Abstract
Background
L-Dopa represents the mainstay of therapy of Parkinson’s disease (PD), but its effectiveness is reduced with continued treatment and disease progression. Accordingly, there remains a need to explore novel treatment strategies to manage the signs and symptoms of the later disease stages. The aim of the study was to evaluate the efficacy and safety of adjunctive perampanel (PER) in patients with PD through a meta-analysis of existing trials.
Methods
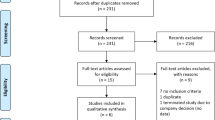
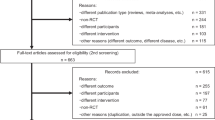
Randomized, placebo-controlled, double- or single-blind, add-on studies of PER in patients with PD were identified through a systematic literature search. The following outcomes were assessed: changes from baseline to final efficacy visit in total daily OFF time, activities of daily living during OFF time and motor function during ON time, incidence of adverse events (AEs), and treatment withdrawal.
Results
Four trials were included involving 2266 participants, 1449 and 817 for PER and placebo treatment groups, respectively. Four PER daily doses were tested, namely 0.5, 1, 2 and 4 mg. There were no significant differences in any efficacy outcome between PER and placebo treated patients. The risk ratios (RRs) for AEs, severe AEs and treatment withdrawal were similar between placebo and PER at 0.5, 1 and 2 mg; the 4 mg daily dose was associated with an increased risk of AEs [RR 1.118 (1.047–1.193)], and withdrawal for AEs [RR 1.345 (1.034–1.749)] and for any reason [RR 1.197 (1.020–1.406)].
Conclusions
In PD patients experiencing motor fluctuations, adjunctive PER did not improve the motor state and was well-tolerated at the lower doses.



Similar content being viewed by others
References
de Lau LM, Breteler MM (2006) Epidemiology of Parkinson’s disease. Lancet Neurol 5(525):35
Jankovic J, Stacy M (2007) Medical management of levodopa-associated motor complications in patients with Parkinson’s disease. CNS Drugs 21:677–692
Klockgether T, Turski L, Honoré T, Zhang ZM, Gash DM, Kurlan R, Greenamyre JT (1991) The AMPA receptor antagonist NBQX has antiparkinsonian effects in monoamine-depleted rats and MPTP-treated monkeys. Ann Neurol 30:717–723
Marin C, Jiménez A, Bonastre M, Vila M, Agid Y, Hirsch EC, Tolosa E (2001) LY293558, an AMPA glutamate receptor antagonist, prevents and reverses levodopa-induced motor alterations in Parkinsonian rats. Synapse 42:40–47
Konitsiotis S, Blanchet PJ, Verhagen L, Lamers E, Chase TN (2000) AMPA receptor blockade improves levodopa-induced dyskinesia in MPTP monkeys. Neurology 54:1589–1595
Eggert K, Squillacote D, Barone P, Dodel R, Katzenschlager R, Emre M, Lees AJ, Rascol O, Poewe W, Tolosa E, Trenkwalder C, Onofrj M, Stocchi F, Nappi G, Kostic V, Potic J, Ruzicka E, Oertel W (2010) German competence network on Parkinson’s disease. Safety and efficacy of perampanel in advanced Parkinson’s disease: a randomized, placebo-controlled study. Mov Disord 25:896–905
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. Higgins JPT, Green S (eds). The Cochrane Collaboration, 2011. http://handbook-5-1.cochrane.org/. Accessed June 2017
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Lattanzi S, Cagnetti C, Foschi N, Provinciali L, Silvestrini M (2016) Brivaracetam add-on for refractory focal epilepsy: a systematic review and meta-analysis. Neurology 86:1344–1352
Lattanzi S, Cagnetti C, Provinciali L, Silvestrini M (2017) How should we lower blood pressure after cerebral hemorrhage? A systematic review and meta-analysis. Cerebrovasc Dis 43:207–213
Lattanzi S, Cagnetti C, Danni M, Provinciali L, Silvestrini M (2017) Oral and intravenous steroids for multiple sclerosis relapse: a systematic review and meta-analysis. J Neurol 264:1697–1704
Lees A, Fahn S, Eggert KM, Jankovic J, Lang A, Micheli F, Mouradian MM, Oertel WH, Olanow CW, Poewe W, Rascol O, Tolosa E, Squillacote D, Kumar D (2012) Perampanel, an AMPA antagonist, found to have no benefit in reducing “off” time in Parkinson’s disease. Mov Disord 27:284–288
