Abstract
Purpose
Patients undergoing laparoscopic colorectal cancer resection have a high incidence of postoperative gastrointestinal dysfunction (POGD). Remote ischemic preconditioning (RIPC) is an organ protection measure. The study investigated the effect of RIPC on postoperative gastrointestinal function.
Methods
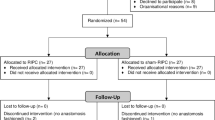
In this single-center, prospective, double-blinded, randomized, parallel-controlled trial, 100 patients undergoing elective laparoscopic colorectal cancer resection were randomly assigned in a 1:1 ratio to receive RIPC or sham RIPC (control). Three cycles of 5-min ischemia/5-min reperfusion induced by a blood pressure cuff placed on the right upper arm served as RIPC stimulus. Patients were followed up continuously for 7 days after surgery. The I-FEED score was used to evaluate the patient’s gastrointestinal function after the surgery. The primary outcome of the study was the I-FEED score on POD3. Secondary outcomes include the daily I-FEED scores, the highest I-FEED score, the incidence of POGD, the changes in I-FABP and the inflammatory markers (IL-6 and TNF-α), and the time to first postoperative flatus.
Results
A total of 100 patients were enrolled in the study, of which 13 patients were excluded. Finally, 87 patients were included in the analysis, 44 patients in the RIPC group and 43 patients in the sham-RIPC group. Patients assigned to the RIPC group had a lower I-FEED score on POD3 compared with the sham-RIPC group (mean difference 0.86; 95% CI: 0.06 to 1.65; P = 0.035). And patients in the RIPC group were also associated with a lower I-FEED score on POD4 vs the sham-RIPC group (mean difference 0.81; 95% CI: 0.03 to 1.60; P = 0.043). Compared with the sham-RIPC group, the incidence of POGD within 7 days after surgery was lower in the RIPC group (P = 0.040). At T1, T2, and T3 time points, inflammatory factors and I-FABP were considerably less in the RIPC group compared to the sham-RIPC group. The time to the first flatus and the first feces was similar in both groups.
Conclusion
RIPC reduced I-FEED scores, decreased the incidence of postoperative gastrointestinal dysfunction, and lowered concentrations of I-FABP and inflammatory factors.


Similar content being viewed by others
Data availability
The data sets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Vather R, Trivedi S, Bissett I (2013) Defining postoperative ileus: results of a systematic review and global survey. J Gastrointest Surg 17(5):962–972
Lam D, Jones O (2020) Changes to gastrointestinal function after surgery for colorectal cancer. Best Pract Res Clin Gastroenterol 48–49
Iyer S, Saunders WB, Stemkowski S (2009) Economic burden of postoperative ileus associated with colectomy in the United States. J Manag Care Pharm JMCP 15(6):485–494
Alhashemi M, Fiore JF Jr, Safa N, Al Mahroos M, Mata J, Pecorelli N, Baldini G, Dendukuri N, Stein BL, Liberman AS et al (2019) Incidence and predictors of prolonged postoperative ileus after colorectal surgery in the context of an enhanced recovery pathway. Surg Endosc 33(7):2313–2322
Gianotti L, Nespoli L, Rocchetti S, Vignali A, Nespoli A, Braga M (2011) Gut oxygenation and oxidative damage during and after laparoscopic and open left-sided colon resection: a prospective, randomized, controlled clinical trial. Surg Endosc 25(6):1835–1843
Ishizaki Y, Bandai Y, Shimomura K, Abe H, Ohtomo Y, Idezuki Y (1993) Changes in splanchnic blood flow and cardiovascular effects following peritoneal insufflation of carbon dioxide. Surg Endosc 7(5):420–423
Vlug MS, Diepenhorst GM, van Koperen PJ, Renooij W, de Smet MB, Slors JF, Boermeester MA, Bemelman WA (2011) Intestinal barrier function in patients undergoing colectomy. Colorectal Dis 13(12):1432–1437
