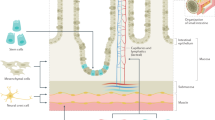
Abstract
Recent advances in culturing of intestinal stem cells and pluripotent stem cells have led to the development of intestinal organoids. These are self-organizing 3D structures, which recapitulate the characteristics and physiological features of in vivo intestinal epithelium. Intestinal organoids have allowed the development of novel in vitro models to study various gastrointestinal diseases expanding our understanding of the pathophysiology of diseases and leading to the development of innovative therapies. This article aims to summarize the current usage of intestinal organoids as a model of gastrointestinal diseases and the potential applications of intestinal organoids in infants and children. Intestinal organoids allow the study of intestinal epithelium responses to stress factors. Mimicking intestinal injury such as necrotizing enterocolitis, intestinal organoids increases the expression of pro-inflammatory cytokine genes and shows disruption of tight junctions after they are injured by lipopolysaccharide and hypoxia. In cystic fibrosis, intestinal organoids derived from rectal biopsies have provided benefits in genetic studies and development of novel therapeutic gene modulation. Transplantation of intestinal organoids via enema has been shown to rescue damaged colonic epithelium in mice. In addition, tissue-engineered small intestine derived from intestinal organoids have been successfully established providing a potential novel treatment and a new hope for children with short bowel syndrome.


Similar content being viewed by others
Abbreviations
- PSCs:
-
Pluripotent stem cells
- ASCs:
-
Adult stem cells
- Lgr5:
-
Leucine-rich repeat containing G-protein-coupled receptor 5
- NEC:
-
Necrotizing enterocolitis
- HIOs:
-
Human intestinal organoids
- TESI:
-
Tissue-engineered small intestine
- ENCCs:
-
Enteric neural crest cells
- CFTR:
-
Cystic transmembrane conductance regulator
References
Eiraku M, Sasai Y (2012) Self-formation of layered neural structures in three-dimensional culture of ES cells. Curr Opin Neurobiol 22:768–777
Lancaster MA, Knoblich JA (2014) Organogenesis in a dish: Modeling development and disease using organoid technologies. Science 345:1247125–1247125
Kretzschmar K, Clevers H (2016) Organoids: modeling development and the stem cell niche in a dish. Dev Cell 38:590–600
Dedhia PH, Bertaux-Skeirik N, Zavros Y et al (2016) Organoid models of human gastrointestinal development and disease. Gastroenterology 150:1098–1112
Clevers H (2016) Modeling development and disease with organoids. Cell 165:1586–1597
Schweiger PJ, Jensen KB (2016) Modeling human disease using organotypic cultures. Curr Opin Cell Biol 43:22–29
Dutta D, Heo I, Clevers H (2017) Disease modeling in stem cell-derived 3d organoid systems. Trends Mol Med 23:393–410
Dekkers JF, Berkers G, Kruisselbrink E et al (2016) Characterizing responses to CFTR-modulating drugs using rectal organoids derived from subjects with cystic fibrosis. Sci Transl Med 8:344ra84
Yui S, Nakamura T, Sato T et al (2012) Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5+ stem cell. Nat Med 18:618–623
Fordham RP, Yui S, Hannan NRF et al (2013) Transplantation of expanded fetal intestinal progenitors contributes to colon regeneration after injury. Cell Stem Cell 13:734–744
Fukuda M, Mizutani T, Mochizuki W et al (2014) Small intestinal stem cell identity is maintained with functional Paneth cells in heterotopically grafted epithelium onto the colon. Genes Dev 28:1752–1757
Huch M, Dorrell C, Boj SF et al (2013) In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature 494:247–250
Huch M, Bonfanti P, Boj SF et al (2013) Unlimited in vitro expansion of adult bi-potent pancreas progenitors through the Lgr5/R-spondin axis. EMBO J 32:2708–2721
Schwank G, Koo B-K, Sasselli V et al (2013) Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell 13:653–658
Volk N, Lacy B (2017) Anatomy and physiology of the small bowel. Gastrointest Endosc Clin N Am 27:1–13
Potten CS, Gandara R, Mahida YR et al (2009) The stem cells of small intestinal crypts: where are they?: stem cells of small intestinal crypts. Cell Prolif 42:731–750