Rascol O, Barone P, Behari M, Emre M, Giladi N, Olanow CW, Ruzicka E, Bibbiani F, Squillacote D, Patten A, Tolosa E (2012) Perampanel in Parkinson disease fluctuations: a double-blind randomized trial with placebo and entacapone. Clin Neuropharmacol 35:15–20
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39:889–909
Galvan A, Wichmann T (2008) Pathophysiology of parkinsonism. Clin Neurophysiol 119:1459–1474
Stayte S, Vissel B (2014) Advances in non-dopaminergic treatments for Parkinson’s disease. Front Neurosci 8:113
Oh JD, Chase TN (2002) Glutamate-mediated striatal dysregulation and the pathogenesis of motor response complications in Parkinson’s disease. Amino Acids 23:133–139
Koutsilieri E, Riederer P (2007) Excitotoxicity and new antiglutamatergic strategies in Parkinson’s disease and Alzheimer’s disease. Parkinsonism Relat Disord 13(Suppl. 3):S329–S331
Engber TM, Papa SM, Boldry RC, Chase TN (1994) NMDA receptor blockade reverses motor response alterations induced by levodopa. NeuroReport 5:2586–2588
Blanchet PJ, Konitsiotis S, Whittemore ER, Zhou ZL, Woodward RM, Chase TN (1999) Differing effects of N-methyl-d-aspartate receptor subtype selective antagonists on dyskinesias in levodopa-treated 1-methyl-4-phenyl-tetrahydropyridine monkeys. J Pharmacol Exp Ther 290:1034–1040
Marin C, Papa S, Engber TM, Bonastre M, Tolosa E, Chase TN (1996) MK-801 prevents levodopa-induced motor response alterations in parkinsonian rats. Brain Res 736:202–205
Chase TN, Oh JD, Konitsiotis S (2000) Antiparkinsonian and antidyskinetic activity of drugs targeting central glutamatergic mechanisms. J Neurol 247(Suppl 2):II36–II42
Varanese S, Howard J, Di Rocco A (2010) NMDA antagonist memantine improves levodopa-induced dyskinesias and “on-off” phenomena in Parkinson’s disease. Mov Disord 25:508–510
Blair HA, Dhillon S (2017) Safinamide: a review in parkinson’s disease. CNS Drugs 31:169–176
Dingledine R, Borges K, Bowie D, Traynelis SF (1999) The glutamate receptor ion channels. Pharmacol Rev 51:7–61
Wiltgen BJ, Royle GA, Gray EE, Abdipranoto A, Thangthaeng N, Jacobs N, Saab F, Tonegawa S, Heinemann SF, O’Dell TJ, Fanselow MS, Vissel B (2010) A role for calcium-permeable AMPA receptors in synaptic plasticity and learning. PLoS One. https://doi.org/10.1371/journal.pone.0012818
Johnson KA, Conn PJ, Niswender CM (2009) Glutamate receptors as therapeutic targets for Parkinson’s disease. CNS Neurol Disord Drug Targ 8:475–491
Marin C, Jimenez A, Bonastre M, Chase TN, Tolosa E (2000) Non-NMDA receptor-mediated mechanisms are involved in levodopa-induced motor response alterations in Parkinsonian rats. Synapse 36:267–274
Matsunaga S, Kishi T, Iwata N (2017) Combination therapy with zonisamide and antiparkinson drugs for parkinson’s disease: a meta-analysis. J Alzheimer’s Dis 56:1229–1239
Hirsch EC, Hunot S (2009) Neuroinflammation in Parkinson’s disease: a target for neuroprotection? Lancet Neurol 8:382–397
Lattanzi S, Silvestrini M, Verbeek MM, Mollenhauer B, Lindqvist D, Janelidze S, Hansson O (2016) Increased CSF biomarkers of angiogenesis in Parkinson disease. Neurology 86:1747–1748
Dias V, Junn E, Mouradian MM (2013) The role of oxidative stress in Parkinson’s disease. J Parkinsons Dis 3:461–491
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
SL and MS declare that they have no conflict of interest. EG is an employee of Eisai s.r.l. FB has received speakers’ honoraria from Eisai and PeerVoice, payment for consultancy from Eisai, and travel support from Eisai, ITALFARMACO, and UCB Pharma.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lattanzi, S., Grillo, E., Brigo, F. et al. Efficacy and safety of perampanel in Parkinson’s disease. A systematic review with meta-analysis. J Neurol 265, 733–740 (2018). https://doi.org/10.1007/s00415-017-8681-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-017-8681-y