Abstract
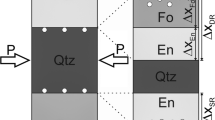
Reaction rims contain a wealth of information that can be used to decipher the P-T-t-X history of metamorphic and metasomatic rocks. One of the most important parameters that controls reaction rim growth is the presence of volatiles, which can affect rim thicknesses, phase stabilities and the development of rim microstructures. In this study, reaction rim growth experiments were performed between periclase and quartz at anhydrous to water-saturated conditions at 3–4 kbar and 1100–1300 °C. Controlled minute amounts of water were added through OH-doped periclase, which enabled us to perform experiments at controlled water-undersaturated conditions. At anhydrous conditions, no reaction rim formed at all implying that water acts as a catalyst, and a minimum fluid threshold is needed to initiate metamorphic reactions. At water-undersaturated conditions extremely small variations in water content are sufficient to change reaction rim growth rates by multiple orders of magnitude. This implies that reaction rims have the potential to monitor variations in the amount of water at those grain boundaries that serve as fast pathways for component transport at water-undersaturated conditions during metamorphic and metasomatic reactions in natural systems, allowing them to be used as sensitive “geohygrometers”. Additionally, the effect of water on relative layer thicknesses may provide an application for reaction rim microstructures to be used as new physico-chemical gauges that will allow us to discriminate between water-undersaturated and water-saturated conditions during metamorphic events.







Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings in this study are available within the article and the supplementary materials (ESM_1-3).
References
Abart R, Petrishcheva E (2011) Thermodynamic model for reaction rim growth: interface reaction and diffusion control. Am J Sci 311:517–527. https://doi.org/10.2475/06.2011.02
Abart R, Kunze K, Milke R, Sperb R, Heinrich W (2004) Silicon and oxygen self diffusion in enstatite polycrystals: the Milke et al. (2001) rim growth experiments revisited. Contrib Mineral Petrol 147:633–646. https://doi.org/10.1007/s00410-004-0596-9
Austrheim H (1987) Eclogitization of lower crustal granulites by fluid migration through shear zones. Earth Planet Sci Lett 81:221–232. https://doi.org/10.1016/0012-821X(87)90158-0
Brady J, McCallister R (1983) Diffusion data for clinopyroxenes from homogenization and self-diffusion experiments. Am Mineral 68:95–105
Cantwell PR, Tang M, Dillon SJ, Luo J, Rohrer GS, Harmer MP (2014) Grain boundary complexions. Acta Mater 62:1–48. https://doi.org/10.1016/j.actamat.2013.07.037
Carlson WD (2010) Dependence of reaction kinetics on H2O activity as inferred from rates of intergranular diffusion of aluminium. J Metamorph Geol 28:735–752. https://doi.org/10.1111/j.1525-1314.2010.00886.x
Clark C, Schmidt Mumm A, Faure K (2005) Timing and nature of fluid flow and alteration during Mesoproterozoic shear zone formation, Olary domain, South Australia. J Metamorph Geol 23:147–164. https://doi.org/10.1111/j.1525-1314.2005.00568.x
Carter RE (1961) Mechanism of solid-state reaction between magnesium oxide and aluminum oxide and between magnesium oxide and ferric oxide. J Am Ceram Soc 44:116–120. https://doi.org/10.1111/j.1151-2916.1961.tb13724.x
Dohmen R, Milke R (2010) Diffusion in polycrystalline materials: grain boundaries, mathematical models, and experimental data. Rev Miner Geochem 72:921–970. https://doi.org/10.2138/rmg.2010.72.21
Farver JR, Yund RA (1995) Grain boundary diffusion of oxygen, potassium and calcium in natural and hot-pressed feldspar aggregates. Contrib Mineral Petrol 118:340–355. https://doi.org/10.1007/s004100050019
Fisher GW (1978) Rate laws in metamorphism. Geochim Cosmochim Acta 42:1035–1050
Fisler DK, Mackwell SJ, Petsch S (1997) Grain boundary diffusion in enstatite. Phys Chem Min 24:264–273. https://doi.org/10.1007/s002690050038
Franke M, Joachim-Mrosko B (2022) The effect of fluorine on reaction rim growth dynamics in the ternary CaO-MgO-SiO2 system. Am Mineral 107:1477–1486. https://doi.org/10.2138/am-2022-8123CCBY