Clevers H (2013) The intestinal crypt, a prototype stem cell compartment. Cell 154:274–284
Pinto D, Gregorieff A, Begthel H et al (2003) Canonical Wnt signals are essential for homeostasis of the intestinal epithelium. Genes Dev 17(14):1709–1713
Clevers H, Nusse R (2012) Wnt/β-catenin signaling and disease. Cell 149:1192–1205
Haegebarth A, Clevers H (2009) Wnt signaling, Lgr5, and stem cells in the intestine and skin. Am J Pathol 174:715–721
Milano J, McKay J, Dagenais C et al (2004) Modulation of notch processing by gamma-secretase inhibitors causes intestinal goblet cellmetaplasia and induction of genes known to specify gut secretory lineage differentiation. Toxicol Sci 82(1):341–358
Wong VWY, Stange DE, Page ME et al (2012) Lrig1 controls intestinal stem-cell homeostasis by negative regulation of ErbB signalling. Nat Cell Biol 14:401–408
Haramis AP, Begthel H, van den Born M et al (2004) De novo crypt formation and juvenile polyposis on BMP inhibition in mouse intestine. Science 303(5664):1684–1686
Kosinski C, Li VS, Chan AS et al (2007) Gene expression patterns of human colon tops and basal crypts and BM antagonists as intestinal stem cell niche factors. Proc Natl Acad Sci USA 104(39):15418–15423
Leushacke M, Barker N (2014) Ex vivo culture of the intestinal epithelium: strategies and applications. Gut 63:1345–1354
Merker SR, Weitz J, Stange DE (2016) Gastrointestinal organoids: How they gut it out. Dev Biol 420:239–250
Grün D, Lyubimova A, Kester L et al (2015) Single-cell messenger RNA sequencing reveals rare intestinal cell types. Nature 525:251–255
Barker N, van Es JH, Kuipers J et al (2007) Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449(7165):1003–1007
Sato T, Vries RG, Snippert HJ et al (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459:262–265
Sato T, van Es JH, Snippert HJ et al (2011) Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 469:415–418
Hughes CS, Postovit LM, Lajoie GA (2010) Matrigel: a complex protein mixture required for optimal growth of cell culture. Proteomics 10:1886–1890
Kim G-A, Spence JR, Takayama S (2017) Bioengineering for intestinal organoid cultures. Curr Opin Biotechnol 47:51–58
Jabaji Z, Sears CM, Brinkley GJ et al (2013) Use of collagen gel as an alternative extracellular matrix for the in vitro and in vivo growth of murine small intestinal epithelium. Tissue Eng Part C Methods 19:961–969
Jabaji Z, Brinkley GJ, Khalil HA et al (2014) Type I collagen as an extracellular matrix for the in vitro growth of human small intestinal epithelium. PLoS ONE 9:e107814
Finkbeiner SR, Freeman JJ, Wieck MM et al (2015) Generation of tissue-engineered small intestine using embryonic stem cell-derived human intestinal organoids. Biol Open 4:1462–1472
Schweinlin M, Wilhelm S, Schwedhelm I et al (2016) Development of an advanced primary human in vitro model of the small intestine. Tissue Eng Part C Methods 22:873–883
de Lau W, Barker N, Low TY et al (2011) Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature 476:293–297
Sato T, Clevers H (2013) Growing self-organizing mini-guts from a single intestinal stem cell: mechanism and applications. Science 340:1190–1194
Sato T, Stange DE, Ferrante M et al (2011) Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and barrett’s epithelium. Gastroenterology 141:1762–1772
Dignass AU, Sturm A (2001) Peptide growth factors in the intestine. Eur J Gastroenterol Hepatol 13(7):763–770
Kosinski C, Li VS, Chan AS et al (2007) Gene expression patterns of human colon tops and basal crypts and BMP antagonists as intestinal stem cell niche factors. Proc Natl Acad Sci USA 104(39):15418–15423
Navis M, Garcia TM, Renes IB et al (2018) Mouse fetal intestinal organoids: new model to study epithelial maturation from suckling to weaning. EMBO Rep. https://doi.org/10.15252/embr.201846221
Ootani A, Li X, Sangiorgi E et al (2009) Sustained in vitro intestinal epithelial culture within a Wnt-dependent stem cell niche. Nat Med 15:701–706
