Abstract
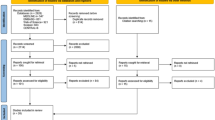
There are no validated criteria for the diagnosis of sensory neuronopathy (SNN) yet. In a preliminary monocenter study a set of criteria relying on clinical and electrophysiological data showed good sensitivity and specificity for a diagnosis of probable SNN. The aim of this study was to test these criteria on a French multicenter study. 210 patients with sensory neuropathies from 15 francophone reference centers for neuromuscular diseases were included in the study with an expert diagnosis of non-SNN, SNN or suspected SNN according to the investigations performed in these centers. Diagnosis was obtained independently from the set of criteria to be tested. The expert diagnosis was taken as the reference against which the proposed SNN criteria were tested. The set relied on clinical and electrophysiological data easily obtainable with routine investigations. 9/61 (16.4 %) of non-SNN patients, 23/36 (63.9 %) of suspected SNN, and 102/113 (90.3 %) of SNN patients according to the expert diagnosis were classified as SNN by the criteria. The SNN criteria tested against the expert diagnosis in the SNN and non-SNN groups had 90.3 % (102/113) sensitivity, 85.2 % (52/61) specificity, 91.9 % (102/111) positive predictive value, and 82.5 % (52/63) negative predictive value. Discordance between the expert diagnosis and the SNN criteria occurred in 20 cases. After analysis of these cases, 11 could be reallocated to a correct diagnosis in accordance with the SNN criteria. The proposed criteria may be useful for the diagnosis of probable SNN in patients with sensory neuropathy. They can be reached with simple clinical and paraclinical investigations.
Similar content being viewed by others
References
Kuntzer T, Antoine JC, Steck AJ (2004) Clinical features and pathophysiological basis of sensory neuronopathies (ganglionopathies). Muscle Nerve 30(3):255–268
Sghirlanzoni A, Pareyson D, Lauria G (2005) Sensory neuron diseases. Lancet Neurol 4(6):349–361
Graus F, Ribalta T, Campo E, Monforte R, Urbano A, Rozman C (1990) Immunohistochemical analysis of the immune reaction in the nervous system in paraneoplastic encephalomyelitis. Neurology 40(2):219–222
Dalmau J, Furneaux HM, Rosenblum MK, Graus F, Posner JB (1991) Detection of the anti-Hu antibody in specific regions of the nervous system and tumor from patients with paraneoplastic encephalomyelitis/sensory neuronopathy. Neurology 41(11):1757–1764
Wanschitz J, Hainfellner JA, Kristoferitsch W, Drlicek M, Budka H (1997) Ganglionitis in paraneoplastic subacute sensory neuronopathy: a morphologic study. Neurology 49(4):1156–1159
Scaravilli F, Sinclair E, Arango JC, Manji H, Lucas S, Harrison MJ (1992) The pathology of the posterior root ganglia in AIDS and its relationship to the pallor of the gracile tract. Acta Neuropathol (Berl) 84(2):163–170
Esiri MM, Morris CS, Millard PR (1993) Sensory and sympathetic ganglia in HIV-1 infection: immunocytochemical demonstration of HIV-1 viral antigens, increased MHC class II antigen expression and mild reactive inflammation. J Neurol Sci 114(2):178–187
Mori K, Iijima M, Koike H, Hattori N, Tanaka F, Watanabe H, Katsuno M, Fujita A, Aiba I, Ogata A, Saito T, Asakura K, Yoshida M, Hirayama M, Sobue G (2005) The wide spectrum of clinical manifestations in Sjogren’s syndrome-associated neuropathy. Brain 128(Pt 11):2518–2534
Griffin JW, Cornblath DR, Alexander E, Campbell J, Low PA, Bird S, Feldman EL (1990) Ataxic sensory neuropathy and dorsal root ganglionitis associated with Sjogren’s syndrome. Ann Neurol 27(3):304–315
Okajima T, Yamamura S, Hamada K, Kawasaki S, Ideta T, Ueno H, Tokuomi H (1983) Chronic sensory and autonomic neuropathy. Neurology 33(8):1061–1064
Sobue G, Yanagi T, Hashizume Y (1988) Chronic progressive sensory ataxic neuropathy with polyclonal gammopathy and disseminated focal perivascular cellular infiltrations. Neurology 38(3):463–467
Hainfellner JA, Kristoferitsch W, Lassmann H, Bernheimer H, Neisser A, Drlicek M, Beer F, Budka H (1996) T-cell-mediated ganglionitis associated with acute sensory neuronopathy. Ann Neurol 39(4):543–547
Kurokawa K, Noda K, Mimori Y, Watanabe C, Katayama S, Nakamura S, Sannomiya K, Yamamoto S, Tahara E (1998) A case of pandysautonomia with associated sensory ganglionopathy. J Neurol Neurosurg Psychiatry 65(2):278–279
Colli BO, Carlotti CG Jr, Assirati JA Jr, Lopes Lda S, Marques W Jr, Chimelli L, Neder L, Barreira AA (2008) Dorsal root ganglionectomy for the diagnosis of sensory neuropathies. Surgical technique and results. Surg Neurol 69(3):266–273 discussion 273
