Abstract
Considering the known disparities in racial representation in psoriasis clinical trials, this study sought to characterize travel distance and time to reach a psoriasis clinical trial site as a potential barrier to trial participation for multiple demographic and geographic variables. We determined travel distance and time from every census tract population center in the United States to the nearest psoriasis clinical trial site using ArcGIS and linked travel estimates to demographic characteristics in each census tract based on 2020 American Community Survey. The average distance and time traveled to reach a psoriasis clinical trial site nationally were 45.6 miles and 51.8 min, respectively. Urban residence and Northeast location had significantly lower travel distance and time relative to their geographic counterparts. Travel burden was significantly greater among Native American and Black races, individuals without college education and Veterans Affairs beneficiaries relative to their counterparts. These findings reveal disparate access regarding rurality, race, education and insurance type, which may encourage investigators to increase travel funding for underrepresented groups and diversity recruitment efforts to promote access to psoriasis clinical trials.

Similar content being viewed by others
References
Ding J, Joseph M, Chawla S et al (2022) Disparities in psoriasis clinical trials: a cross-sectional analysis. J Am Acad Dermatol 87(6):1386–1389
Feng H, Berk-Krauss J, Feng PW, Stein JA (2018) Comparison of dermatologist density between urban and rural counties in the United States. JAMA Dermatol 154(11):1265–1271
Armstrong AW, Mehta MD, Schupp CW, Gondo GC, Bell SJ, Griffiths CEM (2021) Psoriasis prevalence in adults in the United States. JAMA Dermatol 157(8):940–946
Hodges WT, Bhat T, Raval NS et al (2021) Biologics utilization for psoriasis is lower in black compared with white patients. Br J Dermatol 185(1):207–209
Wolfe MK, McDonald NC, Holmes GM (2020) Transportation barriers to health care in the United States: findings from the national health interview survey, 1997–2017. Am J Public Health 110(6):815–822
Funding
None.
Author information
Authors and Affiliations
Contributions
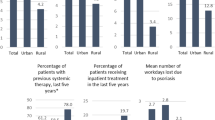
JM and EJB performed data collection. JM performed statistical analysis and prepared Fig. 1. All authors wrote and reviewed the main manuscript text.
Corresponding author
Ethics declarations
Conflict of interest
Hao Feng has consulted for Cytrellis Biosystems, Inc. and Soliton, Inc. The other authors report no conflicts of interest.
IRB approval status
This study did not qualify as human subjects research; therefore, institutional review board approval was not required at the University of Connecticut Health Center.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Masison, J., Beltrami, E.J. & Feng, H. Differential patient travel distance and time to psoriasis clinical trial sites. Arch Dermatol Res 315, 2359–2363 (2023). https://doi.org/10.1007/s00403-023-02633-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-023-02633-2