Abstract
Purpose
To compare the standard treatment, diltiazem gel 2%, with Levorag® Emulgel for chronic anal fissures.
Methods
This was a single-blinded, randomised, controlled, clinical trial with a non-inferiority design. Patients with a chronic anal fissure were randomised to treatment with diltiazem or Levorag® Emulgel twice daily for 8 weeks. Primary endpoint was complete healing of the anal fissure after 12 weeks. Secondary endpoints included incidence of adverse events and efficacy on pain relief.
Results
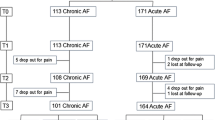
In total, 55 patients were included. Inclusion was terminated prematurely due to a slow inclusion rate. Complete fissure healing at 12 weeks follow-up was overall achieved in 31 of 55 (56%) patients, 18 of 29 (62%) in the diltiazem group compared with 13 of 26 (50%) in the Levorag® Emulgel group (P = 0.424). Pain relief was significantly better at day seven in patients treated with diltiazem (P = 0.040) compared with Levorag® Emulgel, whereas there were no differences in early (3 days) or late (12 weeks) pain relief. Three patients (10.3%) developed severe perianal exanthema during diltiazem treatment, whereas no side effects were observed in the Levorag® Emulgel group.
Conclusion
The study demonstrated statistical non-inferiority of Levorag® Emulgel compared with diltiazem in the treatment of chronic anal fissure. Diltiazem resulted in a more prompt pain relief and also in a substantial number of local allergic reactions. Levorag® Emulgel may therefore be an alternative in these patients.
Trial registration
Clinicaltrials.gov no. NCT02158013


Similar content being viewed by others
Data availability
Only the authors had full and uninhibited access to the data and the final publication right.
References
Ricciardi R, Dykes SL, Madoff RD (2009) Anal fissure. In: Beck D, Roberts P, Rombeau J et al (eds) The ASCRS Manual of Colon and Rectal Surgery, 2nd edn. Springer, pp 203–218
Bennett RC, Goligher JC (1962) Results of internal sphincterotomy for anal fissure. Br Med J 2(5318):1500–1503
Lund JN, Scholefield JH (1996) Aetiology and treatment of anal fissure. Br J Surg 83(10):1335–1344
Kocher HM, Steward M, Leather AJ, Cullen PT (2002) Randomized clinical trial assessing the side-effects of glyceryl trinitrate and diltiazem hydrochloride in the treatment of chronic anal fissure. Br J Surg 89(4):413–417. https://doi.org/10.1046/j.0007-1323.2001.02042.x
Schouten WR, Briel JW, Auwerda JJ (1994) Relationship between anal pressure and anodermal blood flow. The vascular pathogenesis of anal fissures. Dis Colon Rectum 37(7):664–669
Lund JN, Binch C, McGrath J, Sparrow RA, Scholefield JH (1999) Topographical distribution of blood supply to the anal canal. Br J Surg 86(4):496–498. https://doi.org/10.1046/j.1365-2168.1999.01026.x
Nelson RL, Thomas K, Morgan J, Jones A (2012) Non surgical therapy for anal fissure. Cochrane Database Syst Rev 2:CD003431. https://doi.org/10.1002/14651858.CD003431.pub3
Samim M, Twigt B, Stoker L, Pronk A (2012) Topical diltiazem cream versus botulinum toxin a for the treatment of chronic anal fissure: a double-blind randomized clinical trial. Ann Surg 255(1):18–22. https://doi.org/10.1097/SLA.0b013e318225178a
Blackwelder WC (1982) "Proving the null hypothesis" in clinical trials. Control Clin Trials 3(4):345–353
Nelson RL, Manuel D, Gumienny C, Spencer B, Patel K, Schmitt K, Castillo D, Bravo A, Yeboah-Sampong A (2017) A systematic review and meta-analysis of the treatment of anal fissure. Tech Coloproctol 21(8):605–625. https://doi.org/10.1007/s10151-017-1664-2
Renzi A, Brillantino A, Di Sarno G, D’Aniello F, Ziccardi S, Paladino F, Iacobellis F (2015) Myoxinol (hydrolyzed Hibiscus esculentus extract) in the cure of chronic anal fissure: early clinical and functional outcomes. Gastroenterol Res Pract 2015:567920. https://doi.org/10.1155/2015/567920
Digennaro R, Pecorella G, La Manna S, Alderisio A, Alderisio A Jr, De Pascalis B, Pennisi D, Santangelo G, Pezzolla F, Racalbuto A, Serra G, Pulvirenti D’Urso A, Altomare DF (2015) Prospective multicenter observational trial on the safety and efficacy of LEVORAG(R) Emulgel in the treatment of acute and chronic anal fissure. Tech Coloproctol 19(5):287–292. https://doi.org/10.1007/s10151-015-1289-2
Giordano P, Mistrangelo M, Cracco N, D’Elia A, Creperio G, Digito F, Paduano R, Tapparo A, Quinto S, Villa E, Pagano C (2015) Topical application of LEVORAG(R) as first-line treatment for chronic anal fissures: a preliminary multicentric study. Minerva Chir 70(5):319–325
Forkel S, Baltzer AB, Geier J, Buhl T (2018) Contact dermatitis caused by diltiazem cream and cross-reactivity with other calcium channel blockers. Contact Dermatitis 79(4):244–246. https://doi.org/10.1111/cod.13036
Opstrup MS, Guldager S, Zachariae C, Thyssen JP (2017) Systemic allergic dermatitis caused by diltiazem. Contact Dermatitis 76(6):364–365. https://doi.org/10.1111/cod.12719
Saenz de Santa Maria Garcia M, Noguerado-Mellado B, Rojas Perez-Ezquerra P, Hernandez-Aragues I, De Barrio Fernandez M (2016) Acute generalized exanthematous pustulosis due to diltiazem: investigation of cross-reactivity with other calcium channel blockers. J Allergy Clin Immunol Pract 4(4):765–766. https://doi.org/10.1016/j.jaip.2016.02.004
Acknowledgements
Research nurse Tina Lee Brøndum assisted in the generation of the EasyTrial database and management of logistical aspects of this trial. Dr. Susanne Strauss assisted in the inclusion and control of patients.
Funding
The execution of this study was supported financially by Sacomed ApS, Denmark, including the costs for the gels and the randomisation. No financial support was received from the manufacturers of the experimental or control compounds, and none of the investigators had any financial association with Sacomed ApS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All patients gave informed written consent before inclusion. The study was registered and approved by the local Ethical Committee and the Data Registration Authorities (BHH-2014-031, I-Suite No. 02946).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental figure A
Inclusion rate (JPG 825 kb)
Rights and permissions
About this article
Cite this article
Nordholm-Carstensen, A., Perregaard, H., Wahlstrøm, K.L. et al. Treatment of chronic anal fissure: a feasibility study on Levorag® Emulgel versus Diltiazem gel 2%. Int J Colorectal Dis 35, 615–621 (2020). https://doi.org/10.1007/s00384-020-03515-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-020-03515-z