Abstract
Purpose
This study tested the ability of pycnogenol, an extract from the bark of the French maritime pine (Pinus pinaster), to prevent intra-abdominal adhesions.
Methods
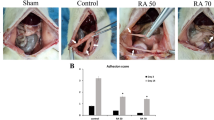
Thirty female Wistar albino rats were separated randomly into three equal groups: Group (1) the control group, which underwent surgery, but was given no drug; Group (2) given 10 mg/kg of pycnogenol dissolved in normal saline intraperitoneally for 10 days after surgery; and Group (3) given 0.1 mL of normal saline for 10 days intraperitoneally after surgery. On post-operative day 10, all of the animals were killed and any adhesions were evaluated macroscopically and histopathologically.
Results
The macroscopic adhesion scores (mean ± SD) for Groups 1, 2, and 3 were 2.5 ± 0.53, 0.60 ± 0.70, and 2.0 ± 0.82, respectively. The macroscopic adhesion score was significantly lower in Group 2 than in Groups 1 and 3 (p < 0.001). All three components of the histopathological evaluation (inflammation, fibrosis, and neovascularization) were significantly lower in Group 2 than in Groups 1 or 3 (p < 0.001, p < 0.001, and p = 0.004, respectively).
Conclusions
Pycnogenol was found to be effective at preventing surgery-related adhesions in an animal model.

Similar content being viewed by others
References
De Wilde RL, Bakkum EA, Brölmann H et al (2014) Consensus recommendations on adhesions (version 2014) for the ESGE adhesions research working group (European Society for Gynecological Endoscopy): an expert opinion. Arch Gynecol Obstet 290(3):581–582
Menzies D, Ellis H (1990) Intestinal obstruction from adhesions—how big is the problem? Ann R Coll Surg Engl 72(1):60–63
Weibel MA, Majno G (1973) Peritoneal adhesions and their relation to abdominal surgery. A postmortem study. Am J Surg 126(3):345–353
Brochhausen C, Schmitt VH, Planck CN et al (2012) Current strategies and future perspectives for intraperitoneal adhesion prevention. J Gastrointest Surg 16(6):1256–1274
Liakakos T, Thomakos N, Fine PM, Dervenis C, Young RL (2001) Peritoneal adhesions: etiology, pathophysiology, and clinical significance. Recent advances in prevention and management. Dig Surg 18(4):260–273 Review
Kamel Remah M (2010) Prevention of postoperative peritoneal adhesions. Eur J Obstet Gynecol Reprod Biol 150:111–118
Ray NF, Denton WG, Thamer M, Henderson SC, Perry S (1998) Abdominal adhesiolysis: inpatient care and expenditures in the United States in 1994. J Am Coll Surg 186(1):1–9
Arung W, Meurisse M, Detry O (2011) Pathophysiology and prevention of postoperative peritoneal adhesions. World J Gastroenterol 17(41):4545–4553
Fletcher NM, Abuanzeh S, Saed MG, Diamond MP, Abu-Soud HM, Saed GM (2014) Nicotinamide adenine dinucleotide phosphate oxidase expression is differentially regulated to favor a pro-oxidant state that contributes to postoperative adhesion development. Reprod Sci 21(8):1050–1059
Maimoona A, Naeem I, Saddiqe Z, Jameel K (2011) A review on biological, nutraceutical and clinical aspects of French maritime pine bark extract. J Ethnopharmacol 133(2):261–277
Iravani S, Zolfaghari B (2011) Pharmaceutical and nutraceutical effects of Pinus pinaster bark extract. Res Pharm Sci 6(1):1–11
Rohdewald PA (2002) review of the French maritime pine bark extract (Pycnogenol), a herbal medication with a diverse clinical pharmacology. Int J Clin Pharmacol Ther 40(4):158–168 Review
Petrassi C, Mastromarino A, Spartera C (2000) PYCNOGENOL in chronic venous insufficiency. Phytomedicine 7(5):383–388
Peng YJ, Lee CH, Wang CC, Salter DM, Lee HS (2012) Pycnogenol attenuates the inflammatory and nitrosative stress on joint inflammation induced by urate crystals. Free Radic Biol Med 52(4):765–774
Torras MA, Faura CA, Schönlau F, Rohdewald P (2005) Antimicrobial activity of Pycnogenol. Phytother Res 19(7):647–648
Evans DM, Mc Aree K, Guyton DP (1993) Dose dependency and wound healing aspects of the use of tissue plasminogen activator in the prevention of intra-abdominal adhesions. Am J Surg 165:229–232
Holmdahl L, Ivarsson ML (1999) The role of cytokines, coagulation, and fibrinolysis in peritoneal tissue repair. Eur J Surg 165(11):1012–1019
Awonuga AO, Belotte J, Abuanzeh S, Fletcher NM, Diamond MP, Saed GM (2014) Advances in the pathogenesis of adhesion development: the role of oxidative stress. Reprod Sci 21(7):823–836
Fletcher NM, Jiang ZL, Diamond MP, Abu-Soud HM, Saed GM (2008) Hypoxia-generated superoxide induces the development of the adhesion phenotype. Free Radic Biol Med 45(4):530–536
