Abstract
Purpose
Inhalation of perillyl alcohol (POH) recently emerged as an investigational promising antiglioma strategy. However, little attention has been paid to its therapeutic potential for other brain tumors, especially in the pediatric setting.
Methods
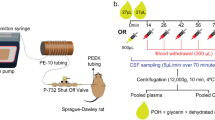
The effects of POH were explored in medulloblastoma cell models belonging to the SHH variant with activation of RAS (ONS-76) or with TP53 mutations (DAOY and UW402), by means of proliferation and invasion assays. Interactions with methotrexate, thiotepa, or ionizing radiation were also assessed. Mice bearing subcutaneous tumors were treated with intraperitoneal injections. Alternatively, animals with intracranial tumors were exposed to intranasal POH alone or combined with radiation. Tumor growth was measured by bioluminescence. Analyses of cytotoxicity to the nasal cavity were also performed, and the presence of POH in the brain, lungs, and plasma was surveyed through chromatography/mass spectrometry.
Results
POH decreased cell proliferation and colony formation, with conspicuous death, though the invasive capacity was only affected in the NRAS-mutated cell line. Median-drug effect analysis displayed synergistic combinations with methotrexate. Otherwise, POH showed to be a reasonable radiosensitizer. In vivo, intraperitoneal injection significantly decreased tumor volume. However, its inhalation did not affect orthotopic tumors, neither alone or followed by cranial irradiation. Nasal cavity epithelium showed unimportant alterations, though, no traces of POH or its metabolites were detected in tissue samples.
Conclusion
POH presents robust in vitro antimedulloblastoma effects and sensitizes cell lines to other conventional therapeutics, reducing tumor volume when administered intraperitoneally. Nevertheless, further improvement of delivery devices and/or drug formulations are needed to better characterize its effectiveness through inhalation.




Similar content being viewed by others
References
Markouli M, Strepkos D, Papavassiliou AG, Piperi C (2020) Targeting of endoplasmic reticulum (ER) stress in gliomas. Pharmacol Res 157:104823. https://doi.org/10.1016/j.phrs.2020.104823
Azimi H, Khakshur AA, Abdollahi M, Rahimi R (2015) Potential new pharmacological agents derived from medicinal plants for the treatment of pancreatic cancer. Pancreas 44:11–15. https://doi.org/10.1097/MPA.0000000000000175
Chung BH, Lee H, Lee JS, Young CYF (2006) Perillyl alcohol inhibits the expression and function of the androgen receptor in human prostate cancer cells. Cancer Lett 236:222–228. https://doi.org/10.1016/j.canlet.2005.05.023
Koyama M, Sowa Y, Hitomi T, Iizumi Y, Watanabe M, Taniguchi T, Ichikawa M, Sakai T (2013) Perillyl alcohol causes G1 arrest through p15INK4b and p21WAF1/Cip1 induction. Oncol Rep 29:779–784. https://doi.org/10.3892/or.2012.2167
Oturanel CE, Kiran I, Özsen Ö et al (2017) Cytotoxic, antiproliferative and apoptotic effects of perillyl alcohol and its biotransformation metabolite on A549 and HepG2 cancer cell lines. Anti Cancer Agents Med Chem 17. https://doi.org/10.2174/1871520617666170103093923
Reddy BS, Wang CX, Samaha H, Lubet R, Steele VE, Kelloff GJ, Rao CV (1997) Chemoprevention of colon carcinogenesis by dietary perillyl alcohol. Cancer Res 57:420–425
Stark MJ, Burke YD, McKinzie JH et al (1995) Chemotherapy of pancreatic cancer with the monoterpene perillyl alcohol. Cancer Lett 96:15–21. https://doi.org/10.1016/0304-3835(95)03912-G
