Abstract
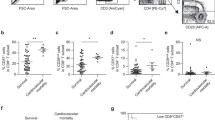
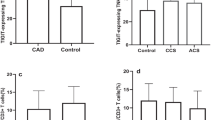
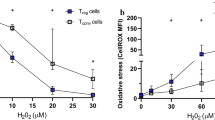
In acute coronary syndrome (ACS), T cell abnormalities are associated to a worse outcome. Loss of inhibitory activity of CD31, an Ig-like adhesion molecule, on peripheral leukocytes has been found to enhance atherosclerosis in experimental models. In this study, we examined the expression of CD31 on T cells, and its role on TCR signaling in 35 patients with non-ST elevation ACS, in 35 patients with stable angina (SA), and in 35 controls. Furthermore, 10 ACS and 10 SA patients were re-analyzed at 1-year follow-up. Flow-cytometry analysis showed that in ACS patients, CD31 expression was reduced on total CD4+ and CD4+CD28null (P < 0.001, ACS vs. SA), on naïve (P < 0.001, ACS vs. SA) and on central-memory and effector-memory CD4+ T cells (P < 0.05, ACS vs. SA and controls). The immunomodulatory effect of CD31 on TCR signaling of CD4+ and CD4+CD28null T cells, was lower in ACS than SA patients (P < 0.05, for both comparisons). At 1-year follow-up, CD31 expression and function increased in ACS becoming similar to that found in SA. CD31 recruitment in the immunological synapse was lower in ACS than controls (P = 0.012). Moreover, CD31 modulated MAPK signaling and reduced the expression of T bet and Rorγ-t, necessary for Th1 and Th17 differentiation. Finally, we studied TCR signaling in CD31+ naïve and primed T cell subsets observing a different pattern of protein phosphorylation. A CD31-mediated regulatory pathway is enhanced in SA and temporarily downregulated in ACS. As CD31 modulates both T cell activation, by increasing the threshold for TCR stimulation, and T cell differentiation, it might represent a novel molecular target to treat T cell abnormalities in ACS.








Similar content being viewed by others
References
Adachi K, Davis MM (2011) T cell receptor ligation induces distinct signaling pathways in naive vs. antigen-experienced T cells. Proc Natl Acad Sci 108:1549–1554. doi:10.1073/pnas.1017340108
Caligiuri G, Groyer E, Khallou-Laschet J, Al Haj Zen A, Sainz J, Urbain D, Gaston AT, Lemitre M, Nicoletti A, Lafont A (2005) Reduced immunoregulatory CD31+ T cells in the blood of atherosclerotic mice with plaque thrombosis. Arterioscler Thromb Vasc Biol 25:1659–1664. doi:10.1161/01.ATV.0000172660.24580.b4
Caligiuri G, Paulsson G, Nicoletti A, Maseri A, Hansson GK (2000) Evidence for antigen-driven T cell response in unstable angina. Circulation 102:1114–1119. doi:10.1161/01.CIR.102.10.1114
Caligiuri G, Rossignol P, Julia P, Groyer E, Mouradian D, Urbain D, Misra N, Ollivier V, Sapoval M, Boutouyrie P, Kaveri SV, Nicoletti A, Lafont A (2006) Reduced immunoregulatory CD31+ T cells in patients with atherosclerotic abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol 26:618–623. doi:10.1161/01.ATV.0000200380.73876.d9
Crea F, Liuzzo G (2013) Pathogenesis of acute coronary syndromes. J Am Coll Cardiol 61:1–11. doi:10.1016/j.jacc.2012.07.064
Cui G, Qin X, Zhang Y, Gong Z, Ge B, Zang YQ (2009) Berberine differentially modulates the activities of ERK, p38 MAPK, and JNK to suppress Th17 and Th1 T cell differentiation in type 1 diabetic mice. J Biol Chem 284:28420–28429. doi:10.1074/jbc.M109.012674
De Palma R, Del Galdo F, Abbate G, Chiariello M, Calabró R, Forte L, Cimmino G, Papa MF, Russo MG, Ambrosio G, Giombolini C, Tritto I, Notaristefano S, Berrino L, Rossi F, Golino P (2006) Patients with acute coronary syndrome show oligoclonal T cell recruitment within unstable plaque: evidence for a local, intracoronary immunologic mechanism. Circulation 113:640–646. doi:10.1161/CIRCULATIONAHA.105.537712
Dumitriu IE, Baruah P, Finlayson CJ, Loftus IM, Antunes RF, Lim P, Bunce N, Kaski JC (2012) High levels of costimulatory receptors OX40 and 4-1BB characterize CD4+ CD28null T cells in patients with acute coronary syndrome. Circ Res 110:857–869. doi:10.1161/CIRCRESAHA.111.261933
