Abstract
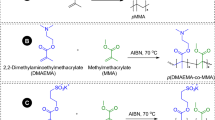
Copolymers of poly(ε-caprolactone) containing zwitterionic sulfobetaine may be used as biomedical materials. The interaction of these materials with proteins is rarely reported. In this study, amphiphilic copolymers PCL20-PDEAS6, PCL20-PEG200-PDEAS6, and PCL20-PEG400-PDEAS6, which contained poly{N-[2-(methacryloyloxy)ethyl]-N,N-diethyl-N-(3-sulfopropyl)-ammonium} (PDEAS), were designed and synthesized in order to explore interaction of these materials and their micelles with proteins. The structure of the copolymers was confirmed by 1H-NMR, Fourier transform infrared spectroscopy (FTIR), and element analysis (EA). The copolymers could form either films or micelles. The adsorption of bovine serum albumin (BSA) and fibrinogen (Fg) on the copolymer films and their micelles was studied. The results showed that BSA adsorbed on the films presented different tendencies from their micelle formulation. The protein adsorption may be related to the hydration in the shell of the micelles and the electrostatic interaction between the charges of zwitterion and the protein. The reason for the different amounts of protein adsorption between film and micelle formulation is the difference of molecular chain arrangement and charge distribution of PCLDEAS copolymer.









Similar content being viewed by others
References
Sakata S, Inoue Y, Ishihara K (2015) Molecular interaction forces generated during protein adsorption to well-defined polymer brush surfaces. Langmuir: ACS J Surf Colloids 31(10):3108–14. doi:10.1021/acs.langmuir.5b00351
Hayward JA, Chapman D (1984) Biomembrane surfaces as models for polymer design: the potential for haemocompatibility. Biomaterials 5(3):135–42. doi:10.1016/0142-9612(84)90047-4
Sheng Y, Liu C, Yuan Y, Tao X, Yang F, Shan X et al (2009) Long-circulating polymeric nanoparticles bearing a combinatorial coating of PEG and water-soluble chitosan. Biomaterials 30(12):2340–8. doi:10.1016/j.biomaterials.2008.12.070
Vonarbourg A, Passirani C, Saulnier P, Benoit JP (2006) Parameters influencing the stealthiness of colloidal drug delivery systems. Biomaterials 27(24):4356–73. doi:10.1016/j.biomaterials.2006.03.039
Ruenraroengsak P, Cook JM, Florence AT (2010) Nanosystem drug targeting: Facing up to complex realities. J Control Release: Off J Control Release Soc 141(3):265–76. doi:10.1016/j.jconrel.2009.10.032
Salvati A, Pitek AS, Monopoli MP, Prapainop K, Bombelli FB, Hristov DR et al (2013) Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nat Nanotechnol 8(2):137–43. doi:10.1038/nnano.2012.237
Ma H, Hyun J, Stiller P, Chilkoti A (2004) “Non-Fouling” oligo(ethylene glycol)-functionalized polymer brushes synthesized by surface-initiated atom transfer radical polymerization. Adv Mater 16(4):338–41. doi:10.1002/adma.200305830
Moghimi SM, Hunter AC, Murray JC (2001) Long-circulating and target-specific nanoparticles: theory to practice. Pharmacol Rev 53(2):283–318
Yao J, Wu H, Ruan Y, Guan J, Wang A, Li H (2011) “Reservoir” and “barrier” effects of ABC block copolymer micelle in hydroxyapatite mineralization control. Polymer 52(3):793–803. doi:10.1016/j.polymer.2010.12.017
Stolnik S, Illum L, Davis SS (2012) Long circulating microparticulate drug carriers. Adv Drug Deliv Rev 64:290–301. doi:10.1016/j.addr.2012.09.029
Xiao K, Luo J, Li Y, Lee JS, Fung G, Lam KS (2011) PEG-oligocholic acid telodendrimer micelles for the targeted delivery of doxorubicin to B-cell lymphoma. J Control Release: Off J Control Release Soc 155(2):272–81. doi:10.1016/j.jconrel.2011.07.018
