Abstract
Purpose
The consumption of refined, fructose-enriched food continuously increases and has been linked to development of obesity, especially in young population. Low-grade inflammation and increased oxidative stress have been implicated in the pathogenesis of obesity-related disorders including type 2 diabetes. In this study, we examined alterations in inflammation and antioxidative defense system in the visceral adipose tissue (VAT) of fructose-fed young female rats, and related them to changes in adiposity and insulin sensitivity.
Methods
We examined the effects of 9-week fructose-enriched diet applied immediately after weaning on nuclear factor κB (NF-κB) intracellular distribution, and on the expression of pro-inflammatory cytokines (IL-1β and TNFα) and key antioxidative enzymes in the VAT of female rats. Insulin signaling in the VAT was evaluated at the level of insulin receptor substrate-1 (IRS-1) protein and its inhibitory phosphorylation on Ser307.
Results
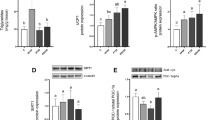
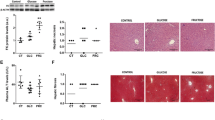
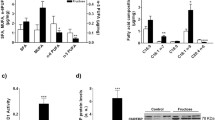
Fructose-fed rats had increased VAT mass along with increased NF-κB nuclear accumulation and elevated IL-1β, but not TNFα expression. The protein levels of antioxidative defense enzymes, mitochondrial manganese superoxide dismutase 2, and glutathione peroxidase, were reduced, while the protein content of IRS-1 and its inhibitory phosphorylation were not altered by fructose diet.
Conclusions
The results suggest that fructose overconsumption-related alterations in pro-inflammatory markers and antioxidative capacity in the VAT of young female rats can be implicated in the development of adiposity, but do not affect inhibitory phosphorylation of IRS-1.




Similar content being viewed by others
References
Havel PJ (2005) Dietary fructose: implications for dysregulation of energy homeostasis and lipid/carbohydrate metabolism. Nutr Rev 63:133–157
Stanhope KL, Schwarz JM, Keim NL, Griffen SC, Bremer AA, Graham JL, Hatcher B et al (2009) Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J Clin Invest 119:1322–1334
Tappy L, Le KA, Tran C, Paquot N (2010) Fructose and metabolic diseases: new findings, new questions. Nutrition 26:1044–1049
Bagul PK, Middela H, Matapally S, Padiya R, Bastia T, Madhusudana K, Reddy BR et al (2012) Attenuation of insulin resistance, metabolic syndrome and hepatic oxidative stress by resveratrol in fructose-fed rats. Pharmacol Res 66:260–268
Miller A, Adeli K (2008) Dietary fructose and the metabolic syndrome. Curr Opin Gastroenterol 24:204–209
Panchal SK, Poudyal H, Brown L (2012) Quercetin ameliorates cardiovascular, hepatic, and metabolic changes in diet-induced metabolic syndrome in rats. J Nutr 142:1026–1032
Qin B, Polansky MM, Anderson RA (2010) Cinnamon extract regulates plasma levels of adipose-derived factors and expression of multiple genes related to carbohydrate metabolism and lipogenesis in adipose tissue of fructose-fed rats. Horm Metab Res 42:187–193
Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, Mullany EC et al (2014) Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 384:766–781
Kahn BB, Flier JS (2000) Obesity and insulin resistance. J Clin Invest 106:473–481
Berg AH, Scherer PE (2005) Adipose tissue, inflammation, and cardiovascular disease. Circ Res 96:939–949
Hotamisligil GS (2006) Inflammation and metabolic disorders. Nature 444:860–867