Dvergsten JA, Mueller RG, Griffin P, Abedin S, Pishko A, Michel JJ, Rosenkranz ME, Reed AM, Kietz DA, Vallejo AN (2013) Premature cell senescence and T cell receptor-independent activation of CD8+ T cells in juvenile idiopathic arthritis. Arthritis Rheum 65:2201–2210. doi:10.1002/art.38015
Fornasa G, Clement M, Groyer E, Gaston AT, Khallou-Laschet J, Morvan M, Guedj K, Kaveri SV, Tedgui A, Michel JB, Nicoletti A, Caligiuri G (2012) A CD31-derived peptide prevents angiotensin II-induced atherosclerosis progression and aneurysm formation. Cardiovasc Res 94:30–37. doi:10.1093/cvr/cvs076
Fornasa G, Groyer E, Clement M, Dimitrov J, Compain C, Gaston AT, Varthaman A, Khallou-Laschet J, Newman DK, Graff-Dubois S, Nicoletti A, Caligiuri G (2010) TCR stimulation drives cleavage and shedding of the ITIM receptor CD31. J Immunol 184:5485–5492. doi:10.4049/jimmunol.0902219
Giubilato S, Liuzzo G, Brugaletta S, Pitocco D, Graziani F, Smaldone C, Montone RA, Pazzano V, Pedicino D, Biasucci LM, Ghirlanda G, Crea F (2011) Expansion of CD4+ CD28null T lymphocytes in diabetic patients: exploring new pathogenetic mechanisms of increased cardiovascular risk in diabetes mellitus. Eur Heart J 32:1214–1226. doi:10.1093/eurheartj/ehq499
Goel R, Schrank BR, Arora S, Boylan B, Fleming B, Miura H, Newman PJ, Molthen RC, Newman DK (2008) Site-specific effects of PECAM-1 on atherosclerosis in LDL receptor-deficient mice. Arterioscler Thromb Vasc Biol 28:1996–2002. doi:10.1161/ATVBAHA.108.172270
Graesser D, Solowiej A, Bruckner M, Osterweil E, Juedes A, Davis S, Ruddle NH, Engelhardt B, Madri JA (2002) Altered vascular permeability and early onset of experimental autoimmune encephalomyelitis in PECAM-1-deficient mice. J Clin Invest 109:383–392. doi:10.1172/JCI13595
Groyer E, Nicoletti A, AiT Oufella H, Khallou-Laschet J, Varthaman A, Gaston AT, Thaunat O, Kaveri SV, Blatny R, Stockinger H, Mallat Z, Caligiuri G (2007) Atheroprotective effect of CD31 receptor globulin through enrichment of circulating regulatory T cells. J Am Coll Cardiol 50:344–350. doi:10.1016/j.jacc.2007.04.040
Hansson GK, Hermansson A (2011) The immune system in atherosclerosis. Nat Immunol 12:204–212. doi:10.1038/ni.2001
Indolfi C, Gasparri C, Vicinanza C, De Serio D, Boncompagni D, Mongiardo A, Spaccarotella C, Agosti V, Torella D, Curcio A (2011) Mitogen-activated protein kinases activation in T lymphocytes of patients with acute coronary syndromes. Basic Res Cardiol 106:667–679. doi:10.1007/s00395-011-0172-1
Kakizaki M, Nobori K, Watanabe H, Iino K, Ishida M, Ito H (2013) Increased circulating CD3+/CD31+ T cells in patients with acute coronary syndrome. Heart Vessels 28:566–569. doi:10.1007/s00380-012-0284-z
Kimmig S, Przybylski GK, Schmidt CA, Laurisch K, Möwes B, Radbruch A, Thiel A (2002) Two subsets of naive T helper cells with distinct T cell receptor excision circle content in human adult peripheral blood. J Exp Med 18:789–794. doi:10.1084/jem.20011756
Kishore M, Ma L, Cornish G, Nourshargh S, Marelli-Berg FM (2012) Primed T cell responses to chemokines are regulated by the immunoglobulin-like molecule CD3. PLoS ONE 7:e39433. doi:10.1371/journal.pone.0039433
Kohler S, Thiel A (2009) Life after the thymus: CD31+ and CD31− human naive CD4+ T cell subsets. Blood 113(4):769–774. doi:10.1182/blood-2008-02-139154
Lahoute C, Herbin O, Mallat Z, Tedgui A (2011) Adaptive immunity in atherosclerosis: mechanisms and future therapeutic targets. Nat Rev Cardiol 8:348–358. doi:10.1038/nrcardio.2011.62
Liuzzo G, Biasucci LM, Trotta G, Brugaletta S, Pinnelli M, Digianuario G, Rizzello V, Rebuzzi AG, Rumi C, Maseri A, Crea F (2007) Unusual CD4+ CD28null T lymphocytes and recurrence of acute coronary events. J Am Coll Cardiol 50:1450–1458. doi:10.1016/j.jacc.2007.06.040
Liuzzo G, Goronzy JJ, Yang H, Kopecky SL, Holmes DR, Frye RL, Weyand CM (2000) Monoclonal T cell proliferation and plaque instability in acute coronary syndromes. Circulation 101:2883–2888. doi:10.1161/01.CIR.101.25.2883
Liuzzo G, Montone RA, Gabriele M, Pedicino D, Giglio AF, Trotta F, Galiffa VA, Previtero M, Severino A, Biasucci LM, Crea F (2013) Identification of unique adaptive immune system signature in acute coronary syndromes. Int J Cardiol 168:564–567. doi:10.1016/j.ijcard.2013.01.009