Herrick SE, Mutsaers SE, Ozua P et al (2000) Human peritoneal adhesions are highly cellular, innervated, and vascularized. J Pathol 192(1):67–72
Epstein JC, Wilson MS, Wilkosz S, Ireland G, O’Dwyer ST, Herrick SE (2006) Human peritoneal adhesions show evidence of tissue remodeling and markers of angiogenesis. Dis Colon Rectum 49(12):1885–1892
Binnebösel M, Klink CD, Serno J et al (2011) Chronological evaluation of inflammatory mediators during peritoneal adhesion formation using a rat model. Langenbecks Arch Surg 396(3):371–378
Keleidari B, Mahmoudieh M, Bahrami F, Mortazavi P, Aslani RS, Toliyat SA (2014) The effect of vitamin A and vitamin C on postoperative adhesion formation: a rat model study. J Res Med Sci 19(1):28–32
de la Portilla F, Ynfante I, Bejarano D et al (2004) Prevention of peritoneal adhesions by intraperitoneal administration of vitamin E: an experimental study in rats. Dis Colon Rectum 47(12):2157–2161
Chu DI, Lim R, Heydrick S et al (2011) N-acetyl-l-cysteine decreases intra-abdominal adhesion formation through the upregulation ofperitoneal fibrinolytic activity and antioxidant defenses. Surgery 149(6):801–812
Hızlı D, Hızlı F, Köşüş A et al (2014) Effect of Hypericum perforatum on intraperitoneal adhesion formation in rats. Arch Med Sci. 10(2):396–400
Yetkin G, Uludag M, Citgez B, Karakoc S, Polat N, Kabukcuoglu F (2009) Prevention of peritoneal adhesions by intraperitoneal administration of vitamin E and human amniotic membrane. Int J Surg 7(6):561–565
Noda Y, Anzai K, Mori A, Kohno M, Shinmei M, Packer L (1997) Hydroxyl and superoxide anion radical scavenging activities of natural source antioxidants using the computerized JES-FR30 ESR spectrometer system. Biochem Mol Biol Int 42(1):35–44
van Jaarsveld H, Kuyl JM, Schulenburg DH, Wiid NM (1996) Effect of flavonoids on the outcome of myocardial mitochondrial ischemia/reperfusion injury. Res Commun Mol Pathol Pharmacol 91(1):65–75
Sivonová M, Zitnanová I, Horáková L et al (2006) The combined effect of pycnogenol with ascorbic acid and trolox on the oxidation of lipids and proteins. Gen Physiol Biophys 25(4):379–396
Schäfer A, Chovanová Z, Muchová J, Sumegová K, Liptáková A, Duracková Z, Högger P (2006) Inhibition of COX-1 and COX-2 activity by plasma of human volunteers after ingestion of French maritime pine bark extract (Pycnogenol). Biomed Pharmacother 60(1):5–9
Saliou C, Rimbach G, Moini H et al (2001) Solar ultraviolet-induced erythema in human skin and nuclear factor-kappa-B-dependent gene expression in keratinocytes are modulated by a French maritime pine bark extract. Free Radic Biol Med 30(2):154–160
Peng Q, Wei Z, Lau BH (2000) Pycnogenol inhibits tumor necrosis factor-alpha-induced nuclear factor kappa B activation and adhesion molecule expression in human vascular endothelial cells. Cell Mol Life Sci 57(5):834–841
Maia H Jr, Haddad C, Casoy J (2013) Combining oral contraceptives with a natural nuclear factor-kappa B inhibitor for the treatment of endometriosis-related pain. Int J Womens Health 21(6):35–39
Blazsó G, Gábor M, Schönlau F, Rohdewald P (2004) Pycnogenol accelerates wound healing and reduces scar formation. Phytother Res 18(7):579–581
Değer KC, Şeker A, Özer I et al (2013) The effects of Pycnogenol® on colon anastomotic healing in rats given preoperative irradiation. Int J Surg 11(9):983–988
Belcaro G, Cesarone MR, Errichi BM et al (2005) Venous ulcers microcirculatory improvement and faster healing with local use of Pycnogenol. Angiology 56(6):699–705
http://abc.herbalgram.org/site/DocServer/Pycnog_FullMono120809_LOW.pdf?docID=1741. Accessed 24 Feb 2015
Rodhewald P (2005) Pycnogenol®—scientific file-section 19. Horphag Research, Geneva
Maritim A, Dene BA, Sanders RA, Watkins JB 3rd (2003) Effects of pycnogenol treatment on oxidative stress in streptozotocin-induced diabetic rats. J Biochem Mol Toxicol 17(3):193–199
Taner G, Aydın S, Bacanlı M et al (2014) Modulating effects of pycnogenol® on oxidative stress and DNA damage ınduced by sepsis in rats. Phytother Res 28(11):1692–1700
Acknowledgments
We thank Assistant Professor Furuzan Kokturk, Bulent Ecevit University School of Medicine, Department of Biostatistics, for the statistical analysis of the data, and Doctor Humeyra Cicekler for her technical support.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahbaz, A., Aynioglu, O., Isik, H. et al. Pycnogenol prevents peritoneal adhesions. Arch Gynecol Obstet 292, 1279–1284 (2015). https://doi.org/10.1007/s00404-015-3764-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-015-3764-4