Stayrook K (1997) Induction of the apoptosis-promoting protein Bak by perillyl alcohol in pancreatic ductal adenocarcinoma relative to untransformed ductal epithelial cells. Carcinogenesis 18:1655–1658. https://doi.org/10.1093/carcin/18.8.1655
Wiseman DA, Werner SR, Crowell PL (2007) Cell cycle arrest by the isoprenoids perillyl alcohol, geraniol, and farnesol is mediated by p21 Cip1 and p27 Kip1 in human pancreatic adenocarcinoma cells. J Pharmacol Exp Ther 320:1163–1170. https://doi.org/10.1124/jpet.106.111666
Yeruva L, Hall C, Elegbede JA, Carper SW (2010) Perillyl alcohol and methyl jasmonate sensitize cancer cells to cisplatin. Anti-Cancer Drugs 21:1–9. https://doi.org/10.1097/CAD.0b013e32832a68ad
Gomes AC, Mello AL, Ribeiro MG, Garcia DG, da Fonseca CO, Salazar MD’A, Schönthal AH, Quirico-Santos T (2017) Perillyl alcohol, a pleiotropic natural compound suitable for brain tumor therapy, targets free radicals. Arch Immunol Ther Exp (Warsz) 65:285–297. https://doi.org/10.1007/s00005-017-0459-5
Berndt N, Hamilton AD, Sebti SM (2011) Targeting protein prenylation for cancer therapy. Nat Rev Cancer 11:775–791. https://doi.org/10.1038/nrc3151
Campbell PM, Groehler AL, Lee KM, Ouellette MM, Khazak V, der CJ (2007) K-Ras promotes growth transformation and invasion of immortalized human pancreatic cells by Raf and phosphatidylinositol 3-kinase signaling. Cancer Res 67:2098–2106. https://doi.org/10.1158/0008-5472.CAN-06-3752
Crowell PL, Chang RR, Ren ZB, Elson CE, Gould MN (1991) Selective inhibition of isoprenylation of 21-26-kDa proteins by the anticarcinogen d-limonene and its metabolites. J Biol Chem 266:17679–17685
Gelb MH, Tamanoi F, Yokoyama K, Ghomashchi F, Esson K, Gould MN (1995) The inhibition of protein prenyltransferases by oxygenated metabolites of limonene and perillyl alcohol. Cancer Lett 91:169–175. https://doi.org/10.1016/0304-3835(95)03747-K
Ren Z, Elson CE, Gould MN (1997) Inhibition of type I and type II geranylgeranyl-protein transferases by the monoterpene perillyl alcohol in NIH3T3 cells. Biochem Pharmacol 54:113–120. https://doi.org/10.1016/S0006-2952(97)00151-2
Stayrook KR, McKinzie JH, Barbhaiya LH, Crowell PL (1998) Effects of the antitumor agent perillyl alcohol on H-Ras vs. K-Ras farnesylation and signal transduction in pancreatic cells. Anticancer Res 18:823–828
Bailey HH, Attia S, Love RR, Fass T, Chappell R, Tutsch K, Harris L, Jumonville A, Hansen R, Shapiro GR, Stewart JA (2008) Phase II trial of daily oral perillyl alcohol (NSC 641066) in treatment-refractory metastatic breast cancer. Cancer Chemother Pharmacol 62:149–157. https://doi.org/10.1007/s00280-007-0585-6
Ripple GH, Gould MN, Arzoomanian RZ, Alberti D, Feierabend C, Simon K, Binger K, Tutsch KD, Pomplun M, Wahamaki A, Marnocha R, Wilding G, Bailey HH (2000) Phase I clinical and pharmacokinetic study of perillyl alcohol administered four times a day. Clin Cancer Res 6:390–396
Ripple GH, Gould MN, Stewart JA, Tutsch KD, Arzoomanian RZ, Alberti D, Feierabend C, Pomplun M, Wilding G, Bailey HH (1998) Phase I clinical trial of perillyl alcohol administered daily. Clin Cancer Res 4:1159–1164
Chen T, da Fonseca C, Schönthal A (2018) Intranasal perillyl alcohol for glioma therapy: molecular mechanisms and clinical development. Int J Mol Sci 19:3905. https://doi.org/10.3390/ijms19123905
da Fonseca CO, Landeiro JA, Clark SS, Quirico-Santos T, da Costa Carvalho MG, Gattass CR (2006) Recent advances in the molecular genetics of malignant gliomas disclose targets for antitumor agent perillyl alcohol. Surg Neurol 65:S2–S8. https://doi.org/10.1016/j.surneu.2005.06.030
