Abstract
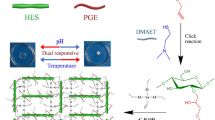
In this study, a novel type of temperature/pH dual responsive polymer PyHES- DMAET ((2-hydroxy-3-(2-propynyloxy) propyl hydroxyethyl starch (PyHES))-2-(dimethylamino) ethanethiol (DMAET)) was synthesized. First, the temperature-responsive polymer PyHES was prepared via hydrophobic modification of hydroxyl groups in hydroxyethyl starch (HES) with propynylglycidyl ether (PGE) subsequently; pH-responsive tertiary amine group was connected to the propynyl group via a thiol-yne click reaction. Because PyHES-DMAET has pH-responsive amino groups and hydrophobic thioether groups, its aqueous solution exhibits excellent temperature/pH dual sensitivity, i.e., a good transference between the hydrophobic (or self-assembly) and hydrophilic (or swelling) state along as a result of changing temperature/pH values; these properties can be exploited, for hydrophobic drug release. The drug release reached 96% at 37 °C and a pH of 6.5. The drug loading capacity of PyHES-DMAET was increased by increasing the degree of substitution (DS) of the hydrophobic propynyl groups in the PyHES. The highest drug loading capacity for doxorubicin (DOX) achieved in this study was 33 wt%.









Similar content being viewed by others
References
Maji T, Banerjee S, Biswas Y, Mandal TK (2015) Dual-stimuli-responsive L -serine-based Zwitter ionic UCST-TypePolymer with tunable thermosensitivity. Macromolecules 48:4957–4966
Zhao Y, Ce S, Yang X, Shen B, Sun Y, Chen Y, Xiaowei X, Sun H, Kui Y, Yang B, Lin Q (2016) pH- and temperature-sensitive hydrogel nanoparticles with dual photoluminescence for bioprobes. ACS Nano 10:5856–5863
Yi L, Terrell JL, Tsao C-Y, Hsuan-Chen W, Javvaji V, Kim E, Yi C, Wang Y, Ulijn RV, Raghavan SR, Rubloff GW, Bentley WE, Payne GF (2012) Biofabricating multifunctional soft matter with enzymes and stimuli-responsive materials. Adv Funct Mater 22:3004–3012
Bogomolova A, Kaberov L, Sedlacek O, Filippov SK, Stepanek P, Král V, Wang XY, Liu SL, Ye XD, Hruby M (2016) Double stimuli-responsive polymer systems: how to use crosstalk between pH- and thermosensitivity for drug depots. Eur Polym J 84:54–64. https://doi.org/10.1016/j.eurpolymj.2016.09.010
Jian-Bo Q, Xu Y-L, Liu J-Y, Zeng J-B, Chen Y-L, Zhou W-Q, Liu J-G (2016) Thermo- and pH-responsive polymer brushes-grafted gigaporous polystyrene microspheres as a high-speed protein chromatography matrix. J Chromatogr A 1441:60–67
Eggers S, Lauterbach F, Abetz V (2016) Synthesis and self-assembly of high molecular weight polystyrene-block-poly[2-(N-morpholino)ethyl methacrylate]: a story about microphase separation, amphiphilicity, and stimuli-responsivity. Polymer 107:357–367. https://doi.org/10.1016/j.polymer.2016.04.066
Jiang J, Ma Y, Cui Z (2017) Smart foams based on dual stimuli-responsive surfactant. Colloids Surf A Physicochem Eng Asp 513:287–291. https://doi.org/10.1016/j.colsurfa.2016.10.056
Deen GR (2012) Solution properties of water-soluble smart poly(N-acryloyl-N′-ethyl piperazine-co-methyl methacrylate). Polymer 4(4):32–45. https://doi.org/10.3390/polym4010032
Xiao CS, Cheng YL, Zhang Y, Ding JX, He CL, Zhuang XL (2014) Side chain impacts on pH- and thermo-responsiveness of tertiary amine functionalized polypeptides. J Polym Sci, Part A: PolymChem 52(5):671–679. https://doi.org/10.1002/pola.27048
Luo C, Wenxin F, Li Z, Zhao B (2016) Multi-responsive polymethacrylamide homopolymers derived from tertiary amine-modified l-alanine. Polymer 10:319–327
Liu M, Hongliang D, Zhang W, Zhai G (2017) Internal stimuli-responsive nanocarriers for drug delivery: design strategies and applications. Mater Sci Eng, C 71:1267–1280. https://doi.org/10.1016/j.msec.2016.11.030
Liang G, Kong T, Huo Y (2016) Dual thermoresponsive and pH-responsive poly(vinyl alcohol) derivatives: synthesis, Phase Transition Study, and Functional Applications. Macromolecules 49:7478–7489