Camdessanche JP, Jousserand G, Ferraud K, Vial C, Petiot P, Honnorat J, Antoine JC (2009) The pattern and diagnostic criteria of sensory neuronopathy: a case-control study. Brain 132(Pt 7):1723–1733
Camdessanche JP, Antoine JC, Honnorat J, Vial C, Petiot P, Convers P, Michel D (2002) Paraneoplastic peripheral neuropathy associated with anti-Hu antibodies. A clinical and electrophysiological study of 20 patients. Brain 125(Pt 1):166–175
European Federation of Neurological Societies (2010) Peripheral Nerve Society Guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society-First Revision (2010). J Peripher Nerv Syst JPNS 15(1):1–9. doi:10.1111/j.1529-8027.2010.00245.x
Sinnreich M, Klein CJ, Daube JR, Engelstad J, Spinner RJ, Dyck PJ (2004) Chronic immune sensory polyradiculopathy: a possibly treatable sensory ataxia. Neurology 63(9):1662–1669
Viala K, Maisonobe T, Stojkovic T, Koutlidis R, Ayrignac X, Musset L, Fournier E, Leger JM, Bouche P (2010) A current view of the diagnosis, clinical variants, response to treatment and prognosis of chronic inflammatory demyelinating polyradiculoneuropathy. J Peripher Nerv Syst JPNS 15(1):50–56. doi:10.1111/j.1529-8027.2010.00251.x
Gorson KC, Herrmann DN, Thiagarajan R, Brannagan TH, Chin RL, Kinsella LJ, Ropper AH (2008) Non-length dependent small fibre neuropathy/ganglionopathy. J Neurol Neurosurg Psychiatry 79(2):163–169
Devigili G, Tugnoli V, Penza P, Camozzi F, Lombardi R, Melli G, Broglio L, Granieri E, Lauria G (2008) The diagnostic criteria for small fibre neuropathy: from symptoms to neuropathology. Brain 131(Pt 7):1912–1925
Willison HJ, O’Leary CP, Veitch J, Blumhardt LD, Busby M, Donaghy M, Fuhr P, Ford H, Hahn A, Renaud S, Katifi HA, Ponsford S, Reuber M, Steck A, Sutton I, Schady W, Thomas PK, Thompson AJ, Vallat JM, Winer J (2001) The clinical and laboratory features of chronic sensory ataxic neuropathy with anti-disialosyl IgM antibodies. Brain 124(Pt 10):1968–1977
Attarian S, Boucraut J, Hubert AM, Uzenot D, Delmont E, Verschueren A, Franques J, Azulay JP, Pouget J (2010) Chronic ataxic neuropathies associated with anti-GD1b IgM antibodies: response to IVIg therapy. J Neurol Neurosurg Psychiatry 81(1):61–64. doi:10.1136/jnnp.2009.185736
Camdessanche JP, Jousserand G, Franques J, Pouget J, Delmont E, Creange A, Kuntzer T, Maisonobe T, Abba K, Antoine JC (2012) A clinical pattern-based etiological diagnostic strategy for sensory neuronopathies: a French collaborative study. J Peripher Nerv Syst JPNS 17(3):331–340. doi:10.1111/j.1529-8027.2012.00411.x
Sheikh SI, Amato AA (2010) The dorsal root ganglion under attack: the acquired sensory ganglionopathies. Pract Neurol 10(6):326–334. doi:10.1136/jnnp.2010.230532
Lauria G, Pareyson D, Grisoli M, Sghirlanzoni A (2000) Clinical and magnetic resonance imaging findings in chronic sensory ganglionopathies. Ann Neurol 47(1):104–109
Conflicts of interest
This study was supported by a grant from the French Ministry of Health (PHRC Inter-Regional 2006 and 2010 API 28-01). None of the authors have financial relationship with the organization that sponsored the research.
Ethical standards
The study was approved by the Ethics Committee of the University Hospital of Saint-Etienne, France. For patients seen before 2011 being a retrospective non interventional study informed consent was not necessary while patients seen from 2011 onward signed an informed consent.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Members of the French CIDP study group are listed in “Appendix”.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
Other members of the French CIDP group who provided cases for the study are, in alphabetical order, Françoise Bouhour, CHU de Lyon, France; Pierre Clavelou, CHU de Clermont-Ferrand, France; Andoni Echaniz-Laguna, CHU de Strasbourg, France; Hélène Gervais-Bernard, CHU de Lyon, France; Joerg Kleeberg, CHUV Lausanne, Switzerland; Emeline Lagrange, CHU de Grenoble, France; Jean-Marc Leger, Hôpital de la Salpetrière, Paris, France; Guillaume Nicolas, CHU Angers, France; François Ochsner, La Chaux-de-Fond, Switzerland; Yann Péréon, CHU de Nantes, France; Philippe Petiot, CHU Lyon, France; Pierre Soichot, CHU de Dijon, France; Guillaume Taïeb, CHU de Nîmes, France; Jean-Michel Vallat, CHU de Limoges, France; Christophe Vial, CHU Lyon, France.
Rights and permissions
About this article
Cite this article
Antoine, JC., Robert-Varvat, F., Maisonobe, T. et al. Testing the validity of a set of diagnostic criteria for sensory neuronopathies: a francophone collaborative study. J Neurol 261, 2093–2100 (2014). https://doi.org/10.1007/s00415-014-7423-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7423-7