Abstract
Objectives
To report pitfalls in the clinical diagnosis of anti-N-methyl-d-aspartate receptor (NMDAR) encephalitis.
Methods
We retrospectively reviewed the clinical information of 221 patients with clinically suspected autoimmune neurological disorders who underwent testing for autoantibodies against neuronal cell-surface antigens between January 1, 2007 and September 10, 2017. Forty-one patients met the diagnostic criteria for probable anti-NMDAR encephalitis (probable criteria), but one was excluded because neither serum nor CSF was examined at the active stage. Thus, in 220 patients, sensitivity and specificity of the probable criteria were assessed.
Results
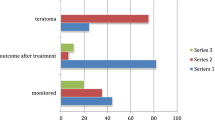
NMDAR-antibodies were detected in 34 of 40 patients (85%) with the probable criteria; however, 2 of the 6 antibody-negative patients had ovarian teratoma. The median age at onset was higher in antibody-negative patients than those with antibodies (49 vs. 27 years, p = 0.015). The age at onset was associated with the probability of antibody detection (p = 0.014); the probability was less than 50% in patients aged 50 years or older. NMDAR-antibodies were also detected in 5 of 180 patients who did not fulfill the probable criteria; these patients presented with isolated epileptic syndrome (n = 2), atypical demyelinating syndrome (n = 2; one with aquaporin 4 antibodies), and autoimmune post-herpes simplex encephalitis (post-HSE) (n = 1). Sensitivity and specificity of the probable criteria was 87.2 and 96.7%, respectively.
Conclusion
The probable criteria are valid, but the diversity of clinical phenotype should be taken into account in diagnosing anti-NMDAR encephalitis particularly in patients aged 50 years or older, or with isolated epileptic syndrome, atypical demyelinating syndrome, or post-HSE.


Similar content being viewed by others
References
Dalmau J, Tüzün E, Wu HY, Masjuan J, Rossi JE, Voloschin A, Baehring JM, Shimazaki H, Koide R, King D, Mason W, Sansing LH, Dichter MA, Rosenfeld MR, Lynch DR (2007) Paraneoplastic anti-N-methyl-d-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol 61:25–36. https://doi.org/10.1002/ana.21050
Lai M, Hughes EG, Peng X, Zhou L, Gleichman AJ, Shu H, Matà S, Kremens D, Vitaliani R, Geschwind MD, Bataller L, Kalb RG, Davis R, Graus F, Lynch DR, Balice-Gordon R, Dalmau J (2009) AMPA receptor antibodies in limbic encephalitis alter synaptic receptor location. Ann Neurol 65:424–434. https://doi.org/10.1002/ana.21589
Lancaster E, Lai M, Peng X, Hughes E, Constantinescu R, Raizer J, Friedman D, Skeen MB, Grisold W, Kimura A, Ohta K, Iizuka T, Guzman M, Graus F, Moss SJ, Balice-Gordon R, Dalmau J (2010) Antibodies to the GABA(B) receptor in limbic encephalitis with seizures: case series and characterisation of the antigen. Lancet Neurol 9:67–76. https://doi.org/10.1016/S1474-4422(09)70324-2
Lai M, Huijbers MG, Lancaster E, Graus F, Bataller L, Balice-Gordon R, Cowell JK, Dalmau J (2010) Investigation of LGI1 as the antigen in limbic encephalitis previously attributed to potassium channels: a case series. Lancet Neurol 9:776–785. https://doi.org/10.1016/S1474-4422(10)70137-X
Lancaster E, Huijbers MG, Bar V, Boronat A, Wong A, Martinez-Hernandez E, Wilson C, Jacobs D, Lai M, Walker RW, Graus F, Bataller L, Illa I, Markx S, Strauss KA, Peles E, Scherer SS, Dalmau J (2011) Investigations of caspr2, an autoantigen of encephalitis and neuromyotonia. Ann Neurol 69:303–311. https://doi.org/10.1002/ana.22297
Lancaster E, Martinez-Hernandez E, Titulaer MJ, Boulos M, Weaver S, Antoine JC, Liebers E, Kornblum C, Bien CG, Honnorat J, Wong S, Xu J, Contractor A, Balice-Gordon R, Dalmau J (2011) Antibodies to metabotropic glutamate receptor 5 in the Ophelia syndrome. Neurology 77:1698–1701. https://doi.org/10.1212/WNL.0b013e3182364a44
Boronat A, Gelfand JM, Gresa-Arribas N, Jeong HY, Walsh M, Roberts K, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R, Graus F, Rudy B, Dalmau J (2013) Encephalitis and antibodies to dipeptidyl-peptidase-like protein-6, a subunit of Kv4.2 potassium channels. Ann Neurol 73:120–128. https://doi.org/10.1002/ana.23756
Petit-Pedrol M, Armangue T, Peng X, Bataller L, Cellucci T, Davis R, McCracken L, Martinez-Hernandez E, Mason WP, Kruer MC, Ritacco DG, Grisold W, Meaney BF, Alcalá C, Sillevis-Smitt P, Titulaer MJ, Balice-Gordon R, Graus F, Dalmau J (2014) Encephalitis with refractory seizures, status epilepticus, and antibodies to the GABAA receptor: a case series, characterisation of the antigen, and analysis of the effects of antibodies. Lancet Neurol 13:276–286. https://doi.org/10.1016/S1474-4422(13)70299-0