Watt SK, Hasselbalch HC, Skov V, Kjaer L, Thomassen M, Kruse TA, Burton M, Poulsen HE, Gogenur I (2020) Increased oxidative stress with substantial dysregulation of genes related to oxidative stress and DNA repair after laparoscopic colon cancer surgery. Surg Oncol 35:71–78
Li C, Li YS, Xu M, Wen SH, Yao X, Wu Y, Huang CY, Huang WQ, Liu KX (2013) Limb remote ischemic preconditioning for intestinal and pulmonary protection during elective open infrarenal abdominal aortic aneurysm repair: a randomized controlled trial. Anesthesiology 118(4):842–852
Zhou H, Yang L, Wang G, Zhang C, Fang Z, Lei G, Shi S, Li J (2019) Remote ischemic preconditioning prevents postoperative acute kidney injury after open total aortic arch replacement: a double-blind, randomized, sham-controlled trial. Anesth Analg 129(1):287–293
Garcia-de-la-Asuncion J, Bruno L, Perez-Griera J, Galan G, Morcillo A, Wins R, Garcia-Del-Olmo E, Guijarro R, Sarria B, Marti F et al (2017) Remote ischemic preconditioning decreases oxidative lung damage after pulmonary lobectomy: a single-center randomized, double-blind, controlled trial. Anesth Analg 125(2):499–506
Lang JA, Kim J (2022) Remote ischaemic preconditioning - translating cardiovascular benefits to humans. J Physiol 600(13):3053–3067
Tapuria N, Kumar Y, Habib MM, Abu Amara M, Seifalian AM, Davidson BR (2008) Remote ischemic preconditioning: a novel protective method from ischemia reperfusion injury–a review. J Surg Res 150(2):304–330
Hummitzsch L, Zitta K, Berndt R, Wong YL, Rusch R, Hess K, Wedel T, Gruenewald M, Cremer J, Steinfath M et al (2019) Remote ischemic preconditioning attenuates intestinal mucosal damage: insight from a rat model of ischemia-reperfusion injury. J Transl Med 17(1):136
Zitta K, Meybohm P, Bein B, Heinrich C, Renner J, Cremer J, Steinfath M, Scholz J, Albrecht M (2012) Serum from patients undergoing remote ischemic preconditioning protects cultured human intestinal cells from hypoxia-induced damage: involvement of matrixmetalloproteinase-2 and -9. Mol Med 18:29–37
Filaretova L, Komkova O, Sudalina M, Yarushkina N (2021) Non-invasive remote ischemic preconditioning may protect the gastric mucosa against ischemia-reperfusion-induced injury through involvement of glucocorticoids. Front Pharmacol 12:682643
Colak T, Turkmenoglu O, Dag A, Polat A, Comelekoglu U, Bagdatoglu O, Polat G, Kanik A, Akca T, Aydin S (2007) The effect of remote ischemic preconditioning on healing of colonic anastomoses. J Surg Res 143(2):200–205
Hausenloy DJ, Candilio L, Evans R, Ariti C, Jenkins DP, Kolvekar S, Knight R, Kunst G, Laing C, Nicholas J et al (2015) Remote ischemic preconditioning and outcomes of cardiac surgery. N Engl J Med 373(15):1408–1417
Struck R, Wittmann M, Muller S, Meybohm P, Muller A, Bagci S (2018) Effect of remote ischemic preconditioning on intestinal ischemia-reperfusion injury in adults undergoing on-pump CABG surgery: a randomized controlled pilot trial. J Cardiothorac Vasc Anesth 32(3):1243–1247
Hedrick TL, McEvoy MD, Mythen MMG, Bergamaschi R, Gupta R, Holubar SD, Senagore AJ, Gan TJ, Shaw AD, Thacker JKM et al (2018) American society for enhanced recovery and perioperative quality initiative joint consensus statement on postoperative gastrointestinal dysfunction within an enhanced recovery pathway for elective colorectal surgery. Anesth Analg 126(6):1896–1907
Alsharqawi N, Alhashemi M, Kaneva P, Baldini G, Fiore JF Jr, Feldman LS, Lee L (2020) Validity of the I-FEED score for postoperative gastrointestinal function in patients undergoing colorectal surgery. Surg Endosc 34(5):2219–2226
Peng LH, Wang WJ, Chen J, Jin JY, Min S, Qin PP (2021) Implementation of the pre-operative rehabilitation recovery protocol and its effect on the quality of recovery after colorectal surgeries. Chin Med J (Engl) 134(23):2865–2873
Schietroma M, Pessia B, Carlei F, Cecilia EM, Amicucci G (2013) Intestinal permeability, systemic endotoxemia, and bacterial translocation after open or laparoscopic resection for colon cancer: a prospective randomized study. Int J Colorectal Dis 28(12):1651–1660
