Abstract
Background and purpose
Induction chemotherapy (IC) plus concurrent chemoradiotherapy (CCRT) has been established as standard of care for locoregionally advanced nasopharyngeal carcinoma (LANPC). No direct comparison between different IC regimens has been performed. We conducted Bayesian network meta-analysis to evaluate the efficacy and safety of IC regimens in LANPC.
Materials and methods
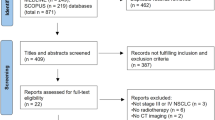
We systematically searched studies comparing different regimens of IC plus CCRT versus CCRT alone for LANPC. Pairwise meta-analysis and Bayesian network meta-analysis were conducted using Review Manger, Stata and R software.
Results
Eight eligible studies with a total of 2382 patients were involved. Compared with CCRT alone, IC + CCRT significantly improved PFS (HR = 0.68 [95% CI 0.59–0.79]) and OS (HR = 0.72 [95% CI 0.61–0.86]) in conventional meta-analysis. In Bayesian network meta-analysis, GP (gemcitabine and cisplatin) had advantage in prolonging PFS, OS and DMFS. GP had adverse but manageable impacts on hemoglobin and platelet. Meanwhile, treatment compliance of GP was higher than that of other regimens.
Conclusion
Based on existing evidences, GP could likely to be recommended as an optimal IC regimen for LANPC.





Similar content being viewed by others
Availability of data and materials
The data and materials in the article are all available.
Abbreviations
- ASCO:
-
American Society for Radiation Oncology
- ASTRO:
-
American Society for Radiation Oncology
- CC:
-
Concurrent chemotherapy
- CCRT:
-
Concurrent chemoradiotherapy
- CEP:
-
Paclitaxel, epirubicin and cis-platinum
- CI:
-
Confidence interval
- CSCO:
-
Chinese Society of Clinical Oncology
- CSTRO:
-
Chinese Society for Radiation Oncology
- DMFS:
-
Distant metastasis free survival
- ESMO:
-
European Society for Medical Oncology
- ESTRO:
-
European Society for Radiotherapy & Oncology
- GCP:
-
Paclitaxel, gemcitabine and carboplatin
- GP:
-
Cisplatin plus gemcitabine
- HR:
-
Hazard ratio
- IC:
-
Induction chemotherapy
- LANPC:
-
Locoregionally advanced nasopharyngeal carcinoma
- LRFS:
-
Locoregional failure survival
- MEPFL:
-
Mitomycin, epirubicin, cisplatin, fluorouracil and leucovorin
- NPC:
-
Nasopharyngeal carcinoma
- OR:
-
Odds ratio
- OS:
-
Overall survival
- PF:
-
Cisplatin plus 5-fluorouracil
- PFS:
-
Progression-free survival
- PX:
-
Cisplatin plus capecitabine
- RCT:
-
Randomized clinical trial
- TP:
-
Docetaxel plus cisplatin
- TPF:
-
Cisplatin, fluorouracil plus docetaxel
References
Sung H et al (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249. https://doi.org/10.3322/caac.21660
Chen W et al (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66(2):115–132. https://doi.org/10.3322/caac.21338
Pan JJ et al (2016) Proposal for the 8th edition of the AJCC/UICC staging system for nasopharyngeal cancer in the era of intensity-modulated radiotherapy. Cancer 122(4):546–558. https://doi.org/10.1002/cncr.29795
Chen YP et al (2021) Chemotherapy in combination with radiotherapy for definitive-intent treatment of stage II–IVA nasopharyngeal carcinoma: CSCO and ASCO Guideline. J Clin Oncol 39(7):840–859. https://doi.org/10.1200/JCO.20.03237
Yang SS et al (2020) Effect of induction chemotherapy in nasopharyngeal carcinoma: an updated meta-analysis. Front Oncol 10:591205. https://doi.org/10.3389/fonc.2020.591205
Chen YP et al (2018) Induction chemotherapy plus concurrent chemoradiotherapy in endemic nasopharyngeal carcinoma: individual patient data pooled analysis of four randomized trials. Clin Cancer Res 24(8):1824–1833. https://doi.org/10.1158/1078-0432.CCR-17-2656
Tang M, Jia Z, Zhang J (2021) The evaluation of adding induction chemotherapy to concurrent chemoradiotherapy for locally advanced nasopharyngeal carcinoma: a meta-analysis. Eur Arch Otorhinolaryngol 278(5):1545–1558. https://doi.org/10.1007/s00405-020-06218-x
