Abstract
Background
Distant metastases from squamous cell cancer of the vulva (VSCC) are encountered rarely and are associated with a poor prognosis. Cerebral metastases have only been described anecdotally.
Case history
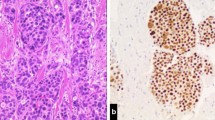
A 51-year old woman was diagnosed with hepatic metastases due to VSCC. Initial therapy comprised wide local excision of the primary tumor with inguino-femoral lymphadenectomy (LAE) followed by stereotactic radiation of the singular hepatic metastasis while adjuvant chemoradiation of the vulva and lymphatics was declined. 3 years later, she subsequently developed lung and cerebral metastases.
Conclusion
The course of metastatic disease in VSCC is poorly understood. Further knowledge of the metastatic patterns in vulvar cancer is required for guidance of future therapeutic interventions.


Similar content being viewed by others
References
National Cancer Institute: SEER cancer statistics factsheets: vulvar cancer. https://seer.cancer.gov/statfacts/html/vulva.html. Accessed 6 Dec 2019
De Witte O et al (1996) Metastases cerebrales d’origine gynecologique. Neurochirurgie 42:216–220
Alkatout I et al (2015) Vulvar cancer: epidemiology, clinical presentation and management options. Int J Womens Health 7:305–313
Ueda Y et al (2011) Two distinct pathways of development of squamous cell carcinoma of the vulva. J Skin Cancer 2011:951250
Gadducci A et al (2006) Old and new perspectives in the management of high-risk, locally advanced or recurrent, and metastatic vulvar cancer. Crit Rev Oncol Hematol 60(3):227–241
Gadducci A et al (2012) Clinico-pathological and biological prognostic variables in squamous cell carcinoma of the vulva. Crit Rev Oncol Hematol 83(1):71–83
Mahner S et al (2015) Adjuvant therapy in lymph node-positive vulvar cancer: the AGO-CaRE-1 study. J Natl Cancer Inst. https://doi.org/10.1093/jnci/dju426
Nooij LS et al (2016) Risk factors and treatment for recurrent vulvar squamous cell carcinoma. Crit Rev Oncol Hematol 106:1–13
Cormio G et al (2010) Groin recurrence in carcinoma of the vulva: management and outcome. Eur J Cancer Care (Engl) 19(3):302–307
Coulter J et al (2003) Local and regional recurrence of vulval cancer:management dilemmas. Best Pract Res Clin Obstet Gynaecol. 17(4):663–681
Prieske K et al (2016) Patterns of distant metastases in vulvar cancer. Gynecol Oncol 142:427–434
Vázquez JP et al (2007) Brain metastasis and carcinomatous meningitis from vulvar squamous cell carcinoma: case report. Eur J Gynaecol Oncol 28(2):152–154
Dursun P et al (2009) Cerebellar metastasis in squamous cell vulvar carcinoma. Arch Gynecol Obstet 279(1):69–71
Tewari KS et al (2014) Improved survival with bevacizumab in advanced cervical cancer. N Engl J Med 370:734–743
Chung HC, Ros W, Delord JP et al (2019) Efficacy and safety of pembrolizumab in previously treated advanced cervical cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol. https://doi.org/10.1200/jco.18.01265
Hollebecque A, Meyer T, Moore KN et al (2017) An open-label, multicohort, phase I/II study of nivolumab in patients with virus-associated tumors (CheckMate 358): efficacy and safety in recurrent or metastatic (R/M) cervical, vaginal, and vulvar cancers. J Clin Oncol 35(15):5504. https://doi.org/10.1200/JCO.2017.35.15
Schuurman MS et al (2013) Trends in incidence and survival of Dutch women with vulvar squamous cell carcinoma. Eur J Cancer 49(18):3872–3880
Hacker NF et al (2012) Cancer of the vulva. Int J Gynaecol Obstet 119(Suppl. 2):S90–S96
van der Stehen S et al (2010) New FIGO staging system of vulvar cancer indeed provides a better reflection of prognosis. Gynecol Oncol 119(3):520–525