Chou AJ, Gupta R, Bell MD, Riewe KO'D, Meyers PA, Gorlick R (2013) Inhaled lipid cisplatin (ILC) in the treatment of patients with relapsed/progressive osteosarcoma metastatic to the lung. Pediatr Blood Cancer 60:580–586. https://doi.org/10.1002/pbc.24438
Huang S, Yang J-Y (2015) Targeting the hedgehog pathway in pediatric medulloblastoma. Cancers (Basel) 7:2110–2123. https://doi.org/10.3390/cancers7040880
Milde T, Lodrini M, Savelyeva L, Korshunov A, Kool M, Brueckner LM, Antunes ASLM, Oehme I, Pekrun A, Pfister SM, Kulozik AE, Witt O, Deubzer HE (2012) HD-MB03 is a novel group 3 medulloblastoma model demonstrating sensitivity to histone deacetylase inhibitor treatment. J Neuro-Oncol 110:335–348. https://doi.org/10.1007/s11060-012-0978-1
Milde T, Oehme I, Korshunov A, Kopp-Schneider A, Remke M, Northcott P, Deubzer HE, Lodrini M, Taylor MD, von Deimling A, Pfister S, Witt O (2010) HDAC5 and HDAC9 in medulloblastoma: novel markers for risk stratification and role in tumor cell growth. Clin Cancer Res 16:3240–3252. https://doi.org/10.1158/1078-0432.CCR-10-0395
Gajjar A, Stewart CF, Ellison DW, Kaste S, Kun LE, Packer RJ, Goldman S, Chintagumpala M, Wallace D, Takebe N, Boyett JM, Gilbertson RJ, Curran T (2013) Phase I study of vismodegib in children with recurrent or refractory medulloblastoma: a pediatric brain tumor consortium study. Clin Cancer Res 19:6305–6312. https://doi.org/10.1158/1078-0432.CCR-13-1425
Robinson GW, Orr BA, Wu G, Gururangan S, Lin T, Qaddoumi I, Packer RJ, Goldman S, Prados MD, Desjardins A, Chintagumpala M, Takebe N, Kaste SC, Rusch M, Allen SJ, Onar-Thomas A, Stewart CF, Fouladi M, Boyett JM, Gilbertson RJ, Curran T, Ellison DW, Gajjar A (2015) Vismodegib exerts targeted efficacy against recurrent sonic hedgehog–subgroup medulloblastoma: results from phase II pediatric brain tumor consortium studies PBTC-025B and PBTC-032. J Clin Oncol 33:2646–2654. https://doi.org/10.1200/JCO.2014.60.1591
Kieran MW, Chisholm J, Casanova M, Brandes AA, Aerts I, Bouffet E, Bailey S, Leary S, MacDonald TJ, Mechinaud F, Cohen KJ, Riccardi R, Mason W, Hargrave D, Kalambakas S, Deshpande P, Tai F, Hurh E, Geoerger B (2017) Phase I study of oral sonidegib (LDE225) in pediatric brain and solid tumors and a phase II study in children and adults with relapsed medulloblastoma. Neuro-Oncology 19:1542–1552. https://doi.org/10.1093/neuonc/nox109
Menyhárt O, Győrffy B (2019) Principles of tumorigenesis and emerging molecular drivers of <scp>SHH</scp>-activated medulloblastomas. Ann Clin Transl Neurol 6:990–1005. https://doi.org/10.1002/acn3.762
Zhao X, Ponomaryov T, Ornell KJ, Zhou P, Dabral SK, Pak E, Li W, Atwood SX, Whitson RJ, Chang ALS, Li J, Oro AE, Chan JA, Kelleher JF, Segal RA (2015) RAS/MAPK activation drives resistance to Smo inhibition, metastasis, and tumor evolution in Shh pathway-dependent tumors. Cancer Res 75:3623–3635. https://doi.org/10.1158/0008-5472.CAN-14-2999-T
Ivanov DP, Coyle B, Walker DA, Grabowska AM (2016) In vitro models of medulloblastoma: choosing the right tool for the job. J Biotechnol 236:10–25. https://doi.org/10.1016/j.jbiotec.2016.07.028
Cruzeiro GAV, Salomão KB, de Biagi Jr CAO, Baumgartner M, Sturm D, Lira RCP, de Almeida Magalhães T, Baroni Milan M, da Silva Silveira V, Saggioro FP, de Oliveira RS, dos Santos Klinger PH, Seidinger AL, Yunes JA, de Paula Queiroz RG, Oba-Shinjo SM, Scrideli CA, Nagahashi SMK, Tone LG, Valera ET (2019) A simplified approach using Taqman low-density array for medulloblastoma subgrouping. Acta Neuropathol Commun 7:33. https://doi.org/10.1186/s40478-019-0681-y