Gresa-Arribas N, Planagumà J, Petit-Pedrol M, Kawachi I, Katada S, Glaser CA, Simabukuro MM, Armangué T, Martínez-Hernández E, Graus F, Dalmau J (2016) Human neurexin-3α antibodies associate with encephalitis and alter synapse development. Neurology 86:2235–2242. https://doi.org/10.1212/WNL.0000000000002775
Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T, Cortese I, Dale RC, Gelfand JM, Geschwind M, Glaser CA, Honnorat J, Höftberger R, Iizuka T, Irani SR, Lancaster E, Leypoldt F, Prüss H, Rae-Grant A, Reindl M, Rosenfeld MR, Rostásy K, Saiz A, Venkatesan A, Vincent A, Wandinger KP, Waters P, Dalmau J (2016) A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol 15:391–404. https://doi.org/10.1016/S1474-4422(15)00401-9
Titulaer MJ, Höftberger R, Iizuka T, Leypoldt F, McCracken L, Cellucci T, Benson LA, Shu H, Irioka T, Hirano M, Singh G, Cobo Calvo A, Kaida K, Morales PS, Wirtz PW, Yamamoto T, Reindl M, Rosenfeld MR, Graus F, Saiz A, Dalmau J (2014) Overlapping demyelinating syndromes and anti-N-methyl-d-aspartate receptor encephalitis. Ann Neurol 75:411–428. https://doi.org/10.1002/ana.24117
Taoka T, Yuh WT, White ML, Quets JP, Maley JE, Ueda T (2001) Sulcal hyperintensity on fluid-attenuated inversion recovery mr images in patients without apparent cerebrospinal fluid abnormality. AJR Am J Roentgenol 176:519–524. https://doi.org/10.2214/ajr.176.2.1760519
Armangue T, Moris G, Cantarín-Extremera V, Conde CE, Rostasy K, Erro ME, Portilla-Cuenca JC, Turón-Viñas E, Málaga I, Muñoz-Cabello B, Torres-Torres C, Llufriu S, González-Gutiérrez-Solana L, González G, Casado-Naranjo I, Rosenfeld M, Graus F, Dalmau J (2015) Autoimmune post-herpes simplex encephalitis of adults and teenagers. Neurology 85:1736–1743. https://doi.org/10.1212/WNL.0000000000002125
Ito S, Mori M, Makino T, Hayakawa S, Kuwabara S (2009) “Cloud-like enhancement” is a magnetic resonance imaging abnormality specific to neuromyelitis optica. Ann Neurol 66:425–428. https://doi.org/10.1002/ana.21753
Zandi MS, Irani SR, Lang B, Waters P, Jones PB, McKenna P, Coles AJ, Vincent A, Lennox BR (2011) Disease-relevant autoantibodies in first episode schizophrenia. J Neurol 258:686–688. https://doi.org/10.1007/s00415-010-5788-9
Niehusmann P, Dalmau J, Rudlowski C, Vincent A, Elger CE, Rossi JE, Bien CG (2009) Diagnostic value of N-methyl-d-aspartate receptor antibodies in women with new-onset epilepsy. Arch Neurol 66:458–464. https://doi.org/10.1001/archneurol.2009.5
Iizuka T, Sakai F, Ide T, Monzen T, Yoshii S, Iigaya M, Suzuki K, Lynch DR, Suzuki N, Hata T, Dalmau J (2008) Anti-NMDA receptor encephalitis in Japan: long-term outcome without tumor removal. Neurology 70:504–511. https://doi.org/10.1212/01.wnl.0000278388.90370.c3
Dalmau J, Gleichman AJ, Hughes EG, Rossi JE, Peng X, Lai M, Dessain SK, Rosenfeld MR, Balice-Gordon R, Lynch DR (2008) Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol 7:1091–1098. https://doi.org/10.1016/S1474-4422(08)70224-2
Kayser MS, Dalmau J (2011) Anti-NMDA receptor encephalitis in psychiatry. Curr Psychiatry Rev 7:189–193. https://doi.org/10.2174/157340011797183184
Titulaer MJ, McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, Honig LS, Benseler SM, Kawachi I, Martinez-Hernandez E, Aguilar E, Gresa-Arribas N, Ryan-Florance N, Torrents A, Saiz A, Rosenfeld MR, Balice-Gordon R, Graus F, Dalmau J (2013) Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol 12:157–165. https://doi.org/10.1016/S1474-4422(12)70310-1
Iizuka T, Kanazawa N, Kaneko J, Tominaga N, Nonoda Y, Hara A, Onozawa Y, Asari H, Hata T, Kaneko J, Yoshida K, Sugiura Y, Ugawa Y, Watanabe M, Tomita H, Kosakai A, Kaneko A, Ishima D, Kitamura E, Nishiyama K (2017) Cryptogenic NORSE: its distinctive clinical features and response to immunotherapy. Neurol Neuroimmunol Neuroinflamm 4:e396. https://doi.org/10.1212/NXI.0000000000000396
Armangue T, Martínez-Hernández E, Graus F, Dalmau J (2017) Brain autoimmunity following herpes simplex encephalitis (HSE): 100 cases (S30.004). Neurology 88(16): Supplement S30.004
Acknowledgements
We particularly thank Professor Josep Dalmau in Service of Neurology, IDIBAPS Hospital Clínic, University of Barcelona, Casanova (Spain) for examining antibodies against neuronal cell-surface antigens, and MOG and AQP4 antibodies. We are extremely grateful to all participants and physicians for their contribution to this study, and acknowledge the efforts of research staffs, who worked on the clinical, and neuroimaging data collection.
