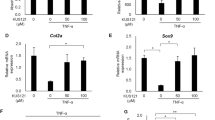
Abstract
Traditional Chinese medicine for invigorating the kidney and promoting blood circulation is commonly prescribed for the treatment of osteoarthritis associated with kidney deficiency and blood stasis. However, the specific mechanisms of these medicines are still unclear. The present study aimed to evaluate the protective effects of Bugu granules against sodium nitroprusside-induced chondrocyte apoptosis and elucidate the underlying molecular mechanisms. Drug-containing serum was prepared by administering rats with Bugu granules and harvesting the serum. Chondrocytes were exposed to different dilutions of serum, and apoptosis assessed by flow cytometry after staining with annexin V‑FITC/PI. Flow cytometry showed that chondrocyte apoptosis increased significantly after incubation with 2 mol/L sodium nitroprusside for 24 h (t = −48.221, P = 0.000), and the apoptotic rate of chondrocytes decreased with increasing concentrations of drug-containing serum (F = 33.965, P = 0.000). Cellular levels of Trx2, ASK1, caspase‑3, and reactive oxygen species (ROS) were detected by enzyme-linked immunosorbent assay. The cellular content of Trx2 increased gradually with increasing concentrations of drug-containing serum (F = 2610.593, P = 0.000), while that of ASK1 (F = 2473.545, P = 0.000), caspase‑3 (F = 209.921, P = 0.000), and ROS (F = 1666.435, P = 0.000) all decreased significantly. The mRNA expression levels were analyzed by RT-qPCR, which revealed that expression levels of Trx2 and caspase‑3 mRNA increased and decreased significantly, respectively, following exposure to Bugu granules in the drug-containing serum (F = 6.974, P = 0.003 and F = 3.691, P = 0.191; respectively), but the expression of ASK1 mRNA was not significantly different between treatment groups (F = 1.784, P = 0.191). Taken together, these results support the hypothesis that the Trx2 signaling pathway is activated by Bugu granules, which in turn inhibits chondrocyte apoptosis. This may play a role in preventing the development of osteoarthritis.
Zusammenfassung
Traditionelle chinesische Arzneimittel für die Stärkung der Niere und Förderung des Blutkreislaufs werden häufig zur Behandlung einer Arthrose assoziiert mit Nierenstörung und Blutstase verschrieben. Die genauen Wirkmechanismen dieser Arzneimittel sind aber noch nicht bekannt. In der vorliegenden Studie sollten die protektiven Wirkungen von Bugu-Granulat bezüglich der Natriumnitroprussid-induzierten Chondrozytenapoptose untersucht und die zugrunde liegenden molekularen Mechanismen aufgeklärt werden. Arzneimittelhaltiges Serum wurde hergestellt, indem Bugu-Granulat in Ratten appliziert und danach das Serum gewonnen wurde. Chondrozyten wurden mit verschiedenen Verdünnungen des Serums versetzt; die Apoptose wurde durchflusszytometrisch nach Färbung mit Annexin V-FITC/PI bestimmt. Die Durchflusszytometrie ergab, dass die Chondrozytenapoptose nach Inkubation mit 2 mol/l Natriumnitroprussid für 24 h signifikant anstieg (t = −48,221; P = 0,000). Die Apoptoserate der Chondrozyten sank mit steigenden Konzentrationen des arzneimittelhaltigen Serums (F = 33,965; P = 0,000). Die zellulären Spiegel von Trx2, ASK1, Caspase‑3 und reaktiven Sauerstoffspezies (ROS) wurden mit einem „enzyme-linked immunosorbent assay“ bestimmt. Der zelluläre Trx2-Gehalt stieg schrittweise mit zunehmender Konzentration des arzneimittelhaltigen Serums (F = 2610,593, P = 0,000), während der von ASK1 (F = 2473,545, P = 0,000), Caspase‑3 (F = 209,921, P = 0,000) und ROS (F = 1666,435, P = 0,000) jeweils signifikant abnahm. Die mRNA-Expressions-Niveaus wurden mithilfe der RT-qPCR analysiert. Diese zeigte, dass die Expressionsniveaus von Trx2- und Caspase-3-mRNA nach Exposition gegenüber Bugu-Granulat im arzneimittelhaltigen Serum signifikant zu- bzw. abnahmen (F = 6,974, P = 0,003 bzw. F = 3,691, P = 0,191). Die Expression von ASK1-mRNA unterschied sich dagegen nicht signifikant zwischen den Behandlungsgruppen (F = 1,784, P = 0,191). In der Zusammenschau stützen diese Ergebnisse die Hypothese, dass der Trx2-Signalweg durch Bugu-Granulat aktiviert wird, was wiederum die Chondrozytenapoptose hemmt. Das könnte mit Blick auf die Prävention der Arthroseentwicklung von Bedeutung sein.