Bobola MS (2005) Apurinic/apyrimidinic endonuclease activity is associated with response to radiation and chemotherapy in medulloblastoma and primitive neuroectodermal tumors. Clin Cancer Res 11:7405–7414. https://doi.org/10.1158/1078-0432.CCR-05-1068
Kunkele A, De Preter K, Heukamp L et al (2012) Pharmacological activation of the p53 pathway by nutlin-3 exerts anti-tumoral effects in medulloblastomas. Neuro-Oncology 14:859–869. https://doi.org/10.1093/neuonc/nos115
Zomerman WW, Plasschaert SLA, Conroy S, Scherpen FJ, Meeuwsen-de Boer TGJ, Lourens HJ, Guerrero Llobet S, Smit MJ, Slagter-Menkema L, Seitz A, Gidding CEM, Hulleman E, Wesseling P, Meijer L, van Kempen LC, van den Berg A, Warmerdam DO, Kruyt FAE, Foijer F, van Vugt MATM, den Dunnen WFA, Hoving EW, Guryev V, de Bont ESJM, Bruggeman SWM (2018) Identification of two protein-signaling states delineating transcriptionally heterogeneous human medulloblastoma. Cell Rep 22:3206–3216. https://doi.org/10.1016/j.celrep.2018.02.089
dos Santos Klinger PH, Delsin LEA, Cruzeiro GAV, Andrade AF, Lira RCP, de Andrade PV, das Chagas PF, de Paula Queiroz RG, Trevisan FA, de Oliveira RS, Scrideli CA, Tone LG, Valera ET (2020) Arsenic trioxide exerts cytotoxic and radiosensitizing effects in pediatric medulloblastoma cell lines of SHH subgroup. Sci Rep 10:6836. https://doi.org/10.1038/s41598-020-63808-9
Lee Y, Shacter E (1999) Oxidative stress inhibits apoptosis in human lymphoma cells. J Biol Chem 274:19792–19798. https://doi.org/10.1074/jbc.274.28.19792
Liang C-C, Park AY, Guan J-L (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2:329–333. https://doi.org/10.1038/nprot.2007.30
Roberto GM, Paiva HH, Botelho de Souza LE, Pezuk JA, Vieira GM, de Oliveira HF, Umezawa K, Tone LG, Brassesco MS (2019) DTCM-glutarimide delays growth and radiosensitizes glioblastoma. Anti Cancer Agents Med Chem 18:1323–1329. https://doi.org/10.2174/1871520618666180423105740
Yuri T, Danbara N, Tsujita-Kyutoku M, Kiyozuka Y, Senzaki H, Shikata N, Kanzaki H, Tsubura A (2004) Perillyl alcohol inhibits human breast cancer cell growth in vitro and in vivo. Breast Cancer Res Treat 84:251–260. https://doi.org/10.1023/B:BREA.0000019966.97011.4d
Gholamin S, Feroze AH, Mitra SS, Kahn SA, Cheshier SH (2013) Establishment of stereotactic orthotopic brain tumor xenografts in mice: technical report. Cureus. https://doi.org/10.7759/cureus.150
D’Alincourt Salazar M, da Silva RF, Da Fonseca CO et al (2014) Intranasal administration of perillyl alcohol activates peripheral and bronchus-associated immune system in vivo. Arch
Geyer JR, Sposto R, Jennings M, Boyett JM, Axtell RA, Breiger D, Broxson E, Donahue B, Finlay JL, Goldwein JW, Heier LA, Johnson D, Mazewski C, Miller DC, Packer R, Puccetti D, Radcliffe J, Tao ML, Shiminski-Maher T, Children's Cancer Group (2005) Multiagent chemotherapy and deferred radiotherapy in infants with malignant brain tumors: a report from the children’s cancer group. J Clin Oncol 23:7621–7631. https://doi.org/10.1200/JCO.2005.09.095
Dhall G, Grodman H, Ji L, Sands S, Gardner S, Dunkel IJ, McCowage GB, Diez B, Allen JC, Gopalan A, Cornelius AS, Termuhlen A, Abromowitch M, Sposto R, Finlay JL (2008) Outcome of children less than three years old at diagnosis with non-metastatic medulloblastoma treated with chemotherapy on the “Head Start” I and II protocols. Pediatr Blood Cancer 50:1169–1175. https://doi.org/10.1002/pbc.21525
