Abstract
Objective
The objective of this study was to determine if high-dose antibiotic prophylaxis with cefazolin decreases the risk of surgical site infection (SSI) after a cesarean delivery.
Methods
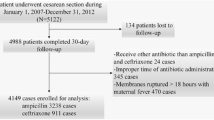
We performed a retrospective cohort study of women who underwent a cesarean section. Two preoperative antibiotic regimens were compared: low dose versus high dose. The primary outcome was SSI. A sample size of 343 patients per group was calculated for a 50% reduction in risk for SSI.
Results
Seven hundred and thirty women were included with an incidence of SSI of 5%. Women who received the high-dose antibiotic regimen had lower rates of risk factors for SSI. The only exception was skin incision closure with staples. The rate of SSI did not differ between the low-dose and high-dose groups, even after adjusting for confounding variables [aOR 1.78, 95% CI (0.82–3.9)].
Conclusions
Higher doses of antibiotic prophylaxis did not decrease the rates of SSI after cesarean delivery.
Similar content being viewed by others
References
Betran AP, Ye J, Moller AB, Zhang J, Gulmezoglu AM, Torloni MR (2016) The increasing trend in caesarean section rates: global, regional and national estimates: 1990–2014. PLoS ONE 11:e0148343
Gibbs RS (1980) Clinical risk factors for puerperal infection. Obstet Gynecol 55:178s–184s
Kirkland KB, Briggs JP, Trivette SL, Wilkinson WE, Sexton DJ (1999) The impact of surgical-site infections in the 1990s: attributable mortality, excess length of hospitalization, and extra costs. Infect Control Hosp Epidemiol 20:725–730
Smaill FM, Grivell RM (2014) Antibiotic prophylaxis versus no prophylaxis for preventing infection after cesarean section. Cochrane Database Syst Rev 2014:Cd007482
Mackeen AD, Packard RE, Ota E, Berghella V, Baxter JK (2014) Timing of intravenous prophylactic antibiotics for preventing postpartum infectious morbidity in women undergoing cesarean delivery. Cochrane Database Syst Rev 2014:9516
Currier JS, Tosteson TD, Platt R (1993) Cefazolin compared with cefoxitin for cesarean section prophylaxis: the use of a two-stage study design. J Clin Epidemiol 46:625–630
ACOG Practice Bulletin No (2011) 120: Use of prophylactic antibiotics in labor and delivery. Obstet Gynecol 117:1472–1483
Young OM, Shaik IH, Twedt R et al (2015) Pharmacokinetics of cefazolin prophylaxis in obese gravidae at time of cesarean delivery. Am J Obstet Gynecol 213(541):e1–e7
Maggio L, Nicolau DP, DaCosta M, Rouse DJ, Hughes BL (2015) Cefazolin prophylaxis in obese women undergoing cesarean delivery: a randomized controlled trial. Obstet Gynecol 125:1205–1210
Stitely M, Sweet M, Slain D et al (2013) Plasma and tissue cefazolin concentrations in obese patients undergoing cesarean delivery and receiving differing pre-operative doses of drug. Surg Infect (Larchmt) 14:455–459
Ahmadzia HK, Patel EM, Joshi D et al (2015) Obstetric surgical site infections: 2 grams compared with 3 grams of cefazolin in morbidly obese women. Obstet Gynecol 126:708–715
Horan TC, Gaynes RP, Martone WJ, Jarvis WR, Emori TG (1992) CDC definitions of nosocomial surgical site infections, 1992: a modification of CDC definitions of surgical wound infections. Am J Infect Control 20:271–274
Bratzler DW, Dellinger EP, Olsen KM et al (2013) Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm 70:195–283
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
MLR project development, data collection, manuscript writing, data analysis. CO project development, data collection. TR project development, data collection. MA project development, data collection. NS project development, data collection. JV project development, data collection. GO project development, manuscript writing, data analysis. GS project development, manuscript writing, data analysis. AS project development, data collection, manuscript writing, data analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Research involving human participants
The protocol was reviewed by the University of Texas Medical Branch Galveston Hospital Institutional Review Board, our research project (IRB protocol #16-0204) and found to meet the exemption criteria and review was waived.
Informed consent
No informed consent was required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
La Rosa, M., Omere, C., Redfern, T. et al. The impact of low-dose versus high-dose antibiotic prophylaxis regimens on surgical site infection rates after cesarean delivery. Arch Gynecol Obstet 301, 69–73 (2020). https://doi.org/10.1007/s00404-019-05370-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-019-05370-y