Pastuła A, Middelhoff M, Brandtner A et al (2016) Three-dimensional gastrointestinal organoid culture in combination with nerves or fibroblasts: a method to characterize the gastrointestinal stem cell niche. Stem Cells International 2016:1–16
Spence JR, Mayhew CN, Rankin SA et al (2011) Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470:105–109
Cao L, Gibson JD, Miyamoto S et al (2011) Intestinal lineage commitment of embryonic stem cells. Differentiation 81:1–10
Wells JM, Spence JR (2014) How to make an intestine. Development 141:752–760
Aurora M, Spence JR (2016) hPSC-derived lung and intestinal organoids as models of human fetal tissue. Dev Biol 420:230–238
Sinagoga KL, Wells JM (2015) Generating human intestinal tissues from pluripotent stem cells to study development and disease. EMBO J 34:1149–1163
Tsai Y-H, Nattiv R, Dedhia PH et al (2017) In vitro patterning of pluripotent stem cell-derived intestine recapitulates in vivo human development. Development 144:1045–1055
Mattei P, Nichol PF, Rollins MD et al (2017) Fundamentals of pediatric surgery, 2nd edn. Springer International Publishing, Berlin
Hill DR, Spence JR (2017) Gastrointestinal organoids: understanding the molecular basis of the host-microbe interface. Cell Mol Gastroenterol Hepatol 3:138–149
Leslie JL, Young VB (2016) A whole new ball game: stem cell-derived epithelia in the study of host–microbe interactions. Anaerobe 37:25–28
Senger S, Ingano L, Freire R et al (2018) Human fetal-derived enterospheres provide insights on intestinal development and a novel model to study necrotizing enterocolitis (NEC). Cell Mol Gastroenterol Hepatol 5:549–568
Li B, Lee C, Cadete M et al (2019) Neonatal intestinal organoids as an ex vivo approach to study early intestinal epithelial disorders. Pediatr Surg Int 35:3–7
Wu RY, Li B, Koike Y et al (2018) Human milk oligosaccharides increase mucin expression in experimental necrotizing enterocolitis. Mol Nutr Food Res 63:e1800658
Leslie JL, Huang S, Opp JS et al (2015) Persistence and toxin production by clostridium difficile within human intestinal organoids result in disruption of epithelial paracellular barrier function. Infect Immun 83:138–145
Wales PW, Christison-Lagay ER (2010) Short bowel syndrome: epidemiology and etiology. Semin Pediatr Surg 19:3–9
Wales PW, de Silva N, Kim J et al (2004) Neonatal short bowel syndrome: population-based estimates of incidence and mortality rates. J Pediatr Surg 39(5):690–695
Schalamon J, Mayr JM, Höllwarth ME (2003) Mortality and economics in short bowel syndrome. Best Pract Res Clin Gastroenterol 17(6):931–942
Duro D, Kalish LA, Johnston P et al (2010) Risk factors for intestinal failure in infants with necrotizing enterocolitis: a glaser pediatric research network study. J Pediatr 157:203–208.e1
Ganousse-Mazeron S, Lacaille F, Colomb-Jung V et al (2015) Assessment and outcome of children with intestinal failure referred for intestinal transplantation. Clin Nutr 34:428–435
Goulet O, Baglin-Gobet S, Talbotec C et al (2005) Outcome and long-term growth after extensive small bowel resection in the neonatal period: a survey of 87 children. Eur J Pediatr Surg 15(2):95–101
Fullerton BS, Hong CR, Jaksic T (2017) Long-term outcomes of pediatric intestinal failure. Semin Pediatr Surg 26:328–335
Coletta R, Khalil BA, Morabito A (2014) Short bowel syndrome in children: surgical and medical perspectives. Semin Pediatr Surg 23:291–297
Grant CN, Grikscheit TC (2013) Tissue engineering: a promising therapeutic approach to necrotizing enterocolitis. Semin Pediatr Surg 22:112–116
Barthel ER, Speer AL, Levin DE et al (2012) Tissue engineering of the intestine in a murine model. J Visual Exp
Levin DE, Barthel ER, Speer AL et al (2013) Human tissue-engineered small intestine forms from postnatal progenitor cells. J Pediatr Surg 48:129–137
Suita S, Taguchi T, Ieiri S et al (2005) Hirschsprung's disease in Japan: analysis of 3852 patients based on a nationwide survey in 30 years. J Pediatr Surg 40(1):197–201 (discussion 201–2)
Langer JC (2013) Hirschsprung disease. Curr Opin Pediatr 25:368–374