Furstoss J, Hirel P, Carrez P, Cordier P (2022) Complexions and stoichiometry of the 60.8°//[100](011) symmetrical tilt grain boundary in Mg2SiO4 forsterite: a combined empirical potential and first-principles study. Am Mineral. https://doi.org/10.2138/am-2022-8420
Gardés E, Heinrich W (2011) Growth of multilayered polycrystalline reaction rims in the MgO-SiO2 system, part II: modelling. Contrib Mineral Petrol 162:37–49. https://doi.org/10.1007/s00410-010-0581-4
Gardés E, Wunder B, Wirth R, Heinrich W (2011) Growth of multilayered polycrystalline reaction rims in the MgO-SiO2 system, part I: experiments. Contrib Mineral Petrol 161:1–12. https://doi.org/10.1007/s00410-010-0517-z
Gardés E, Wunder B, Marquardt K, Heinrich W (2012) The effect of water on intergranular mass transport: new insights from diffusion-controlled reaction rims in the MgO-SiO2 system. Contrib Mineral Petrol 164:1–16. https://doi.org/10.1007/s00410-012-0721-0
González R, Chen Y (2002) Transport of hydrogenic species in crystalline oxides: radiation and electric-field-enhanced diffusion. J Phys Condens Matter 14:R1143–R1173. https://doi.org/10.1088/0953-8984/14/45/201
Götze LC, Abart R, Rybacki E, Keller LM, Petrishcheva E, Dresen G (2010) Reaction rim growth in the system MgO-Al2O3-SiO2 under uniaxial stress. Mineral Petrol 99:263–277. https://doi.org/10.1007/s00710-009-0080-3
Incel S, Milke R, Wunder B (2022) Orthopyroxene rim growth during reaction of (Co, Ni, Mn, Zn)-doped forsterite and quartz: Experimental constraints on element distribution and grain boundary diffusion. Mineral Petrol 116:137–149. https://doi.org/10.1007/s00710-022-00773-3
Joachim B (2011) Reaction rim growth in the systems MgO-SiO2 and CaO-MgO-SiO2: diffusion pathways and the effect of water. Dissertation, German Research Centre of Geosciences, Potsdam
Joachim B, Gardés E, Abart R, Heinrich W (2011) Experimental growth of åkermanite reaction rims between wollastonite and monticellite: evidence for volume diffusion control. Contrib Mineral Petrol 161:389–399. https://doi.org/10.1007/s00410-010-0538-7
Joachim B, Gardés E, Velickov B, Abart R, Heinrich W (2012) Experimental growth of diopside + merwinite reaction rims: The effect of water on microstructure development. Am Mineral 97:220–230. https://doi.org/10.2138/am.2011.3819
Joachim B, Wohlers A, Norberg N, Gardés E, Petrishcheva E, Abart R (2013) Diffusion and solubility of hydrogen and water in periclase. Phys Chem Miner 40:19–27. https://doi.org/10.1007/s00269-012-0542-8
Joachim B, Heinrich W, Höschen C, Abart R (2019) The effect of H2O fluid on relative component mobilities in a bimineralic reaction rim in the system CaO-MgO-SiO2. Eur J Mineral 31:61–72. https://doi.org/10.1127/ejm/2019/0031-2796
Joesten R (1977) Evolution of mineral assemblage zoning in diffusion metasomatism. Geochim Cosmochim Acta 41:649–670. https://doi.org/10.1016/0016-7037(77)90303-9
Keller LM, Götze LC, Rybacki E, Dresen G, Abart R (2010) Enhancement of solid-state reaction rates by non-hydrostatic stress effects on polycrystalline diffusion kinetics. Am Mineral 95:1399–1407. https://doi.org/10.2138/am.2010.3372
Libowitzky E, Rossman GR (1997) An IR absorption calibration for water in minerals. Am Mineral 82:1111–1115. https://doi.org/10.2138/am-1997-11-1208
Liu M, Peterson J, Yund RA (1997) Diffusion-controlled growth of albite and pyroxene reaction rims. Contrib Mineral Petrol 126:217–223. https://doi.org/10.1007/s004100050245
Milke R, Heinrich W (2002) Diffusion-controlled growth of wollastonite rims between quartz and calcite: Comparison between nature and experiment. J Metamorph Geol 20:467–480. https://doi.org/10.1046/j.1525-1314.2002.00384.x
Milke R, Wiedenbeck M, Heinrich W (2001) Grain boundary diffusion of Si, Mg, and O in enstatite reaction rims: a SIMS study using isotopically doped reactants. Contrib Mineral Petrol 142:15–26. https://doi.org/10.1007/s004100100277
Milke R, Dohmen R, Becker HW, Wirth R (2007) Growth kinetics of enstatite reaction rims studied on nano-scale, Part I: Methodology, microscopic observations and the role of water. Contrib Mineral Petrol 154:519–533. https://doi.org/10.1007/s00410-007-0207-7
