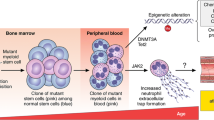
Abstract
Aims
To investigate the frequency of clonal hematopoiesis of indeterminate potential (CHIP) and evaluate its impacts on outcomes in patients with acute coronary syndrome (ACS) undergoing percutaneous coronary intervention (PCI) in the absence of traditional cardiovascular risk factors (CVRFs).
Methods
Whole-exome sequencing was performed to detect the presence of CHIP in 183 patients underwent PCI for the treatment of ACS. The association between CHIP-related mutations and major adverse cardiac or cerebral events (MACCEs, a composite of all-cause mortality, coronary revascularization, myocardial infarction, or stroke) was analyzed in such cohort.
Results
Of 179 patients [median age, 65 years; 84 female (46.9%)] included in this analysis, CHIP-related mutations were detected in 36 (20.1%) patients. The somatic mutations most frequently occurred in the genes DNMT3A (17 mutations), TET2 (6 mutations), and ASXL1 (4 mutations). Clinical outcomes at median 635 follow-up days showed that DNMT3A/TET2/ASXL1-CHIP mutations were associated with significantly higher risk of MACCEs, compared with non-CHIP carriers in the CVRFs-absent ACS cohort (26.1% vs. 4.2%, log-rank P = 0.001). Multivariable regression showed that DNMT3A/TET2/ASXL1-CHIP driver mutations (HR 4.015; 95% CI 1.236–13.046; P = 0.021) were independent predictors of adverse clinical outcomes.
Conclusion
The most frequent CHIP-related mutations, DNMT3A, TET2, and ASXL1 are significantly associated with increased risk of recurrent cardiovascular events. Our study may be valuable target to reduce residual risk in patients with ACS carrying specific mutations.
Graphical abstract





Similar content being viewed by others
References
Steensma DP, Bejar R, Jaiswal S, Lindsley RC, Sekeres MA, Hasserjian RP, Ebert BL (2015) Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood 126:9–16. https://doi.org/10.1182/blood-2015-03-631747
Dorsheimer L, Assmus B, Rasper T, Ortmann CA, Ecke A, Abou-El-Ardat K, Schmid T, Brüne B, Wagner S, Serve H, Hoffmann J, Seeger F, Dimmeler S, Zeiher AM, Rieger MA (2019) Association of mutations contributing to clonal hematopoiesis with prognosis in chronic ischemic heart failure. JAMA Cardiol 4:25–33. https://doi.org/10.1001/jamacardio.2018.3965
Pascual-Figal DA, Bayes-Genis A, Díez-Díez M, Hernández-Vicente Á, Vázquez-Andrés D, de la Barrera J, Vazquez E, Quintas A, Zuriaga MA, Asensio-López MC, Dopazo A, Sánchez-Cabo F, Fuster JJ (2021) Clonal hematopoiesis and risk of progression of heart failure with reduced left ventricular ejection fraction. J Am Coll Cardiol 77:1747–1759. https://doi.org/10.1016/j.jacc.2021.02.028
Jaiswal S, Natarajan P, Silver AJ, Gibson CJ, Bick AG, Shvartz E, McConkey M, Gupta N, Gabriel S, Ardissino D, Baber U, Mehran R, Fuster V, Danesh J, Frossard P, Saleheen D, Melander O, Sukhova GK, Neuberg D, Libby P, Kathiresan S, Ebert BL (2017) Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N Engl J Med 377:111–121. https://doi.org/10.1056/NEJMoa1701719
Fuster JJ, MacLauchlan S, Zuriaga MA, Polackal MN, Ostriker AC, Chakraborty R, Wu CL, Sano S, Muralidharan S, Rius C, Vuong J, Jacob S, Muralidhar V, Robertson AA, Cooper MA, Andrés V, Hirschi KK, Martin KA, Walsh K (2017) Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 355:842–847. https://doi.org/10.1126/science.aag1381
Sano S, Oshima K, Wang Y, Katanasaka Y, Sano M, Walsh K (2018) CRISPR-mediated gene editing to assess the roles of Tet2 and Dnmt3a in clonal hematopoiesis and cardiovascular disease. Circ Res 123:335–341. https://doi.org/10.1161/CIRCRESAHA.118.313225
