruption. J Neurol 268, 169–177 (2021). https://doi.org/10.1007/s00415-020-10110-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-10110-3