Funding
This work was supported in part by a grant from Japan Epilepsy Research Foundation (JERFTENKAN 17002).
Author information
Authors and Affiliations
Contributions
Atsushi Kaneko: Study concept or design, data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Juntaro Kaneko: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Naomi Tominaga: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Naomi Kanazawa: Data acquisition, analysis or interpretation of the data, statistical analysis, drafting/revising the manuscript. Kasumi Hattori: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Yoshikazu Ugawa: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Arata Moriya: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Daiske Kuzume: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Daisuke Ishima: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Eiji Kitamura: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Kazutoshi Nishiyama: Data acquisition, analysis or interpretation of the data, drafting/revising the manuscript. Takahiro Iizuka: Study concept or design, data acquisition, analysis or interpretation of the data, statistical analysis, drafting/revising the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by Institutional Review Boards of Kitasato University (B17-144).
Conflicts of interest
A. Kaneko, J. Kaneko, N. Tominaga, N. Kanazawa, and K. Hattori report no disclosures; Y. Ugawa received grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan, the Ministry of Health, Labour and Welfare of Japan, the Support Center for Advanced Telecommunications Technology Research, the Association of Radio Industries Businesses, Ministry of Internal Affairs and Communications/grants from the Committee of the Study of Human Exposure to EMF, and the Novartis Foundation (Japan) for the Promotion of Science, Nihon Kohden, Ltd., Takeda Pharmaceutical Company Limited, Nippon Boehringer Ingelheim Co., Ltd., Mitsubishi Tanabe Pharma Corporation received honoraria from the Taiwan Movement Disorders Society, Chinese Neurology Society, Astellas Pharma Inc., Eisai Co., Ltd., FP Pharmaceutical Corporation, Otsuka Pharmaceutical Co., Ltd., Elsevier Japan K. K., Kissei Pharmaceutical Co., Ltd., Kyorin Pharmaceutical Co., Ltd., Kyowa Hakko Kirin Co., Ltd., GlaxoSmithKline K. K., Sanofi-Aventis K.K., Daiichi Sankyo Co., Ltd., Dainippon Sumitomo Pharma Co., Ltd., Takeda Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Teijin Pharma Ltd., Nippon Chemiphar Co., Ltd., Nihon Pharmaceutical Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., Novartis Pharma K.K., Bayer Yakuhin, Ltd., and Mochida Pharmaceutical Co., Ltd. Y. Ugawa also received royalties from CHUGAI-IGAKUSHA, Igaku-Shoin Ltd., Medical View Co. Ltd., and Blackwell Publishing K.K.; A. Moriya, D. Kuzume, D. Ishima, and E. Kitamura reports no disclosures; K. Nishiyama received grants from Nippon Boehringer Ingelheim Co., Ltd., Daiichi Sankyo Co., Ltd., Astellas Pharma Inc., Otsuka Pharmaceutical Co., Ltd., Kyorin Pharmaceutical Co., Ltd., Dainippon Sumitomo Pharma Co., Ltd., Teijin Pharma Limited., Nihon Pharmaceutical Co., Ltd., Pfizer Inc., Bristol-Myers Squibb, Japan Blood Products Organization, MSD, and Nihon Medi-Physics Co., Ltd; T. Iizuka is an editorial board member for Current Treatment Options in Neurology, and Rinsho Shinkeigaku, and received a grant from Japan Epilepsy Research Foundation.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaneko, A., Kaneko, J., Tominaga, N. et al. Pitfalls in clinical diagnosis of anti-NMDA receptor encephalitis. J Neurol 265, 586–596 (2018). https://doi.org/10.1007/s00415-018-8749-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-8749-3