Abplanalp WT, Mas-Peiro S, Cremer S, John D, Dimmeler S, Zeiher AM (2020) Association of clonal hematopoiesis of indeterminate potential with inflammatory gene expression in patients with severe degenerative aortic valve stenosis or chronic postischemic heart failure. JAMA Cardiol 5:1170–1175. https://doi.org/10.1001/jamacardio.2020.2468
Jaiswal S, Fontanillas P, Flannick J, Manning A, Grauman PV, Mar BG, Lindsley RC, Mermel CH, Burtt N, Chavez A, Higgins JM, Moltchanov V, Kuo FC, Kluk MJ, Henderson B, Kinnunen L, Koistinen HA, Ladenvall C, Getz G, Correa A, Banahan BF, Gabriel S, Kathiresan S, Stringham HM, McCarthy MI, Boehnke M, Tuomilehto J, Haiman C, Groop L, Atzmon G, Wilson JG, Neuberg D, Altshuler D, Ebert BL (2014) Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med 371:2488–2498. https://doi.org/10.1056/NEJMoa1408617
Bick AG, Weinstock JS, Nandakumar SK et al (2020) Inherited causes of clonal haematopoiesis in 97,691 whole genomes. Nature 586:763–768. https://doi.org/10.1038/s41586-020-2819-2
Khot UN, Khot MB, Bajzer CT, Sapp SK, Ohman EM, Brener SJ, Ellis SG, Lincoff AM, Topol EJ (2003) Prevalence of conventional risk factors in patients with coronary heart disease. JAMA 290:898–904. https://doi.org/10.1001/jama.290.7.898
Figtree GA, Vernon ST, Hadziosmanovic N, Sundström J, Alfredsson J, Arnott C, Delatour V, Leósdóttir M, Hagström E (2021) Mortality in STEMI patients without standard modifiable risk factors: a sex-disaggregated analysis of SWEDEHEART registry data. Lancet 397:1085–1094. https://doi.org/10.1016/S0140-6736(21)00272-5
Vernon ST, Coffey S, D’Souza M, Chow CK, Kilian J, Hyun K, Shaw JA, Adams M, Roberts-Thomson P, Brieger D, Figtree GA (2019) ST-segment-elevation myocardial infarction (STEMI) patients without standard modifiable cardiovascular risk factors-how common are they, and what are their outcomes? J Am Heart Assoc 8:e013296. https://doi.org/10.1161/JAHA.119.013296
Libby P (2013) Mechanisms of acute coronary syndromes and their implications for therapy. N Engl J Med 368:2004–2013. https://doi.org/10.1056/NEJMra1216063
Kalkman DN, Aquino M, Claessen BE, Baber U, Guedeney P, Sorrentino S, Vogel B, de Winter RJ, Sweeny J, Kovacic JC, Shah S, Vijay P, Barman N, Kini A, Sharma S, Dangas GD, Mehran R (2018) Residual inflammatory risk and the impact on clinical outcomes in patients after percutaneous coronary interventions. Eur Heart J 39:4101–4108. https://doi.org/10.1093/eurheartj/ehy633
Han Y, Li Y, Li Y (2019) The Department of Cardiology of General Hospital of Northern Theater Command of Chinese People’s Liberation Army, Shenyang, China. Eur Heart J 40:3816–3818. https://doi.org/10.1093/eurheartj/ehz840
Valent P, Horny HP, Bennett JM, Fonatsch C, Germing U, Greenberg P, Haferlach T, Haase D, Kolb HJ, Krieger O, Loken M, van de Loosdrecht A, Ogata K, Orfao A, Pfeilstöcker M, Rüter B, Sperr WR, Stauder R, Wells DA (2007) Definitions and standards in the diagnosis and treatment of the myelodysplastic syndromes: consensus statements and report from a working conference. Leuk Res 31:727–736. https://doi.org/10.1016/j.leukres.2006.11.009
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25:1754–1760. https://doi.org/10.1093/bioinformatics/btp324
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R, 1000 Genome Project Data Processing Subgroup (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Tarasov A, Vilella AJ, Cuppen E, Nijman IJ, Prins P (2015) Sambamba: fast processing of NGS alignment formats. Bioinformatics 31:2032–2034. https://doi.org/10.1093/bioinformatics/btv098
Wang K, Li M, Hakonarson H (2010) ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38:e164. https://doi.org/10.1093/nar/gkq603