Milke R, Kolzer K, Koch-Müller M, Wunder B (2009) Orthopyroxene rim growth between olivine and quartz at low temperatures (750–950°C) and low water concentration. Mineral Petrol 97:223–232. https://doi.org/10.1007/s00710-009-0093-y
Milke R, Neusser G, Kolzer K, Wunder B (2013) Very little water is necessary to make a dry solid silicate system wet. Geology 41:247–250. https://doi.org/10.1130/G33674.1
Mindaleva D, Uno M, Higashino F, Nagaya T, Okamoto A, Tsuchiya N (2020) Rapid fluid infiltration and permeability enhancement during middle-lower crustal fracturing: evidence from amphibolite-granulite-facies fluid-rock reaction zones, Sør Rondane Mountains. East Antarctica Lithos 372–373:105521. https://doi.org/10.1016/j.lithos.2020.105521
Phillips BBA, Busing WR (1956) Comparison of the infrared and Raman spectra of some crystalline hydroxides. J Phys Chem 61:502
Putnis A, Austrheim H (2010) Fluid-induced processes: metasomatism and metamorphism. Geofluids 10:254–269. https://doi.org/10.1111/j.1468-8123.2010.00285.x
Rossi RC, Fulrath RM (1963) Epitaxial growth of spinel by the reaction in solid state. J Am Ceram Soc 46:145–149
Rubie DC (1986) The catalysis of mineral reactions by water and restrictions on the presence of aqueous fluid during metamorphism. Mineral Mag 50:399–415. https://doi.org/10.1180/minmag.1986.050.357.05
Rybacki E, Helpa V (2019) Influence of differential stress on the growth of wet enstatite and enstatite-forsterite reaction rims. Mineral Petrol 113:433–448. https://doi.org/10.1007/s00710-019-00667-x
Schorn S, Diener JFA (2017) Details of the gabbro-to-eclogite transition determined from microtextures and calculated chemical potential relationships. J Metamorph Geol 35:55–75. https://doi.org/10.1111/jmg.12220
Truckenbrodt J, Johannes W (1999) H2O loss during piston-cylinder experiments. Am Mineral 84:1333–1335. https://doi.org/10.2138/am-1999-0909
Tursi F (2022) The key role of µH2O gradients in deciphering microstructures and mineral assemblages of mylonites: examples from the Calabria polymetamorphic terrane. Mineral Petrol 116:1–14. https://doi.org/10.1007/s00710-021-00766-8
Watson EB, Brenan JM (1987) Fluids in the lithosphere, 1. Experimentally-determined wetting characteristics of CO2H2O fluids and their implications for fluid transport, host-rock physical properties, and fluid inclusion formation. Earth Planet Sci Letter 85:497–515. https://doi.org/10.1016/0012-821X(87)90144-0
Watson EB, Price JD (2002) Kinetics of the reaction MgO + Al2O3 → MgAl2O4 and Al-Mg interdiffusion in spinel at 1200 to 2000°C and 1.0 to 4.0 GPa. Geochim Cosmochim Acta 66:2123–2138. https://doi.org/10.1016/S0016-7037(02)00827-X
Whitney WP, Stubican VS (1971) Interdiffusion studies in the system MgOAl2O3. J Phys Chem Solids 32:305–312. https://doi.org/10.1016/0022-3697(71)90015-1
Yund RA (1997) Rates of grain boundary diffusion through enstatite and forsterite reaction rims. Contrib Mineral Petrol 126:224–236. https://doi.org/10.1007/s004100050246
Zaraysky G, Balashov V, Lebedeva M (1989) Macrokinetic model of metasomatic zonality. Geokhimiya 10:1386–1395
Acknowledgements
We thank M. Tribus for her assistance with sample preparation and microprobe analysis. We are grateful for detailed reviews by R. Abart and an anonymous reviewer who significantly improved the quality of this paper. This research was funded by the Austrian Science Fund (FWF) project P 31787.
Funding
This study was funded by the Austrian Science Fund (FWF) project P 31787.
Author information
Authors and Affiliations
Contributions
Conceptualization: MGF, BJ-M; methodology: MGF, BJ-M; formal analysis and investigation: MGF, BJ-M, BCS, RS; funding acquisition: BJ-M; resources: MGF, BCS, RS; supervision: BJ-M; visualization: MGF; project administration: MGF, BJ-M; writing—original draft preparation: MGF; writing—review and editing: MGF, BCS, RS, BJ-M.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Communicated by Othmar Müntener.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Franke, M.G., Schmidt, B.C., Stalder, R. et al. Metamorphic reaction kinetics at anhydrous to water-saturated conditions in the binary MgO-SiO2 system. Contrib Mineral Petrol 178, 87 (2023). https://doi.org/10.1007/s00410-023-02064-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00410-023-02064-2