Peng Q, Wei XQ, Shao XR, Zhang T, Zhang S, Fu N et al (2014) Nanocomplex based on biocompatible phospholipids and albumin for long-circulation applications. ACS Appl Mater Interfaces 6(16):13730–7. doi:10.1021/am503179a
Amoozgar Z, Yeo Y (2012) Recent advances in stealth coating of nanoparticle drug delivery systems. Wiley interdiscip Rev Nanomed Nanobiotechnol 4(2):219–33. doi:10.1002/wnan.1157
Knop K, Hoogenboom R, Fischer D, Schubert US (2010) Poly(ethylene glycol) in drug delivery: pros and cons as well as potential alternatives. Angew Chem 49(36):6288–308. doi:10.1002/anie.200902672
Zhang Z, Zhang M, Chen S, Horbett TA, Ratner BD, Jiang S (2008) Blood compatibility of surfaces with superlow protein adsorption. Biomaterials 29(32):4285–91. doi:10.1016/j.biomaterials.2008.07.039
Lalani R, Liu L (2011) Synthesis, characterization, and electrospinning of zwitterionic poly(sulfobetaine methacrylate). Polymer 52(23):5344–54. doi:10.1016/j.polymer.2011.09.015
Zhao C, Zhao J, Li X, Wu J, Chen S, Chen Q et al (2013) Probing structure–antifouling activity relationships of polyacrylamides and polyacrylates. Biomaterials 34(20):4714–24. doi:10.1016/j.biomaterials.2013.03.028
Ladd J, Zhang Z, Chen S, Hower JC, Jiang S (2008) Zwitterionic polymers exhibiting high resistance to nonspecific protein adsorption from human serum and plasma. Biomacromolecules 9(5):1357–61. doi:10.1021/bm701301s
Chang Y, Chen WY, Yandi W, Shih YJ, Chu WL, Liu YL et al (2009) Dual-thermo responsive phase behavior of blood compatible zwitterionic copolymers containing nonionic poly(N-isopropyl acrylamide). Biomacromolecules 10(8):2092–100. doi:10.1021/bm900208u
Chang Y, Chang WJ, Shih YJ, Wei TC, Hsiue GH (2011) Zwitterionic sulfobetaine-grafted poly (vinylidene fluoride) membrane with highly effective blood compatibility via atmospheric plasma-induced surface copolymerization. ACS Appl Mater Interfaces 3(4):1228–37. doi:10.1021/am200055k
Zhang Z, Chen S, Jiang S (2006) Dual-functional biomimetic materials: nonfouling poly(carboxybetaine) with active functional groups for protein immobilization. Biomacromolecules 7(12):3311–5. doi:10.1021/bm060750m
Yuan J, Mao C, Zhou J, Shen J, Lin SC, Zhu W et al (2003) Chemical grafting of sulfobetaine onto poly(ether urethane) surface for improving blood compatibility. Polym Int 52(12):1869–75. doi:10.1002/pi.1277
Jiang H, Wang XB, Li CY, Li JS, Xu FJ, Mao C et al (2011) Improvement of hemocompatibility of polycaprolactone film surfaces with zwitterionic polymer brushes. Langmuir: ACS J Surf Colloids 27(18):11575–81. doi:10.1021/la202101q
Cao J, Chen YW, Wang X, Luo XL (2011) Enhancing blood compatibility of biodegradable polymers by introducing sulfobetaine. J Biomed Mater Res Part A 97(4):472–9. doi:10.1002/jbm.a.33060
Cao J, Lu A, Li C, Cai M, Chen Y, Li S et al (2013) Effect of architecture on the micellar properties of poly (varepsilon-caprolactone) containing sulfobetaines. Colloids Surf B: Biointerfaces 112:35–41. doi:10.1016/j.colsurfb.2013.07.038
Cao J, Xiu KM, Zhu K, Chen YW, Luo XL (2012) Copolymer nanoparticles composed of sulfobetaine and poly(epsilon-caprolactone) as novel anticancer drug carriers. J Biomed Mater Res Part A 100(8):2079–87. doi:10.1002/jbm.a.34120
Zhai S, Ma Y, Chen Y, Li D, Cao J, Liu Y et al (2014) Synthesis of an amphiphilic block copolymer containing zwitterionic sulfobetaine as a novel pH-sensitive drug carrier. Polym Chem 5(4):1285. doi:10.1039/c3py01325a
