Abstract
Purpose
The CpG island methylator phenotype (CIMP), important for carcinogenesis, is a predictor of prognosis and chemotherapy sensitivity in colorectal cancer (CRC). However, there is a lack of consensus on CIMP markers, and thus, more comprehensive methylation markers are required to reliably predict the clinical outcomes. This study aimed to clarify the effects of genome-wide DNA methylation status on clinical outcomes in patients with metastatic CRC (mCRC) treated with epidermal growth factor receptor (EGFR) inhibitors.
Methods
We enrolled 241 patients with mCRC, who received chemotherapy plus EGFR inhibitors as a first-line treatment. We analyzed the incidence and clinicopathological characteristics of highly methylated CRC (HMCC) and associations between genome-wide DNA methylation status and response rate (RR), progression-free survival (PFS), and overall survival (OS).
Results
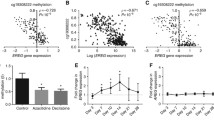
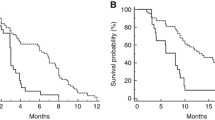
In total, 169 patients were included in the final analyses. The frequency of HMCC was 8.9% (15/169). The characteristics of patients with HMCC included right-sided primary tumor location (P = 0.042), undifferentiated histology (P = 0.047), and BRAF V600E mutation (P < 0.0001). Patients with HMCC showed worse clinical outcomes than those with low-methylated CRC in terms of RR (P = 0.017), PFS (P = 0.004), and OS (P = 0.019). In the multivariate analysis, peritoneal metastasis (P = 0.017), methylation status (P = 0.037), and BRAF V600E mutations (P = 0.0001) were independent factors for shorter PFS.
Conclusions
Genome-wide DNA methylation status is an independent factor associated with PFS in patients with mCRC treated with first-line EGFR inhibitors.



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Sharma S, Kelly TK, Jones PA (2010) Epigenetics in cancer. Carcinogenesis 31:27–36. https://doi.org/10.1093/carcin/bgp220
Toyota M, Ahuja N, Ohe-Toyota M, Herman JG, Baylin SB, Issa JP (1999) CpG island methylator phenotype in colorectal cancer. Proc Natl Acad Sci U S A 96:8681–8686. https://doi.org/10.1073/pnas.96.15.8681
Bird A (2002) DNA methylation patterns and epigenetic memory. Genes Dev 16:6–21. https://doi.org/10.1101/gad.947102
Turker MS (1999) The establishment and maintenance of DNA methylation patterns in mouse somatic cells. Semin Cancer Biol 9:329–337. https://doi.org/10.1006/scbi.1999.0133
Shen L, Toyota M, Kondo Y, Lin E, Zhang L, Guo Y, Hernandez NS, Chen X, Ahmed S, Konishi K, Hamilton SR, Issa JPJ (2007) Integrated genetic and epigenetic analysis identifies three different subclasses of colon cancer. Proc Natl Acad Sci U S A 104:18654–18659. https://doi.org/10.1073/pnas.0704652104
Weisenberger DJ, Siegmund KD, Campan M, Young J, Long TI, Faasse MA, Kang GH, Widschwendter M, Weener D, Buchanan D, Koh H, Simms L, Barker M, Leggett B, Levine J, Kim M, French AJ, Thibodeau SN, Jass J, Haile R, Laird PW (2006) CpG island methylator phenotype underlies sporadic microsatellite instability and is tightly associated with BRAF mutation in colorectal cancer. Nat Genet 38:787–793. https://doi.org/10.1038/ng1834
Yamauchi M, Lochhead P, Morikawa T, Huttenhower C, Chan AT, Giovannucci E, Fuchs C, Ogino S (2012) Colorectal cancer: a tale of two sides or a continuum? Gut 61:794–797. https://doi.org/10.1136/gutjnl-2012-302014