Kern PA, Saghizadeh M, Ong JM, Bosch RJ, Deem R, Simsolo RB (1995) The expression of tumor necrosis factor in human adipose tissue. Regulation by obesity, weight loss, and relationship to lipoprotein lipase. J Clin Invest 95:2111–2119
Ziccardi P, Nappo F, Giugliano G, Esposito K, Marfella R, Cioffi M, D’Andrea F et al (2002) Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 105:804–809
Juge-Aubry CE, Somm E, Chicheportiche R, Burger D, Pernin A, Cuenod-Pittet B, Quinodoz P et al (2004) Regulatory effects of interleukin (IL)-1, interferon-beta, and IL-4 on the production of IL-1 receptor antagonist by human adipose tissue. J Clin Endocrinol Metab 89:2652–2658
Moschen AR, Molnar C, Enrich B, Geiger S, Ebenbichler CF, Tilg H (2011) Adipose and liver expression of interleukin (IL)-1 family members in morbid obesity and effects of weight loss. Mol Med 17:840–845
Esser N, L’Homme L, De Roover A, Kohnen L, Scheen AJ, Moutschen M, Piette J et al (2013) Obesity phenotype is related to NLRP3 inflammasome activity and immunological profile of visceral adipose tissue. Diabetologia 56:2487–2497
Donath MY, Shoelson SE (2011) Type 2 diabetes as an inflammatory disease. Nat Rev Immunol 11:98–107
Stienstra R, Tack CJ, Kanneganti TD, Joosten LA, Netea MG (2012) The inflammasome puts obesity in the danger zone. Cell Metab 15:10–18
Lagathu C, Yvan-Charvet L, Bastard JP, Maachi M, Quignard-Boulange A, Capeau J, Caron M (2006) Long-term treatment with interleukin-1beta induces insulin resistance in murine and human adipocytes. Diabetologia 49:2162–2173
Jager J, Gremeaux T, Cormont M, Le Marchand-Brustel Y, Tanti JF (2007) Interleukin-1beta-induced insulin resistance in adipocytes through down-regulation of insulin receptor substrate-1 expression. Endocrinology 148:241–251
Aye IL, Jansson T, Powell TL (2013) Interleukin-1beta inhibits insulin signaling and prevents insulin-stimulated system A amino acid transport in primary human trophoblasts. Mol Cell Endocrinol 381:46–55
Wen H, Gris D, Lei Y, Jha S, Zhang L, Huang MT, Brickey WJ et al (2011) Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat Immunol 12:408–415
Wei Y, Pagliassotti MJ (2004) Hepatospecific effects of fructose on c-jun NH2-terminal kinase: implications for hepatic insulin resistance. Am J Physiol Endocrinol Metab 287:E926–E933
Hotamisligil GS, Peraldi P, Budavari A, Ellis R, White MF, Spiegelman BM (1996) IRS-1-mediated inhibition of insulin receptor tyrosine kinase activity in TNF-alpha- and obesity-induced insulin resistance. Science 271:665–668
Kanety H, Feinstein R, Papa MZ, Hemi R, Karasik A (1995) Tumor Necrosis Factor α-induced Phosphorylation of Insulin Receptor Substrate-1 (IRS-1): possible mechanism for suppression of insulin-stimulated tyrosine phosphorylation of IRS-1. J Biol Chem 270:23780–23784
Uysal KT, Wiesbrock SM, Marino MW, Hotamisligil GS (1997) Protection from obesity-induced insulin resistance in mice lacking TNF-alpha function. Nature 389:610–614
Trede NS, Tsytsykova AV, Chatila T, Goldfeld AE, Geha RS (1995) Transcriptional activation of the human TNF-alpha promoter by superantigen in human monocytic cells: role of NF-kappa B. J Immunol 155:902–908
Hiscott J, Marois J, Garoufalis J, D’Addario M, Roulston A, Kwan I, Pepin N et al (1993) Characterization of a functional NF-kappa B site in the human interleukin 1 beta promoter: evidence for a positive autoregulatory loop. Mol Cell Biol 13:6231–6240