da Fonseca CO, Simão M, Lins IR, Caetano RO, Futuro D, Quirico-Santos T (2011) Efficacy of monoterpene perillyl alcohol upon survival rate of patients with recurrent glioblastoma. J Cancer Res Clin Oncol 137:287–293. https://doi.org/10.1007/s00432-010-0873-0
da Fonseca CO, Schwartsmann G, Fischer J, Nagel J, Futuro D, Quirico-Santos T, Gattass CR (2008) Preliminary results from a phase I/II study of perillyl alcohol intranasal administration in adults with recurrent malignant gliomas. Surg Neurol 70:259–266. https://doi.org/10.1016/j.surneu.2007.07.040
Chaudhary SC, Alam MS, Siddiqui MS, Athar M (2009) Perillyl alcohol attenuates Ras-ERK signaling to inhibit murine skin inflammation and tumorigenesis. Chem Biol Interact 179:145–153. https://doi.org/10.1016/j.cbi.2008.12.016
Lluria-Prevatt M, Morreale J, Gregus J et al (2002) Effects of perillyl alcohol on melanoma in the TPras mouse model. Cancer Epidemiol Biomark Prev 11:573–579
Sundin T, Peffley DM, Gauthier D, Hentosh P (2012) The isoprenoid perillyl alcohol inhibits telomerase activity in prostate cancer cells. Biochimie 94:2639–2648. https://doi.org/10.1016/j.biochi.2012.07.028
Castro-Gamero AM, Borges KS, Lira RC, Andrade AF, Fedatto PF, Cruzeiro GAV, Silva RB, Fontes AM, Valera ET, Bobola M, Scrideli CA, Tone LG (2013) Chromosomal heterogeneity and instability characterize pediatric medulloblastoma cell lines and affect neoplastic phenotype. Cytotechnology 65:871–885. https://doi.org/10.1007/s10616-012-9529-z
He XM, Ostrowski LE, von Wronski MA, Friedman HS, Wikstrand CJ, Bigner SH, Rasheed A, Batra SK, Mitra S, Brent TP (1992) Expression of O6-methylguanine-DNA methyltransferase in six human medulloblastoma cell lines. Cancer Res 52:1144–1148
Siddik ZH (2005) Mechanisms of action of cancer chemotherapeutic agents: DNA-interactive alkylating agents and antitumour platinum-based drugs. In: The Cancer Handbook. John Wiley & Sons, Ltd, Chichester
Rajesh D, Stenzel RA, Howard SP (2003) Perillyl alcohol as a radio-/chemosensitizer in malignant glioma. J Biol Chem 278:35968–35978. https://doi.org/10.1074/jbc.M303280200
Gómez-Contreras PC, Hernández-Flores G, Ortiz-Lazareno PC, del Toro-Arreola S, Delgado-Rizo V, Lerma-Díaz JM, Barba-Barajas M, Domínguez-Rodríguez JR, Bravo Cuellar A (2006) In vitro induction of apoptosis in U937 cells by perillyl alcohol with sensitization by pentoxifylline: increased BCL-2 and BAX protein expression. Chemotherapy 52:308–315. https://doi.org/10.1159/000096003
Cho H-Y, Wang W, Jhaveri N, Torres S, Tseng J, Leong MN, Lee DJ, Goldkorn A, Xu T, Petasis NA, Louie SG, Schönthal AH, Hofman FM, Chen TC (2012) Perillyl alcohol for the treatment of temozolomide-resistant gliomas. Mol Cancer Ther 11:2462–2472. https://doi.org/10.1158/1535-7163.MCT-12-0321
Rajesh D, Howard SP (2003) Perillyl alcohol mediated radiosensitization via augmentation of the Fas pathway in prostate cancer cells. Prostate 57:14–23. https://doi.org/10.1002/pros.10269
Djupesland PG (2013) Nasal drug delivery devices: characteristics and performance in a clinical perspective—a review. Drug Deliv Transl Res 3:42–62. https://doi.org/10.1007/s13346-012-0108-9
Ris MD, Packer R, Goldwein J, Jones-Wallace D, Boyett JM (2001) Intellectual outcome after reduced-dose radiation therapy plus adjuvant chemotherapy for medulloblastoma: a children’s cancer group study. J Clin Oncol 19:3470–3476. https://doi.org/10.1200/JCO.2001.19.15.3470
Mulhern RK, Palmer SL, Reddick WE, Glass JO, Kun LE, Taylor J, Langston J, Gajjar A (2001) Risks of young age for selected neurocognitive deficits in medulloblastoma are associated with white matter loss. J Clin Oncol 19:472–479. https://doi.org/10.1200/JCO.2001.19.2.472