Li WF et al (2019) Concurrent chemoradiotherapy with/without induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: long-term results of phase 3 randomized controlled trial. Int J Cancer 145(1):295–305. https://doi.org/10.1002/ijc.32099
Sun Y et al (2016) Induction chemotherapy plus concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: a phase 3, multicentre, randomised controlled trial. Lancet Oncol 17(11):1509–1520. https://doi.org/10.1016/S1470-2045(16)30410-7
Zhang Y et al (2019) Gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma. N Engl J Med 381(12):1124–1135. https://doi.org/10.1056/NEJMoa1905287
Hui EP et al (2009) Randomized phase II trial of concurrent cisplatin-radiotherapy with or without neoadjuvant docetaxel and cisplatin in advanced nasopharyngeal carcinoma. J Clin Oncol 27(2):242–249. https://doi.org/10.1200/JCO.2008.18.1545
Cao SM et al (2017) Neoadjuvant chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: a phase III multicentre randomised controlled trial. Eur J Cancer 75:14–23. https://doi.org/10.1016/j.ejca.2016.12.039
Hong RL et al (2018) Final results of a randomized phase III trial of induction chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in patients with stage IVA and IVB nasopharyngeal carcinoma-Taiwan Cooperative Oncology Group (TCOG) 1303 Study. Ann Oncol 29(9):1972–1979. https://doi.org/10.1093/annonc/mdy249
Tonin FS et al (2017) Network meta-analysis: a technique to gather evidence from direct and indirect comparisons. Pharm Pract (Granada) 15(1):943. https://doi.org/10.18549/PharmPract.2017.01.943
Catala-Lopez F et al (2014) Network meta-analysis for comparing treatment effects of multiple interventions: an introduction. Rheumatol Int 34(11):1489–1496. https://doi.org/10.1007/s00296-014-2994-2
Higgins JP et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. https://doi.org/10.1136/bmj.d5928
Shim SR et al (2019) Network meta-analysis: application and practice using R software. Epidemiol Health 41:e2019013. https://doi.org/10.4178/epih.e2019013
Neupane B et al (2014) Network meta-analysis using R: a review of currently available automated packages. PLoS One 9(12):e115065. https://doi.org/10.1371/journal.pone.0115065
Al-Sarraf M et al (1998) Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: phase III randomized Intergroup study 0099. J Clin Oncol 16(4):1310–1317. https://doi.org/10.1200/JCO.1998.16.4.1310
Blanchard P et al (2015) Chemotherapy and radiotherapy in nasopharyngeal carcinoma: an update of the MAC-NPC meta-analysis. Lancet Oncol 16(6):645–655. https://doi.org/10.1016/S1470-2045(15)70126-9
Yang Q et al (2019) Induction chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: long-term results of a phase III multicentre randomised controlled trial. Eur J Cancer 119:87–96. https://doi.org/10.1016/j.ejca.2019.07.007
Zhong YH et al (2013) Phase II trial of neoadjuvant docetaxel and cisplatin followed by intensity-modulated radiotherapy with concurrent cisplatin in locally advanced nasopharyngeal carcinoma. Cancer Chemother Pharmacol 71(6):1577–1583. https://doi.org/10.1007/s00280-013-2157-2
Lee AWM et al (2020) NPC-0501 trial on the value of changing chemoradiotherapy sequence, replacing 5-fluorouracil with capecitabine, and altering fractionation for patients with advanced nasopharyngeal carcinoma. Cancer 126(16):3674–3688. https://doi.org/10.1002/cncr.32972
Frikha M et al (2018) A randomized trial of induction docetaxel-cisplatin-5FU followed by concomitant cisplatin-RT versus concomitant cisplatin-RT in nasopharyngeal carcinoma (GORTEC 2006–02). Ann Oncol 29(3):731–736. https://doi.org/10.1093/annonc/mdx770