Cogswell JP, Godlevski MM, Wisely GB, Clay WC, Leesnitzer LM, Ways JP, Gray JG (1994) NF-kappa B regulates IL-1 beta transcription through a consensus NF-kappa B binding site and a nonconsensus CRE-like site. J Immunol 153:712–723
Mercurio F, Manning AM (1999) NF-kappaB as a primary regulator of the stress response. Oncogene 18:6163–6171
Meigs JB, Larson MG, Fox CS, Keaney JF Jr, Vasan RS, Benjamin EJ (2007) Association of oxidative stress, insulin resistance, and diabetes risk phenotypes: the Framingham Offspring Study. Diabetes Care 30:2529–2535
Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O et al (2004) Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest 114:1752–1761
Schreck R, Rieber P, Baeuerle PA (1991) Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J 10:2247–2258
Armutcu F, Ataymen M, Atmaca H, Gurel A (2008) Oxidative stress markers, C-reactive protein and heat shock protein 70 levels in subjects with metabolic syndrome. Clin Chem Lab Med 46:785–790
Keaney JF Jr, Larson MG, Vasan RS, Wilson PW, Lipinska I, Corey D, Massaro JM et al (2003) Obesity and systemic oxidative stress: clinical correlates of oxidative stress in the Framingham Study. Arterioscler Thromb Vasc Biol 23:434–439
Olusi SO (2002) Obesity is an independent risk factor for plasma lipid peroxidation and depletion of erythrocyte cytoprotectic enzymes in humans. Int J Obes Relat Metab Disord 26:1159–1164
Chrysohoou C, Panagiotakos DB, Pitsavos C, Skoumas I, Papademetriou L, Economou M, Stefanadis C (2007) The implication of obesity on total antioxidant capacity in apparently healthy men and women: the ATTICA study. Nutr Metab Cardiovasc Dis 17:590–597
Hartwich J, Goralska J, Siedlecka D, Gruca A, Trzos M, Dembinska-Kiec A (2007) Effect of supplementation with vitamin E and C on plasma hsCRP level and cobalt-albumin binding score as markers of plasma oxidative stress in obesity. Genes Nutr 2:151–154
Rayssiguier Y, Gueux E, Nowacki W, Rock E, Mazur A (2006) High fructose consumption combined with low dietary magnesium intake may increase the incidence of the metabolic syndrome by inducing inflammation. Magnes Res 19:237–243
Rutledge AC, Adeli K (2007) Fructose and the metabolic syndrome: pathophysiology and molecular mechanisms. Nutr Rev 65:S13–S23
Giriş M, Doğru-Abbasoğlu S, Kumral A, Olgaç V, Koçak-Toker N, Uysal M (2014) Effect of carnosine alone or combined with α-tocopherol on hepatic steatosis and oxidative stress in fructose-induced insulin-resistant rats. J Physiol Biochem 70:385–395
Bray GA, Popkin BM (2013) Calorie-sweetened beverages and fructose: what have we learned 10 years later. Pediatr Obes 8:242–248
Zheng H, Yde CC, Arnberg K, Molgaard C, Michaelsen KF, Larnkjær A et al (2014) NMR-based metabolomic profiling of overweight adolescents: an elucidation of the effects of inter-/intra-individual differences, gender, and pubertal development. BioMed Res Int 2014:537157. doi:10.1155/2014/537157
Kovačević S, Nestorov J, Matić G, Elaković I (2014) Dietary fructose-related adiposity and glucocorticoid receptor function in visceral adipose tissue of female rats. Eur J Nutr 53:1409–1420
Moutsatsou P, Psarra AM, Tsiapara A, Paraskevakou H, Davaris P, Sekeris CE (2001) Localization of the glucocorticoid receptor in rat brain mitochondria. Arch Biochem Biophys 386:69–78