Ma L, Mauro C, Cornish GH, Chai JG, Coe D, Fu H, Patton D, Okkenhaug K, Franzoso G, Dyson J, Nourshargh S, Marelli-Berg FM (2010) Ig gene-like molecule CD31 plays a non redundant role in the regulation of T cell immunity and tolerance. Proc Natl Acad Sci USA 107:19461–19466. doi:10.1073/pnas.1011748107
Marelli-Berg FM, Clement M, Mauro C, Caligiuri G (2013) An immunologist’s guide to CD31 function in T cells. J Cell Sci 126:2343–2352. doi:10.1242/jcs.124099
Mor A, Luboshits G, Planer D, Keren G, George J (2006) Altered status of CD4(+) CD25(+) regulatory T cells in patients with acute coronary syndromes. Eur Heart J 27:2530–2537. doi:10.1093/eurheartj/ehl222
Nakayama T, Yamashita M (2010) The TCR-mediated signaling pathways that control the direction of helper T cell differentiation. Semin Immunol 22:303–309. doi:10.1016/j.smim.2010.04.010
Narducci ML, Grasselli A, Biasucci LM, Farsetti A, Mulè A, Liuzzo G, La Torre G, Niccoli G, Mongiardo R, Pontecorvi A, Crea F (2007) High telomerase activity in neutrophils from unstable coronary plaques. J Am Coll Cardiol 50:2369–2374. doi:10.1016/j.jacc.2007.08.048
Newman PJ (1999) Switched at birth: a new family for PECAM-1. J Clin Invest 103:5–9. doi:10.1172/JCI5928
Newton-Nash DK, Newman PJ (1999) A new role for plateleT endothelial cell adhesion molecule-1 (CD31): inhibition of TCR-mediated signal transduction. J Immunol 163:682–688
Pedicino D, Giglio AF, Galiffa VA, Cialdella P, Trotta F, Graziani F, Liuzzo G (2012) Infections, immunity and atherosclerosis: pathogenic mechanisms and unsolved questions. Int J Cardiol 166:572–583. doi:10.1016/j.ijcard.2012.05.098
Privratsky JR, Newman DK, Newman PJ (2010) PECAM-1: conflicts of interest in inflammation. Life Sci 87:69–82. doi:10.1016/j.lfs.2010.06.001
Pryshchep S, Goronzy JJ, Parashar S, Weyand CM (2010) Insufficient deactivation of the protein tyrosine kinase lck amplifies T cell responsiveness in acute coronary syndrome. Circ Res 106:769–778. doi:10.1161/CIRCRESAHA.109.206052
Rincón M, Enslen H, Raingeaud J, Recht M, Zapton T, Su MS, Penix LA, Davis RJ, Flavell RA (1998) Interferon-gamma expression by Th1 effector T cells mediated by the p38 MAP kinase signaling pathway. EMBO J 17:2817–2829. doi:10.1093/emboj/17.10.2817
Wong MX, Hayball JD, Hogarth PM, Jackson DE (2005) The inhibitory co-receptor, PECAM-1 provides a protective effect in suppression of collagen-induced arthritis. J Clin Immunol 25:19–28. doi:10.1007/s10875-005-0354-7
Woodfin A, Voisin MB, Nourshargh S (2007) PECAM-1: a multi-functional molecule in inflammation and vascular biology. Arterioscler Thromb Vasc Biol 27:2514–2523. doi:10.1161/ATVBAHA.107.151456
Yonekawa K, Neidhart M, Altwegg LA, Wyss CA, Corti R, Vogl T, Grigorian M, Gay S, Lüscher TF, Maier W (2011) Myeloid related proteins activate Toll-like receptor 4 in human acute coronary syndromes. Atherosclerosis. 218:486–492. doi:10.1016/j.atherosclerosis.2011.06.020
Zal B, Kaski JC, Arno G, Akiyu JP, Xu Q, Cole D, Whelan M, Russell N, Madrigal JA, Dodi IA, Baboonian C (2004) HeaT shock protein 60-reactive CD4+ CD28null T cells in patients with acute coronary syndromes. Circulation 109:1230–1235. doi:10.1161/01.CIR.0000118476.29352.2A
Zhang WC, Wang J, Shu YW, Tang TT, Zhu ZF, Xia N, Nie SF, Liu J, Zhou SF, Li JJ, Xiao H, Yuan J, Liao MY, Cheng LX, Liao YH, Cheng X (2012) Impaired thymic export and increased apoptosis account for regulatory T cell defects in patients with non-ST segment elevation acute coronary syndrome. J Biol Chem 287:34157–34166. doi:10.1074/jbc.M112.382978
Conflict of interest
On behalf of all the authors, the corresponding author states that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
G. Liuzzo and F. Crea contributed equally.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Flego, D., Severino, A., Trotta, F. et al. Altered CD31 expression and activity in helper T cells of acute coronary syndrome patients. Basic Res Cardiol 109, 448 (2014). https://doi.org/10.1007/s00395-014-0448-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-014-0448-3