Ogino S, Nosho K, Kirkner GJ, Kawasaki T, Meyerhardt JA, Loda M, Giovannucci EL, Fuchs CS (2009) CpG island methylator phenotype, microsatellite instability, BRAF mutation and clinical outcome in colon cancer. Gut 58:90–96. https://doi.org/10.1136/gut.2008.155473
Ward RL, Cheong K, Ku SL, Meagher A, O’Connor T, Hawkins NJ (2003) Adverse prognostic effect of methylation in colorectal cancer is reversed by microsatellite instability. J Clin Oncol 21:3729–3736. https://doi.org/10.1200/JCO.2003.03.123
van Rijnsoever M, Elsaleh H, Joseph D, McCaul K, Iacopetta B (2003) CpG island methylator phenotype is an independent predictor of survival benefit from 5-fluorouracil in stage III colorectal cancer. Clin Cancer Res 9:2898–2903
Shen L, Catalano PJ, Benson AB 3rd, O’Dwyer P, Hamilton SR, Issa JP (2007) Association between DNA methylation and shortened survival in patients with advanced colorectal cancer treated with 5-fluorouracil based chemotherapy. Clin Cancer Res 13:6093–6098. https://doi.org/10.1158/1078-0432.CCR-07-1011
Jover R, Nguyen TP, Pérez-Carbonell L, Zapater P, Payá A, Alenda C, Rojas E, Cubiella J, Balaguer F, Morillas JD, Clofent J, Bujanda L, Reñé JM, Bessa X, Xicola RM, Nicolás-Pérez D, Castells A, Andreu M, Llor X, Boland CR, Goel A (2011) 5-Fluorouracil adjuvant chemotherapy does not increase survival in patients with CpG island methylator phenotype colorectal cancer. Gastroenterology 140:1174–1181. https://doi.org/10.1053/j.gastro.2010.12.035
Ouchi K, Takahashi S, Yamada Y, Tsuji S, Tatsuno K, Takahashi H, Takahashi N, Takahashi M, Shimodaira H, Aburatani H, Ishioka C (2015) DNA methylation status as a biomarker of anti-epidermal growth factor receptor treatment for metastatic colorectal cancer. Cancer Sci 106:1722–1729. https://doi.org/10.1111/cas.12827
Issa JP (2004) CpG island methylator phenotype in cancer. Nat Rev Cancer 4:988–993. https://doi.org/10.1038/nrc1507
Issa JP (2008) Colon cancer: it’s CIN or CIMP. Clin Cancer Res 14:5939–5940. https://doi.org/10.1158/1078-0432.CCR-08-1596
Ouchi K, Takahashi S, Okita A, Sakamoto Y, Muto O, Amagai K, Okada T, Ohori H, Shinozaki E, Ishioka C (2022) A modified MethyLight assay predicts the clinical outcomes of anti-epidermal growth factor receptor treatment in metastatic colorectal cancer. Cancer Sci 113(3):1057–1068. https://doi.org/10.1111/cas.15252
Saghafinia S, Mina M, Riggi N, Hanahan D, Ciriello G (2018) Pan-cancer landscape of aberrant DNA methylation across human tumors. Cell Rep 25:1066-1080.e8. https://doi.org/10.1016/j.celrep.2018.09.082
Japanese Society for Cancer of the Colon and Rectum (2019) Japanese Classification of Colorectal, Appendiceal, and Anal Carcinoma: the 3d English Edition [Secondary Publication]. J Anus Rectum Colon, English 3:175–195. https://doi.org/10.23922/jarc.2019-018
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 48:452–458. https://doi.org/10.1038/bmt.2012.244
Lee MS, McGuffey EJ, Morris JS, Manyam G, Baladandayuthapani V, Wei W, Morris VK, Overman MJ, Maru DM, Jiang ZQ, Hamilton SR, Kopetz S (2016) Association of CpG island methylator phenotype and EREG/AREG methylation and expression in colorectal cancer. Br J Cancer 114:1352–1361. https://doi.org/10.1038/bjc.2016.87