Wieck MM, El-Nachef WN, Hou X et al (2016) Human and murine tissue-engineered colon exhibit diverse neuronal subtypes and can be populated by enteric nervous system progenitor cells when donor colon is aganglionic. Tissue Eng Part A 22:53–64
Fattahi F, Steinbeck JA, Kriks S et al (2016) Deriving human ENS lineages for cell therapy and drug discovery in Hirschsprung disease. Nature 531:105–109
Workman MJ, Mahe MM, Trisno S et al (2017) Engineered human pluripotent-stem-cell-derived intestinal tissues with a functional enteric nervous system. Nat Med 23:49–59
Schlieve CR, Fowler KL, Thornton M et al (2017) Neural crest cell implantation restores enteric nervous system function and alters the gastrointestinal transcriptome in human tissue-engineered small intestine. Stem Cell Rep 9:883–896
Ratjen F, Bell SC, Rowe SM et al (2015) Cystic fibrosis. Nat Rev Dis Prim (Article number 15010)
Milla CE, Moss RB (2015) Recent advances in cystic fibrosis. Curr Opin Pediatr 27:317–324
Brodlie M, Haq IJ, Roberts K et al (2015) Targeted therapies to improve CFTR function in cystic fibrosis. Genome Medicine 7:101
Davies JC (2015) The future of CFTR modulating therapies for cystic fibrosis. Curr Opin Pulm Med 21:579–584
Dekkers JF, Wiegerinck CL, de Jonge HR et al (2013) A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat Med 19:939–945
Dekkers JF, van der Ent CK, Beekman JM (2013) Novel opportunities for CFTR-targeting drug development using organoids. Rare Dis 1:e27112
Duncan AW, Dorrell C, Grompe M (2009) Stem cells and liver regeneration. Gastroenterology 137:466–481
Miyajima A, Tanaka M, Itoh T (2014) Stem/progenitor cells in liver development, homeostasis, regeneration, and reprogramming. Cell Stem Cell 14:561–574
Theise ND, Saxena R, Portmann BC et al (1999) The canals of Hering and hepatic stem cells in humans. Hepatology 30(6):1425–1433
Kuwahara R, Kofman AV, Landis CS et al (2008) The hepatic stem cell niche: Identification by label-retaining cell assay. Hepatology 47:1994–2002
Zhang L, Theise N, Chua M, Reid LM (2008) The stem cell niche of human livers: symmetry between development and regeneration. Hepatology 48:1598–1607
Kordes C, Häussinger D (2013) Hepatic stem cell niches. J Clin Investig 123:1874–1880
Hindley CJ, Cordero-Espinoza L, Huch M (2016) Organoids from adult liver and pancreas: stem cell biology and biomedical utility. Dev Biol 420:251–261
Huch M, Gehart H, van Boxtel R et al (2015) Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 160:299–312
Asai A, Miethke A, Bezerra JA (2015) Pathogenesis of biliary atresia: defining biology to understand clinical phenotypes. Nat Rev Gastroenterol Hepatol 12:342–352
Si-Tayeb K, Noto FK, Nagaoka M et al (2010) Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology 51:297–305
Zhang W, Li W, Liu B et al (2012) Efficient generation of functional hepatocyte-like cells from human fetal hepatic progenitor cells in vitro. J Cell Physiol 227:2051–2058
Ma X, Duan Y, Tschudy-Seney B et al (2013) Highly efficient differentiation of functional hepatocytes from human induced pluripotent stem cells. Stem Cells Transl Med 2:409–419
Nagamoto Y, Takayama K, Tashiro K et al (2015) Efficient engraftment of human induced pluripotent stem cell-derived hepatocyte-like cells in uPA/SCID mice by overexpression of FNK, a Bcl-xLMutant gene. Cell Transplant 24:1127–1138
Ye J, Shirakigawa N, Ijima H (2015) Hybrid organoids consisting of extracellular matrix gel particles and hepatocytes for transplantation. J Biosci Bioeng 120:231–237
Ye J, Shirakigawa N, Ijima H (2016) Fetal liver cell-containing hybrid organoids improve cell viability and albumin production upon transplantation. J Biosci Bioeng 121:701–708
Acknowledgements
Prof. Agostino Pierro was supported by the Canadian Institutes of Health Research (CIHR) Foundation Grant (#353857) and the Robert M. Filler Chair of Surgery, The Hospital for Sick Children.
Funding
This study did not receive any sources of funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chusilp, S., Li, B., Lee, D. et al. Intestinal organoids in infants and children. Pediatr Surg Int 36, 1–10 (2020). https://doi.org/10.1007/s00383-019-04581-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-019-04581-3