Elaković I, Nestorov J, Kovačević S, Matić G (2012) Selection of reference genes for normalization of real time PCR data in visceral adipose tissue of female rats on fructose enriched diet. Arch Biol Sci 64:1247–1259
Padiya R, Chowdhury D, Borkar R, Srinivas R, Pal Bhadra M, Banerjee SK (2014) Garlic attenuates cardiac oxidative stress via activation of PI3 K/AKT/Nrf2-Keap1 pathway in fructose-fed diabetic rat. PLoS ONE 9:e94228
Velickovic N, Djordjevic A, Vasiljevic A, Bursac B, Milutinovic DV, Matic G (2013) Tissue-specific regulation of inflammation by macrophage migration inhibitory factor and glucocorticoids in fructose-fed Wistar rats. Br J Nutr 110:456–465
Castro MC, Francini F, Schinella G, Caldiz CI, Zubiria MG, Gagliardino JJ, Massa ML (2012) Apocynin administration prevents the changes induced by a fructose-rich diet on rat liver metabolism and the antioxidant system. Clin Sci (Lond) 123:681–692
Huang HY, Korivi M, Tsai CH, Yang JH, Tsai YC (2013) Supplementation of Lactobacillus plantarum K68 and Fruit-Vegetable Ferment along with High Fat-Fructose Diet Attenuates Metabolic Syndrome in Rats with Insulin Resistance. Evid Based Complement Altern Med 2013:943020
Madani Z, Louchami K, Sener A, Malaisse WJ, Ait Yahia D (2012) Dietary sardine protein lowers insulin resistance, leptin and TNF-alpha and beneficially affects adipose tissue oxidative stress in rats with fructose-induced metabolic syndrome. Int J Mol Med 29:311–318
Sivaraman K, Senthilkumar GP, Sankar P, Bobby Z (2013) Attenuation of oxidative stress, inflammation and insulin resistance by Allium sativum in fructose-fed male rats. J Clin Diagn Res 7:1860–1862
Hsieh F-C, Lee C-L, Chai C-Y, Chen W-T, Lu Y-C, Wu C-S (2013) Oral administration of Lactobacillus reuteri GMNL-263 improves insulin resistance and ameliorates hepatic steatosis in high fructose-fed rats. Nutr Metab 10:35
Tack CJ, Stienstra R, Joosten LA, Netea MG (2012) Inflammation links excess fat to insulin resistance: the role of the interleukin-1 family. Immunol Rev 249:239–252
Hotamisligil GS, Arner P, Caro JF, Atkinson RL, Spiegelman BM (1995) Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J Clin Invest 95:2409–2415
Hotamisligil GS, Shargill NS, Spiegelman BM (1993) Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science 259:87–91
Sethi JK, Hotamisligil GS (1999) The role of TNF alpha in adipocyte metabolism. Semin Cell Dev Biol 10:19–29
Xing H, Northrop JP, Grove JR, Kilpatrick KE, Su JL, Ringold GM (1997) TNF alpha-mediated inhibition and reversal of adipocyte differentiation is accompanied by suppressed expression of PPARgamma without effects on Pref-1 expression. Endocrinology 138:2776–2783
Williams PM, Chang DJ, Danesch U, Ringold GM, Heller RA (1992) CCAAT/enhancer binding protein expression is rapidly extinguished in TA1 adipocyte cells treated with tumor necrosis factor. Mol Endocrinol 6:1135–1141
De Bosscher K, Vanden Berghe W, Vermeulen L, Plaisance S, Boone E, Haegeman G (2000) Glucocorticoids repress NF-kappaB-driven genes by disturbing the interaction of p65 with the basal transcription machinery, irrespective of coactivator levels in the cell. Proc Natl Acad Sci USA 97:3919–3924
Tomlinson JW, Moore J, Cooper MS, Bujalska I, Shahmanesh M, Burt C, Strain A et al (2001) Regulation of expression of 11beta-hydroxysteroid dehydrogenase type 1 in adipose tissue: tissue-specific induction by cytokines. Endocrinology 142:1982–1989