Jacobs B, De Roock W, Piessevaux H, Van Oirbeek R, Biesmans B, De Schutter J, Fieuws S, Vandesompele J, Peeters M, Van Laethem JL, Humblet Y, Pénault-Llorca F, De Hertogh G, Laurent-Puig P, Van Cutsem E, Tejpar S (2009) Amphiregulin and epiregulin mRNA expression in primary tumors predicts outcome in metastatic colorectal cancer treated with cetuximab. J Clin Oncol 27:5068–5074. https://doi.org/10.1200/JCO.2008.21.3744
Jonker DJ, Karapetis CS, Harbison C, O’Callaghan CJ, Tu D, Simes RJ, Malone DP, Langer C, Tebbutt N, Price TJ, Shapiro J, Siu LL, Wong RPW, Bjarnason G, Moore MJ, Zalcberg JR, Khambata-Ford S (2014) Epiregulin gene expression as a biomarker of benefit from cetuximab in the treatment of advanced colorectal cancer. Br J Cancer 110:648–655. https://doi.org/10.1038/bjc.2013.753
Khambata-Ford S, Garrett CR, Meropol NJ, Basik M, Harbison CT, Wu S, Wong TW, Huang X, Takimoto CH, Godwin AK, Tan BR, Krishnamurthi SS, Burris HA, Poplin EA, Hidalgo M, Baselga J, Clark EA, Mauro DJ (2007) Expression of epiregulin and amphiregulin and K-ras mutation status predict disease control in metastatic colorectal cancer patients treated with cetuximab. J Clin Oncol 25:3230–3237. https://doi.org/10.1200/JCO.2006.10.5437
Tejpar S, Stintzing S, Ciardiello F, Tabernero J, Van Cutsem EV, Beier F, Esser R, Lenz HJ, Heinemann V (2017) Prognostic and predictive relevance of primary tumor location in patients with RAS wild-type metastatic colorectal cancer: retrospective analyses of the CRYSTAL and FIRE-3 trials. JAMA Oncol 3:194–201. https://doi.org/10.1001/jamaoncol.2016.3797
Arnold D, Lueza B, Douillard JY, Peeters M, Lenz HJ, Venook A, Heinemann V, Van Cutsem EV, Pignon JP, Tabernero J, Cervantes A, Ciardiello F (2017) Prognostic and predictive value of primary tumour side in patients with RAS wild-type metastatic colorectal cancer treated with chemotherapy and EGFR directed antibodies in six randomized trials. Ann Oncol 28:1713–1729. https://doi.org/10.1093/annonc/mdx175
Yoshino T, Arnold D, Taniguchi H, Pentheroudakis G, Yamazaki K, Xu RH, Kim TW, Ismail F, Tan IB, Yeh KH, Grothey A, Zhang S, Ahn JB, Mastura MY, Chong D, Chen LT, Kopetz S, Eguchi-Nakajima T, Ebi H, Ohtsu A, Cervantes A, Muro K, Tabernero J, Minami H, Ciardiello F, Douillard JY (2018) Pan-Asian adapted ESMO consensus guidelines for the management of patients with metastatic colorectal cancer: a JSMO-ESMO initiative endorsed by CSCO, KACO, MOS, SSO and TOS. Ann Oncol 29:44–70. https://doi.org/10.1093/annonc/mdx738
Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T, Hasegawa K, Hotta K, Ishida H, Ishiguro M, Ishihara S, Kanemitsu Y, Kinugasa Y, Murofushi K, Nakajima TE, Oka S, Tanaka T, Taniguchi H, Tsuji A, Uehara K, Ueno H, Yamanaka T, Yamazaki K, Yoshida M, Yoshino T, Itabashi M, Sakamaki K, Sano K, Shimada Y, Tanaka S, Uetake H, Yamaguchi S, Yamaguchi N, Kobayashi H, Matsuda K, Kotake K, Sugihara K, Japanese Society for Cancer of the Colon and Rectum (2020) Japanese society for cancer of the colon and rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Guidelines:1–42. Int J Clin Oncol 25:1–42. https://doi.org/10.1007/s10147-019-01485-z