Homesley HD et al (1986) Radiation therapy versus pelvic node resection for carcinoma of the vulva with positive groin nodes. Obstet Gynecol 68:733–740
Oonk MH et al (2010) Size of sentinel-node metastasis and chances of non-sentinel-node involvement and survival in early stage vulvar cancer: results from GROINSS-V, a multicentre observational study. Lancet Oncol 11:646–652
Moore DH et al (1998) Preoperative chemoradiation for advanced vulvar cancer: a phase II study of the gynaecologic oncology group. Int J Radiat Oncol Biol Phys 42:79–85
Lupi G et al (1996) Combined preoperative chemoradiotherapy followed by radical surgery in locally advanced vulvar carcinoma. Cancer 77:1472–1478
Te Grootenhuis NC et al (2016) Sentinel nodes in vulvar cancer: long-term follow-up of the GROningen INternational Study on Sentinel nodes in Vulvar cancer (GROINSS-V) I. Gynecol Oncol 140(1):157–159
Woelber L et al (2009) Clinicopathological prognostic factors and patterns of recurrence in vulvar cancer. Anticancer Res 29(2):545–552
Woolderink JM et al (2006) Patterns and frequency of recurrences of squamous cell carcinoma of the vulva. Gynecol Oncol 103(1):293–299
Konidaris S, Bakas P, Gregoriou O et al (2011) Surgical management of invasive carcinoma of the vulva A retrospective analysis and review. Eur J Gynaecol Oncol 32(5):505–508
Tabbaa ZM, Gonzalez J, Sznurkowski JJ et al (2012) Impact of the new FIGO 2009 staging classification for vulvar cancer on prognosis and stage distribution. Gynecol Oncol 127(1):147
Frey JN et al (2016) Should groin recurrence still be considered as a palliative situation in vulvar cancer patients? A brief report. Int J Gynecol Cancer 26(3):575–579
Gonzalez BJ et al (2005) Long-term survival and disease recurrence in patients with primary squamous cell carcinoma of the vulva. Gynecol Oncol 97(3):828–833
Witteveen PO et al (2009) Phase II study on paclitaxel in patients with recurrent, metastatic or locallyadvanced vulvar cancer not amenable to surgery or radiotherapy: a study of the EORTC-GCG (European Organisation for Research and Treatment of Cancer-Gynaecological Cancer Group). Ann Oncol 20(9):1511–1516
Salmon EM et al (2002) Recurrent vulvar cancer. Curr Treat Options Oncol 3(2):143–153
Raffetto N et al (2003) Radiotherapy alone and chemoirradiation in recurrent squamous cell carcinoma of the vulva. Anticancer Res 23(3):3105–3108
Durrant KR et al (1990) Bleomycin, methotrexate, and CCNU in advanced in operable squamous cell carcinoma of the vulva: a phase II study of the EORTC Gynaecological Cancer Cooperative Group (GCCG). Gynecol Oncol 37:359–362
Benedetti-Panici P et al (1992) Cisplatin (P), bleomycin(B), and methotrexate (M) preoperative chemotherapy in locally advanced vulvar carcinoma. Gynecol Oncol 50:49–53
Geisler JP et al (2006) Neoadjuvant chemotherapy in vulvar cancer: avoiding primary exenteration. Gynecol Oncol 100:53–57
Wagenaar HC et al (2001) Bleomycin, methotrexate, and CCNU in locally advanced or recurrent, inoperable squamous-cell carcinoma of the vulva: an EORTC Gynaecological Cancer Cooperative Group study. Gynecol Oncol 81:348
Thigpen JT et al (1986) Phase II trials of cisplatin and piperazinedione in advanced or recurrent squamous cell carcinoma of the vulva: a Gynecologic Oncology Group study. Gynecol Oncol 23:358–363
Muss HB et al (1989) Mitoxantrone in the treatment of advanced vulvar and vaginal carcinoma. A Gynecologic Oncology Group study. Am J Clin Oncol 12:142–144
Deppe G et al (1979) Chemotherapy of squamous cell carcinoma of the vulva: a review. Gynecol Oncol 7:345–348
Monk BJ et al (2009) Phase III trial of four cisplatin-containing doublet combinations in stage IVB, recurrent, or persistent cervical carcinoma: a Gynecologic Oncology Group Study. J Clin Oncol 27:4649–4655