Yuan M, Konstantopoulos N, Lee J, Hansen L, Li ZW, Karin M, Shoelson SE (2001) Reversal of obesity- and diet-induced insulin resistance with salicylates or targeted disruption of Ikkbeta. Science 293:1673–1677
Korićanac G, Djordjevic A, Žakula Z, Vojnović-Milutinović D, Tepavčević S, Veličković N, Milosavljević T et al (2013) Gender modulates development of the metabolic syndrome phenotype in fructose-fed rats. Arch Biol Sci 65:455–464
Teff KL, Elliott SS, Tschop M, Kieffer TJ, Rader D, Heiman M, Townsend RR et al (2004) Dietary fructose reduces circulating insulin and leptin, attenuates postprandial suppression of ghrelin, and increases triglycerides in women. J Clin Endocrinol Metab 89:2963–2972
Rebolledo OR, Marra CA, Raschia A, Rodriguez S, Gagliardino JJ (2008) Abdominal adipose tissue: early metabolic dysfunction associated to insulin resistance and oxidative stress induced by an unbalanced diet. Horm Metab Res 40:794–800
Schulze MB, Manson JE, Ludwig DS, Colditz GA, Stampfer MJ, Willett WC, Hu FB (2004) Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA 292:927–934
Tran LT, Yuen VG, McNeill JH (2009) The fructose-fed rat: a review on the mechanisms of fructose-induced insulin resistance and hypertension. Mol Cell Biochem 332:145–159
Ozanne SE, Nave BT, Wang CL, Shepherd PR, Prins J, Smith GD (1997) Poor fetal nutrition causes long-term changes in expression of insulin signaling components in adipocytes. Am J Physiol 273:E46–E51
Swinburn BA, Nyomba BL, Saad MF, Zurlo F, Raz I, Knowler WC, Lillioja S et al (1991) Insulin resistance associated with lower rates of weight gain in Pima Indians. J Clin Invest 88:168–173
Shi H, Kokoeva MV, Inouye K, Tzameli I, Yin H, Flier JS (2006) TLR4 links innate immunity and fatty acid-induced insulin resistance. J Clin Invest 116:3015–3025
Mellouk Z, Hupkens E, Antoine MH, Sener A, Ait Yahia D, Malaisse WJ (2012) The metabolic syndrome of fructose-fed rats: effects of long-chain polyunsaturated omega3 and omega6 fatty acids. VI. Further post-mortem investigations. Mol Med Rep 6:1404–1408
Fields M, Lewis CG (1995) Antioxidant defense mechanisms in the female rat: interactions with alcohol, copper, and type of dietary carbohydrate. Alcohol 12:227–231
Cavarape A, Feletto F, Mercuri F, Quagliaro L, Daman G, Ceriello A (2001) High-fructose diet decreases catalase mRNA levels in rat tissues. J Endocrinol Invest 24:838–845
Faure P, Rossini E, Lafond JL, Richard MJ, Favier A, Halimi S (1997) Vitamin E improves the free radical defense system potential and insulin sensitivity of rats fed high fructose diets. J Nutr 127:103–107
Simons SS Jr, Pratt WB (1995) Glucocorticoid receptor thiols and steroid-binding activity. Methods Enzymol 251:406–422
Okamoto K, Tanaka H, Ogawa H, Makino Y, Eguchi H, Hayashi S, Yoshikawa N et al (1999) Redox-dependent regulation of nuclear import of the glucocorticoid receptor. J Biol Chem 274:10363–10371
Acknowledgments
Funding sources: Ministry of Education, Science and Technological Development of the Republic of Serbia, Grant III41009.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Kovačević, S., Nestorov, J., Matić, G. et al. Fructose-enriched diet induces inflammation and reduces antioxidative defense in visceral adipose tissue of young female rats. Eur J Nutr 56, 151–160 (2017). https://doi.org/10.1007/s00394-015-1065-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-015-1065-0