Similar content being viewed by others
References
Yiqun B, Wang H, Shui S (2018) Taurine alleviates endoplasmic reticulum stress in the chondrocytes from patients with osteoarthritis. Redox Rep 23(1):118–124
Hwang Hyun S, Kim, Ah H (2015) Chondrocyte Apoptosis in the pathogenesis of osteoarthritis. Int J Mol Sci 16(11):26035–26054
Siming L, Xiaohong Y, Zhencheng F et al (2018) Catalase enhances viability of human Chondrocytes in culture by reducing reactive oxygen species and counteracting tumor necrosis factor-α-induced Apoptosis. Cell Physiol Biochem 49(6):2427–2442
Cai X‑Y, Xia Y, Yang S‑H et al (2015) Ropivacaine- and bupivacaine-induced death of rabbit annulus fibrosus cells in vitro: involvement of the mitochondrial apoptotic pathway. Osteoarthritis Cartilage 23(10):1763–1775
Sun N, Hao JR, Li XY et al (2013) GluR6-FasL-Trx2 mediates denitrosylation and activation of procaspase‑3 in cerebral ischemia/reperfusion in rats. Cell Death Dis 4:e771
Gogvadze V, Orrenius S, Zhivotovsky B (2015) Analysis of mitochondrial dysfunction during cell death. Methods Mol Biol 1264(1):385–393
Huang Q, Zhou Huanjiao J, Zhang H et al (2015) Thioredoxin‑2 inhibits mitochondrial reactive oxygen species generation and apoptosis stress kinase‑1 activity to maintain cardiac function. Circulation 131(12):1082–1097
Yan-Yan L, Yin X, Zhang S et al (2017) Thioredoxin‑2 protects against oxygen-glucose deprivation/reperfusion injury by inhibiting autophagy and apoptosis in H9c2 cardiomyocytes. Am J Transl Res 9(3):1471–1482
Hu C, Zhang H, Qiao Z et al (2018) Loss of thioredoxin 2 alters mitochondrial respiratory function and induces cardiomyocyte hypertrophy. Exp Cell Res 372(1):61–72
Hai-Ying S, Hu Y‑J, Zhao X‑Y et al (2015) Age-related changes in mitochondrial antioxidant enzyme Trx2 and TXNIP-Trx2-ASK1 signal pathways in the auditory cortex of a mimetic aging rat model: changes to Trx2 in the auditory cortex. Febs J 282(14):2758–2774
Cheleschi S, De Palma Pascarelli ANA et al (2017) Could oxidative stress regulate the expression of microRNA-146a and microRNA-34a in human osteoarthritic Chondrocyte cultures. Int J Mol Sci 18(12):1–15
Zhang K, Jiang D (2017) RhoA inhibits the hypoxia-induced apoptosis and mitochondrial dysfunction in chondrocytes via positively regulating the CREB phosphorylation. Biosci Rep 37(2):1–11
Qixin C, Xifan M, Guanying H et al (2015) Xanthan gum protects rabbit articular chondrocytes against sodium nitroprusside-induced apoptosis in vitro. Carbohydr Polym 131(1):363–369
Alvarez-Garcia O, Matsuzaki T, Olmer M et al (2017) Regulated in development and DNA damage response 1 deficiency impairs autophagy and Mitochondrial Biogenesis in articular cartilage and increases the severity of experimental osteoarthritis. Arthritis Rheumatol 69(7):1418–1428
Tang Q, Zheng G, Feng Z et al (2017) Trehalose ameliorates oxidative stress-mediated mitochondrial dysfunction and ER stress via selective autophagy stimulation and autophagic flux restoration in osteoarthritis development. Cell Death Dis 8(10):e3081