Libby P, Sidlow R, Lin AE, Gupta D, Jones LW, Moslehi J, Zeiher A, Jaiswal S, Schulz C, Blankstein R, Bolton KL, Steensma D, Levine RL, Ebert BL (2019) Clonal hematopoiesis: crossroads of aging, cardiovascular disease, and cancer: JACC review topic of the week. J Am Coll Cardiol 74:567–577. https://doi.org/10.1016/j.jacc.2019.06.007
Silvain J, Kerneis M, Zeitouni M, Lattuca B, Galier S, Brugier D, Mertens E, Procopi N, Suc G, Salloum T, Frisdal E, Le Goff W, Collet JP, Vicaut E, Lesnik P, Montalescot G, Guerin M (2020) Interleukin-1β and risk of premature death in patients with myocardial infarction. J Am Coll Cardiol 76:1763–1773. https://doi.org/10.1016/j.jacc.2020.08.026
Nidorf SM, Fiolet ATL, Mosterd A, Eikelboom JW, Schut A, Opstal TSJ, The SHK, Xu XF, Ireland MA, Lenderink T, Latchem D, Hoogslag P, Jerzewski A, Nierop P, Whelan A, Hendriks R, Swart H, Schaap J, Kuijper AFM, van Hessen MWJ, Saklani P, Tan I, Thompson AG, Morton A, Judkins C, Bax WA, Dirksen M, Alings M, Hankey GJ, Budgeon CA, Tijssen JGP, Cornel JH, Thompson PL (2020) Colchicine in patients with chronic coronary disease. N Engl J Med 383:1838–1847. https://doi.org/10.1056/NEJMoa2021372
Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ (2017) Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 377:1119–1131. https://doi.org/10.1056/NEJMoa1707914
Svensson EC, Madar A, Campbell CD, He Y, Sultan M, Healey ML, Xu H, D’Aco K, Fernandez A, Wache-Mainier C, Libby P, Ridker PM, Beste MT, Basson CT (2022) TET2-driven clonal hematopoiesis and response to canakinumab: an exploratory analysis of the CANTOS randomized clinical Trial. JAMA Cardiol. https://doi.org/10.1001/jamacardio.2022.0386
Bick AG, Pirruccello JP, Griffin GK, Gupta N, Gabriel S, Saleheen D, Libby P, Kathiresan S, Natarajan P (2020) Genetic interleukin 6 signaling deficiency attenuates cardiovascular risk in clonal hematopoiesis. Circulation 141:124–131. https://doi.org/10.1161/CIRCULATIONAHA.119.044362
Ketkar S, Verdoni AM, Smith AM, Bangert CV, Leight ER, Chen DY, Brune MK, Helton NM, Hoock M, George DR, Fronick C, Fulton RS, Ramakrishnan SM, Chang GS, Petti AA, Spencer DH, Miller CA, Ley TJ (2020) Remethylation of Dnmt3a-/- hematopoietic cells is associated with partial correction of gene dysregulation and reduced myeloid skewing. Proc Natl Acad Sci U S A 117:3123–3134. https://doi.org/10.1073/pnas.1918611117
Acknowledgements
We thank Prof. Siddhartha Jaiswal (Department of Pathology, Stanford University School of Medicine, USA) for constructive suggestions on CHIP-related gene list aspects.
Funding
This study was supported by the National Key Research and Development Project in China, Grant/Award Numbers: 2016YFC1301300, 2016YFC1301303.
Author information
Authors and Affiliations
Contributions
ZXJ and YL: designed the study, acquired and analyzed data, and wrote the manuscript; CHY, XLZ, QYZ, JL, XXT, MHQ and ZYL: assisted with study design; MHQ: assisted with data analysis; SCM, KN, ZQL, SBC, YZ, ZZQ and XYL: clinically examined study participants; and YLH: supervised the study, critically revised the manuscript, and obtained research funding.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
This study have been approved by the ethics review board of the General Hospital of Northern Theater Command and have, therefore, been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jiang, Z., Li, Y., Yan, C. et al. Clonal hematopoiesis of indeterminate potential in patients with acute coronary syndrome undergoing percutaneous coronary intervention in the absence of traditional risk factors. Clin Res Cardiol 112, 506–517 (2023). https://doi.org/10.1007/s00392-022-02039-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-022-02039-6