Johnson GA et al (1997) Epidermal growth factor receptor in vulvar malignancies and its relationship to metastasis and patient survival. Gynecol Oncol 65:425–429
Woelber L et al (2012) EGFR gene copy number increase in vulvar carcinomas is linked with poor clinical outcome. J Clin Pathol 65:133–139
Horowitz NS et al (2012) Phase II trial of erlotinib in women with squamous cell carcinoma of the vulva. Gynecol Oncol 127:141–146
Shields LBE et al (2019) Pembrolizumab in recurrent squamous cell carcinoma of the vulva: case report and review of the literature. Gynecol Obstet Invest 84(1):94–98. https://doi.org/10.1159/000491090(Epub 2018 Jul 17)
Kottke-Marchant K et al (1992) Early brain metastases in endometrial carcinoma. Gynecol Oncol 41:67–73
Mahmoud-Ahmed AS et al (2002) Brain metastases from gynecological cancers: factors that affect overall survival. Technol Cancer Res Treat 1:305–310
Patchell RA et al (1990) A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med 322:494–500
Noordijk EM et al (1994) The choice of treatment of single brain metastasis should be based on extracranial tumor activity and age. Int J Radiut Oncol Biol Phys 29:711–717
Sause WT et al (1990) Solitary brain metastasis: results of RTOG/SWOG protocol evaluation surgery + RT versus RT alone. Am J Clin Oncol 13:427–432
Smalley SR et al (1992) Resection of solitary brain metastasis: role of adjuvant radiation and prognostic variables in 229 patients. J Neurosurg 77:531–540
Zimm S et al (1981) Intracerebral metastases in solid-tumor patients: natural history and results of treatment. Cancer 48:384–394
Markesbery WR et al (1978) Treatment for patients with cerebral metastases. Arch Neurol 35:754–756
Ruderman NB et al (1965) Use of glucocorticoids in the palliative treatment of met. brain tumors. Cancer 18:298–306
Bindal RK et al (1993) Surgical treatment of multiple brain metastases. J Neurosurg 79:210–216
Loeffler JS et al (1990) The treatment of recurrent brain metastases with stereotactic radiosurgery. J Clin Oncol 8:576–582
Tangjitgamil S et al (2004) Role of surgical resection for lung, liver, and central nervous system metastases in patients with gynecological cancer: a literature review. Int J Gynecol Cancer 14:399–422
Hatiboglu MA et al (2005) Brain metastasis from an adenoid cystic carcinoma of the Bartholin gland: case report. J Neurosurg 102(3):543–546
Ferrandina G et al (2005) Skull metastasis in primary vulvar adenocarcinoma of the Bartholin’s gland: a case report. Gynecol Oncol 98:322–324
Author information
Authors and Affiliations
Contributions
All authors have made substantial intellectual contributions to the article, provided critical feedback and given approval of the final manuscript to be published. LW and AJ took the lead in writing the manuscript and were responsible for the conception/design of the article as well as for data collection and interpretation. MB, DG and AG participated in the acquisition of the data and reviewing the literature. JS and CP helped to draft the manuscript by taking responsibility in the analysis and interpretation of the patients data. KP, VM and BS supervised the project by providing substantial feedback and revising it critically for intellectual content.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jaeger, A., Biermann, M., Prieske, K. et al. Cerebral metastasis in recurrent squamous cell carcinoma of the vulva: case report and review of the literature. Arch Gynecol Obstet 301, 327–332 (2020). https://doi.org/10.1007/s00404-019-05403-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-019-05403-6