Cao J, Zhai S, Li C, He B, Lai Y, Chen Y et al (2013) Novel pH-sensitive micelles generated by star-shape copolymers containing zwitterionic sulfobetaine for efficient cellular internalization. J Biomed Nanotechnol 9(11):1847–61. doi:10.1166/jbn.2013.1686
Chang Y, Yandi W, Chen WY, Shih YJ, Yang CC, Chang Y et al (2010) Tunable bioadhesive copolymer hydrogels of thermoresponsive poly(N-isopropyl acrylamide) containing zwitterionic polysulfobetaine. Biomacromolecules 11(4):1101–10. doi:10.1021/bm100093g
Zhu Z, Xie C, Liu Q, Zhen X, Zheng X, Wu W et al (2011) The effect of hydrophilic chain length and iRGD on drug delivery from poly(epsilon-caprolactone)-poly(N-vinylpyrrolidone) nanoparticles. Biomaterials 32(35):9525–35. doi:10.1016/j.biomaterials.2011.08.072
Rubatat L, Li C, Dietsch H, Nykänen A, Ruokolainen J, Mezzenga R (2008) Structure−properties relationship in proton conductive sulfonated polystyrene−polymethyl methacrylate block copolymers (sPS − PMMA). Macromolecules 41(21):8130–7. doi:10.1021/ma801543q
Leng C, Han X, Shao Q, Zhu Y, Li Y, Jiang S et al (2014) In situ probing of the surface hydration of zwitterionic polymer brushes: structural and environmental effects. J Phys Chem C 118(29):15840–5. doi:10.1021/jp504293r
Luengo-Alonso C, Torrado JJ, Ballesteros MP, Malfanti A, Bersani S, Salmaso S et al (2015) A novel performing PEG-cholane nanoformulation for amphotericin B delivery. Int J Pharm 495(1):41–51. doi:10.1016/j.ijpharm.2015.08.070
Seo JH, Matsuno R, Konno T, Takai M, Ishihara K (2008) Surface tethering of phosphorylcholine groups onto poly(dimethylsiloxane) through swelling—deswelling methods with phospholipids moiety containing ABA-type block copolymers. Biomaterials 29(10):1367–76. doi:10.1016/j.biomaterials.2007.11.039
Lee JH, Ju YM, Kim DM (2000) Platelet adhesion onto segmented polyurethane film surfaces modified by addition and crosslinking of PEO-containing block copolymers. Biomaterials 21:683–91
Liu P-S, Chen Q, Wu S-S, Shen J, Lin S-C (2010) Surface modification of cellulose membranes with zwitterionic polymers for resistance to protein adsorption and platelet adhesion. J Membr Sci 350(1–2):387–94. doi:10.1016/j.memsci.2010.01.015
Gombotz WR, Wang GH, Horbett TA, Hoffman AS (1991) Protein adsorption to poly(ethylene oxide) surfaces. J Biomed Mater Res 25(12):1547–62. doi:10.1002/jbm.820251211
Zhang Z, Chen S, Chang Y, Jiang S (2006) Surface grafted sulfobetaine polymers via atom transfer radical polymerization as superlow fouling coatings. J Phys Chem B 110(22):10799–804. doi:10.1021/jp057266i
Ishihara K, Nomura H, Mihara T, Kurita K, Iwasaki Y, Nakabayashi N (1998) Why do phospholipid polymers reduce protein adsorption? J Biomed Mater Res 39(2):323–30. doi:10.1002/(sici)1097-4636(199802)39:2<323::aid-jbm21>3.0.co;2-c
Song W, Chen H (2007) Protein adsorption on materials surfaces with nano-topography. Chin Sci Bull 52(23):3169–73. doi:10.1007/s11434-007-0504-6
Acknowledgments
This work was supported by the National Natural Science Foundation of China (nos. 51473099 and 51273125). We would also like to appreciate our laboratory members for the generous help and gratefully acknowledge the Analytical and Testing Center at Sichuan University for the tests.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Lu, A., Li, C., Wu, Z. et al. The interaction between poly(ε-caprolactone) copolymers containing sulfobetaines and proteins. Colloid Polym Sci 294, 1887–1899 (2016). https://doi.org/10.1007/s00396-016-3942-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-016-3942-3