Benson AB, Venook AP, Al-Hawary MM, Arain Ma CY-J, Ciombor KK, Cohen S, Cooper HS, Deming D, Farkas L, Garrido-Laguna I, Grem JL, Gunn A, Hecht JR, Hoffe S, Hubbard J, Hunt S, Johung KL, Kirilcuk N, Krishnamurthi S, Messersmith WA, Meyerhardt J, Miller ED, Mulcahy MF, Nurkin S, Overman MJ, Parikh A, Patel H, Pedersen K, Saltz L, Schneider C, Shibata D, Skibber JM, Sofocleous CT, Stoffel EM, Stotsky-Himelfarb E, Willett CG, Gregory KM, Gurski LA (2021) Colon cancer. J Natl Compr Canc Network JNCCN, version 2.2021. NCCN Clinical Practice Guidelines in Oncology 19:329–359
Wu C, Bekaii-Saab T (2012) CpG island methylation, microsatellite instability, and BRAF mutations and their clinical application in the treatment of colon cancer. Chemother Res Pract 2012. https://doi.org/10.1155/2012/359041
Acknowledgements
We thank Ms. Hitomi Hannan, Ms. Yukie Naito, and Ms. Yuki Horiike for data management. We also thank Dr Hiroshi Kawachi for tissue sample preparation.
Funding
This work was supported by a grant from the Project for Development of Innovative Research on Cancer Therapeutics (P-DIRECT) (grant number 11110018) and the Project for Cancer Research and Therapeutic Evolution (P-CREATE) by the Japan Agency for Medical Research and Development (grant number 16770660).
Author information
Authors and Affiliations
Contributions
Conception and design: HO and ES. Development of methodology: KO, ST, and CI. Acquisition of data: HO, KO, ES, ST, and CI. Analysis and interpretation of data (e.g., statistical analysis, biostatistics, and computational analysis): HO and ES. Writing, review, and/or revision of the manuscript: all authors. Administrative, technical, or material support (i.e., reporting or organizing data, and constructing databases): KO, ST, and CI. Study supervision: ES.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Institutional Review Board of the Japanese Foundation for Cancer Research (Tokyo, Japan, registry number 2019–1034). The protocol was described on the hospital website, and subjects were provided the opportunity to opt out; therefore, no additional consent was required from patients. All methods were performed in accordance with the Declaration of Helsinki.
Conflict of interest
Dr. Takahashi has received honoraria from Taiho, Chugai, Asahikasei, Bayer, Japan Blood Products Organization, Medicon, Termo, Sanofi, Merckbiopharma, Nippon-kayaku, Takeda, Ono, and Yakult and research grant from Merckbiopharma; Ono. Dr. Shinozaki has received honoraria from Taiho, Chugai, Merckbiopharma, Takeda, Sanofi, Eli Lilly, and Daiichi-Sankyo; and Ono. Pr. Ishioka has received honoraria from Chugai, Taiho, Ono, Merckbiopharma, Novartis, Asahi Kasei, Sanofi, Nippon-Kayaku, Daiichi Sankyo, Takeda, Bayer, Eisai, Eli Lilly, Teijin, Hitachi, Bristol-Myers Squibb, Konica Minolta, Pfizer, and Yakult as well as research grant from Hitachi, Riken Genesis, Yakult, Taiho, Ono, Asahi Kasei, Sanofi, Takeda, Eisai, Chugai, Kyowa Kirin, Tsumura, Eli Lilly, Otsuka, Shionogi, Novartis, Daiichi Sankyo, and Nippon-Kayaku. All other authors declared no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Osumi, H., Ouchi, K., Shinozaki, E. et al. Effect of DNA methylation status on first-line anti-epidermal growth factor receptor treatment in patients with metastatic colorectal cancer. Int J Colorectal Dis 37, 1439–1447 (2022). https://doi.org/10.1007/s00384-022-04177-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-022-04177-9