Shelar Sandeep B, Kaminska Kamila K, Reddy Shridhivya A et al (2015) Thioredoxin-dependent regulation of AIF-mediated DNA damage. Free Radic Biol Med 87(1):125–136
Jidong Y, Xu J, Fei Y et al (2016) TrxR2 deficiencies promote chondrogenic differentiation and induce apoptosis of chondrocytes through mitochondrial reactive oxygen species. Exp Cell Res 344(1):67–75
Fengbo L, Xiaolei S, Jianxiong M et al (2014) Naringin prevents ovariectomy-induced osteoporosis and promotes osteoclasts apoptosis through the mitochondria-mediated apoptosis pathway. Biochem Biophys Res Commun 452(3):629–635
Pan L, Zhang Y, Chen N et al (2017) Icariin regulates cellular functions and gene expression of osteoarthritis patient-derived human Fibroblast-like Synoviocytes. Int J Mol Sci 18(12):1–11
Zheng YS, Wu ZS, Ni HB et al (2014) Codonopsis pilosula polysaccharide attenuates cecal ligation and puncture sepsis via circuiting regulatory T cells in mice. Shock 41(3):250–255
Zhou L, Zhou C, Feng Z et al (2018) Triptolide-induced hepatotoxicity can be alleviated when combined with Panax notoginseng saponins and Catapol. J Ethnopharmacol 214:232–239
Zhuang C, Xu N‑W, Gao G‑M et al (2016) Polysaccharide from Angelica sinensis protects chondrocytes from H2O2-induced apoptosis through its antioxidant effects in vitro. Int J Biol Macromol 87:322–328
Yanyan L, Xiang Yin, Zhang S et al (2017) Intramyocardial injection of thioredoxin 2‑expressing lentivirus alleviates myocardial ischemia-reperfusion injury in rats. Am J Transl Res 9(10):4428–4439
Funding
This work was supported by the Fuzhou Science and Technology Plan Project of China (grant no. 2017-S-130-5) and Fujian Provincial Natural Science Foundation of China (grant no. 2016J01597).
Author information
Authors and Affiliations
Contributions
Guang-Shu Yu participated in the design of the study and performed the statistical analysis. Yan-Bin Lin conceived of the study, participated in its design and coordination and helped to draft the manuscript. Hong-Bin Xu participated in the literature search and data integration. Shou-Xiong Zhang, Jie-Hui Li and Hai-Yang Wang participated in the experiment and statistical analysis of data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Guang-Shu Yu, Yan-Bin Lin, Hong-Bin Xu, Shou-Xiong Zhang, Jie-Hui Li and Hai-Yang Wang declare that they have no competing interests.
For this article no studies with human participants were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case. The present study was approved by the Animal Care and Use Committee of the Fuzhou Second Hospital Affiliated to Xiamen University.
Additional information
Redaktion
U. Müller-Ladner, Bad Nauheim
U. Lange, Bad Nauheim
Availability of data and materials
The datasets used during the present study are available from the corresponding author on reasonable request.
Rights and permissions
About this article
Cite this article
Yu, GS., Lin, YB., Xu, HB. et al. Effect of Bugu granules in a drug-containing serum on chondrocyte apoptosis and the Trx2 signaling pathway. Z Rheumatol 79, 304–311 (2020). https://doi.org/10.1007/s00393-019-00688-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-019-00688-z