Patra S, Roy E, Karfa P, Kumar S, Madhuri R, Sharma PK (2015) Dual-responsive polymer coated superparamagnetic nanoparticle for targeted drug delivery and hyperthermia treatment. ACS Appl Mater Interfaces 7:9235–9246
Bai Y, Zhang Z, Zhang A, Chen L, He C, Zhuang X, Chen X (2012) Novel thermo- and pH-responsive hydroxypropyl cellulose- and poly (l-glutamicacid)-based microgels for oral insulin controlled release. Carbohydr Polym 89(4):1207–1121. https://doi.org/10.1016/j.carbpol.2012.03.095
He M, Chu C-C (2013) Dual stimuli responsive glycidyl methacrylate chitosan-quaternary ammonium hybrid hydrogel and its bovine serum albumin release. J Appl Polym Sci 130:3736–3745
Wei Q-B, Luo Y-L, Feng F, Zhang Y-Q, Ma R-X (2013) Synthesis, characterization, and swelling kinetics of ph-responsive and temperature-responsive carboxymethyl chitosan/polyacrylamide hydrogels. J Appl Polym Sci 129:806–814
Alvarez-Lorenzo C, Blanco-Fernandez B, Puga AM, Concheiro A (2013) Crosslinked ionic polysaccharides for stimuli-sensitive drug delivery. Adv Drug Deliv Rev 65(9):1148–1171. https://doi.org/10.1016/j.addr.2013.04.016
Lin M, Kang H, Liu R, Huang Y (2010) Smart assembly behaviors of hydroxypropylcellulose-graft-poly(4-vinylpyridine) copolymers in aqueous solution by thermo and pH stimuli. Langmuir 26:18519–18525
Carreira AS, Gonçalves FAMM, Mendonça PV, Gil MH, Coelho JFJ (2010) Temperature and pH responsive polymers based on chitosan: applications and new graft copolymerization strategies based on living radical polymerization. Carbohydr Polym 80(3):618–630. https://doi.org/10.1016/j.carbpol.2009.12.047
Yuan W, Zou H, Shen J (2016) Amphiphilic graft copolymers with ethyl cellulose backbone: synthesis, self-assembly and tunable temperature–CO2 response. Carbohydr Polym 136:216–223. https://doi.org/10.1016/j.carbpol.2015.09.052
Xu Y, Benzhi J, Zhang S (2014) Novel pH- and temperature-responsive polymer: tertiary amine starch ether. Carbohydr Polym 114:530–536
Lowe AB (2010) Thiol-ene “click” reactions and recent applications in polymer and materials synthesis. Polym Chem 1(1):17–36. https://doi.org/10.1039/B9PY00216B
Lee J, McGrath AJ, Hawker CJ, Kim B-S (2016) pH-tunable thermoresponsive PEO-based functional polymers with pendant amine groups. ACS Macro Lett 5:1391–1396
Wang Y, Heinze T, Zhang K (2016) Stimuli-responsive nanoparticles from ionic cellulose derivatives. Nano 8:648–657
Jamróz-Piegza M, Utrata-Wesołek A, Trzebicka B, Dworak A (2006) Hydrophobic modification of high molar mass polyglycidol to thermosensitive polymers. Eur Polym J 42(10):2497–2506. https://doi.org/10.1016/j.eurpolymj.2006.04.017
Kuo C-Y, Don T-M, Hsu S-C, Lee C-F, Chiu W-Y, Huang C-Y (2016) Thermo- and pH-induced self-assembly of P(AA-b-NIPAAm-b-AA) triblock copolymers synthesized via RAFT polymerization. J Polym Sci Part A: Polym Chem 54:1109–1118
Liu S, Armes SP (2003) Synthesis and aqueous solution behavior of pH-responsive schizophrenic diblock copolymer. Langmuir 19(10):4432–4438. https://doi.org/10.1021/la020951l
Park HW, Jin H-S, Yang SY, Kim J-D (2009) Tunable phase transition behaviors of pH-sensitive polyaspartamides having various cationic pendant groups. Colloid Polym Sci 287(8):919–926. https://doi.org/10.1007/s00396-009-2046-8
Funding
This work was supported by the National Natural Science Foundation of China (21376041, 21076033, 21536002), Fund for innovative research groups of the National Natural Science Fund Committee of Science (21421005), the Fundamental Research Funds for the Central Universites (No.DUT15ZD224, DUT2016TB12) and Open Fund of Key Laboratory of Biotechnology and Bioresources Utilization (Dalian Minzu University), State Ethnic Affairs Commission & Ministry of Education, China (KF2015004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Jong, K., Ju, B., Xiu, J. et al. Temperature and pH dual responsive 2-(dimethylamino)ethanethiol modified starch derivatives via a thiol-yne reaction for drug delivery. Colloid Polym Sci 296, 627–636 (2018). https://doi.org/10.1007/s00396-018-4284-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-018-4284-0