Gabani P et al (2018) Induction chemotherapy in the treatment of nasopharyngeal carcinoma: clinical outcomes and patterns of care. Cancer Med 7(8):3592–3603. https://doi.org/10.1002/cam4.1626
OuYang PY et al (2019) A pairwise meta-analysis of induction chemotherapy in nasopharyngeal carcinoma. Oncologist 24(4):505–512. https://doi.org/10.1634/theoncologist.2018-0522
Zhang Y, Sun Y, Ma J (2019) Induction gemcitabine and cisplatin in locoregionally advanced nasopharyngeal carcinoma. Cancer Commun (Lond) 39(1):39. https://doi.org/10.1186/s40880-019-0385-5
Zang J et al (2020) Gemcitabine and cisplatin versus docetaxel and cisplatin as induction chemotherapy followed by concurrent chemoradiotherapy in locoregionally advanced nasopharyngeal carcinoma from non-endemic area of China. J Cancer Res Clin Oncol 146(9):2369–2378. https://doi.org/10.1007/s00432-020-03229-3
Zhu J et al (2019) Comparison of GP and TPF induction chemotherapy for locally advanced nasopharyngeal carcinoma. Oral Oncol 97:37–43. https://doi.org/10.1016/j.oraloncology.2019.08.001
Zeng Z et al (2018) Assessment of concurrent chemoradiotherapy plus induction chemotherapy in advanced nasopharyngeal carcinoma: cisplatin, fluorouracil, and docetaxel versus gemcitabine and cisplatin. Sci Rep 8(1):15581. https://doi.org/10.1038/s41598-018-33614-5
Yang Y (2015) Cancer immunotherapy: harnessing the immune system to battle cancer. J Clin Investig 125(9):3335–3337. https://doi.org/10.1172/JCI83871
Fang W et al (2018) Camrelizumab (SHR-1210) alone or in combination with gemcitabine plus cisplatin for nasopharyngeal carcinoma: results from two single-arm, phase 1 trials. Lancet Oncol 19(10):1338–1350. https://doi.org/10.1016/S1470-2045(18)30495-9
Mai HQ et al (2021) Toripalimab or placebo plus chemotherapy as first-line treatment in advanced nasopharyngeal carcinoma: a multicenter randomized phase 3 trial. Nat Med 27(9):1536–1543. https://doi.org/10.1038/s41591-021-01444-0
Yang Y et al (2021) Camrelizumab versus placebo in combination with gemcitabine and cisplatin as first-line treatment for recurrent or metastatic nasopharyngeal carcinoma (CAPTAIN-1st): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol 22(8):1162–1174. https://doi.org/10.1016/S1470-2045(21)00302-8
Funding
Funding was provided by Health and Family Planning Commission of Hubei Province (WJ2019H060), Science, Technology and Innovation Seed Fund of Zhongnan Hospital of Wuhan University (znpy2018123).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing interests.
Ethics approval and consent to participate
Not applicable.
Consent for publication
All the authors consent the publication of this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
405_2022_7435_MOESM2_ESM.pdf
Figure S2 Results of pairwise meta-analysis aiming to compare effectiveness of IC+CCRT to that of CCRT alone. (A) PFS; (B) OS; (C) DMFS; (D) LRFS. (PDF 1671 kb)
405_2022_7435_MOESM3_ESM.pdf
Figure S3 Results of pairwise meta-analysis aiming to compare treatment-related toxicities of IC+CCRT to that of CCRT alone. (A) Neutropenia; (B) Leukopenia; (C) Nausea and vomiting; (D) Anemia; (E) Thrombocytopenia; (F) Mucositis. (PDF 2556 kb)
405_2022_7435_MOESM5_ESM.pdf
Figure S5 Bayesian network meta-analysis for treatment-related toxicities. (A) Leukopenia; (B) Neutropenia; (C) Anemia; (D) Thrombocytopenia; (E) Nausea and vomiting; (F) Mucositis. (PDF 277 kb)
Rights and permissions
About this article
Cite this article
Wu, Q., Li, S., Liu, J. et al. Optimal induction chemotherapy regimen for locoregionally advanced nasopharyngeal carcinoma: an update Bayesian network meta-analysis. Eur Arch Otorhinolaryngol 279, 5057–5069 (2022). https://doi.org/10.1007/s00405-022-07435-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-022-07435-2