Reddy BS, Gatt M, Sowdi R, MacFie J (2006) Surgical manipulation of the large intestine increases bacterial translocation in patients undergoing elective colorectal surgery. Colorectal Dis 8(7):596–600
Braga M, Vignali A, Gianotti L, Zuliani W, Radaelli G, Gruarin P, Dellabona P, Di Carlo V (2002) Laparoscopic versus open colorectal surgery: a randomized trial on short-term outcome. Ann Surg 236(6):759–766
Leventi A, Argyra E, Avraamidou A, Marinis A, Asonitis S, Perrea D, Voros D, Theodoraki K (2015) Attenuation of oxidative stress by ischemic preconditioning in an experimental model of intraabdominal hypertension. J Invest Surg 28(5):253–260
Wu MY, Yiang GT, Liao WT, Tsai AP, Cheng YL, Cheng PW, Li CY, Li CJ (2018) Current mechanistic concepts in ischemia and reperfusion injury. Cell Physiol Biochem 46(4):1650–1667
Deitch EA (2012) Gut-origin sepsis: evolution of a concept. Surgeon 10(6):350–356
Lenaerts K, Ceulemans LJ, Hundscheid IH, Grootjans J, Dejong CH, Olde Damink SW (2013) New insights in intestinal ischemia-reperfusion injury: implications for intestinal transplantation. Curr Opin Organ Transplant 18(3):298–303
Li G, Zhang Y, Fan Z (2021) Cellular signal transduction pathways involved in acute lung injury induced by intestinal ischemia-reperfusion. Oxid Med Cell Longev 2021:9985701
Koike Y, Li B, Ganji N, Zhu H, Miyake H, Chen Y, Lee C, Janssen Lok M, Zozaya C, Lau E et al (2020) Remote ischemic conditioning counteracts the intestinal damage of necrotizing enterocolitis by improving intestinal microcirculation. Nat Commun 11(1):4950
Evennett NJ, Petrov MS, Mittal A, Windsor JA (2009) Systematic review and pooled estimates for the diagnostic accuracy of serological markers for intestinal ischemia. World J Surg 33(7):1374–1383
Khadaroo RG, Fortis S, Salim SY, Streutker C, Churchill TA, Zhang H (2014) I-FABP as biomarker for the early diagnosis of acute mesenteric ischemia and resultant lung injury. PLoS ONE 9(12):e115242
Memet O, Zhang L, Shen J (2019) Serological biomarkers for acute mesenteric ischemia. Ann Transl Med 7(16):394
Kuhn KA, Manieri NA, Liu TC, Stappenbeck TS (2014) IL-6 stimulates intestinal epithelial proliferation and repair after injury. PLoS ONE 9(12):e114195
Jeffery V, Goldson AJ, Dainty JR, Chieppa M, Sobolewski A (2017) IL-6 signaling regulates small intestinal crypt homeostasis. J Immunol 199(1):304–311
Li A, Xiong J, Chen Z (2012) IL-6, TNF-α, and iNOS is associated with decreased colonic contraction in rats with multiple organ dysfunction syndrome. J Surg Res 178(2):e51–e57
Wehner S, Schwarz NT, Hundsdoerfer R, Hierholzer C, Tweardy DJ, Billiar TR, Bauer AJ, Kalff JC (2005) Induction of IL-6 within the rodent intestinal muscularis after intestinal surgical stress. Surgery 137(4):436–446
Zhu P, Jiang H, Fu J, Chen W, Wang Z, Cui L (2013) Cytokine levels in abdominal exudate predict prolonged postoperative ileus following surgery for colorectal carcinoma. Oncol Lett 6(3):835–839
Hampson A, Raj N, Lingamanaicker V et al (2021) Serum cytokine levels as markers of paralytic ileus following robotic radical prostatectomy at different pneumoperitoneum pressures. Curr Urol 15(2):91–94
Author information
Authors and Affiliations
Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Approval for this study was obtained from the ethics committee of the Affiliated Lianyungang Hospital of Xuzhou Medical University, and each patient provided written informed consent.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yi, M., Wu, Y., Li, M. et al. Effect of remote ischemic preconditioning on postoperative gastrointestinal function in patients undergoing laparoscopic colorectal cancer resection. Int J Colorectal Dis 38, 68 (2023). https://doi.org/10.1007/s00384-023-04346-4
Accepted:
Published:
DOI: https://doi.org/10.1007/s00384-023-04346-4