Jamison S, Lin Y, Lin W (2015) Pancreatic endoplasmic reticulum kinase activation promotes medulloblastoma cell migration and invasion through induction of vascular endothelial growth factor A. PLoS One 10:e0120252. https://doi.org/10.1371/journal.pone.0120252
DA Fonseca CO, Teixeira RM, Silva JCT et al (2013) Long-term outcome in patients with recurrent malignant glioma treated with perillyl alcohol inhalation. Anticancer Res 33:5625–5631
Zhang Z, Chen H, Chan KK, Budd T, Ganapathi R (1999) Gas chromatographic-mass spectrometric analysis of perillyl alcohol and metabolites in plasma. J Chromatogr B Biomed Sci Appl 728:85–95
Murren JR, Pizzorno G, DiStasio SA et al (2002) Phase I study of perillyl alcohol in patients with refractory malignancies. Cancer Biol Ther 1:130–135. https://doi.org/10.4161/cbt.57
Lima DC, Rodrigues SV, Boaventura GT, Cho HY, Chen TC, Schönthal AH, da Fonseca CO (2020) Simultaneous measurement of perillyl alcohol and its metabolite perillic acid in plasma and lung after inhalational administration in Wistar rats. Drug Test Anal 12:268–279. https://doi.org/10.1002/dta.2722
Anselmo AC, Gokarn Y, Mitragotri S (2019) Non-invasive delivery strategies for biologics. Nat Rev Drug Discov 18:19–40. https://doi.org/10.1038/nrd.2018.183
Dhuria SV, Hanson LR, Frey WH (2010) Intranasal delivery to the central nervous system: mechanisms and experimental considerations. J Pharm Sci 99:1654–1673. https://doi.org/10.1002/jps.21924
Chen T, Da Fonseca C, Schönthal A (2016) Perillyl alcohol and its drug-conjugated derivatives as potential novel methods of treating brain metastases. Int J Mol Sci 17:1463. https://doi.org/10.3390/ijms17091463
Chen T, Chan N, Labib S et al (2018) Induction of pro-apoptotic endoplasmic reticulum stress in multiple myeloma cells by NEO214, perillyl alcohol conjugated to rolipram. Int J Mol Sci
Acknowledgments
The authors would like to thank Ieda Regina dos Santos and Cleide Lucia Araujo Silva for their assistance with the in vivo model, and Elderes de Paula, who performed the irradiation of animals. We also thank Adriana de Andrade Batista Murashima and Marina Zilio Fantucci for technical support.
Funding
This study was supported by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) Proc. N° 2013/23881-2 and 2016/08125-5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed were in accordance with the ethical standards of the institutional Committee on Ethics in the Use of Animals (CEUA) from the University of São Paulo Ribeirão Preto Campus (Protocol 14.1.541.53.8).
Informed consent
For this type of study, formal consent was not required.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 14 kb)
Supplementary figure 1
Chromatograms with retention time of standard POH samples and spectrometer with fragmentation and mass of this compound. A) POH at a concentration of 500μg/ml; B) POH at a concentration of 50μg/ml. (PNG 75 kb)
Supplementary figure 2
Representative chromatograms of brain, lung and plasma samples from control and POH-treated mice. No POH could be detected in any of the samples. A) Control group without receiving any treatment B) Group treated with 84μg POH collected after 15 min; C) Samples from the group treated with a dose of 84 μg POH, collected after 2 hours. (PNG 56 kb)
Rights and permissions
About this article
Cite this article
Silva, M., de Sousa, G.R., Simões, S.C. et al. Perillyl alcohol for pediatric TP53- and RAS-mutated SHH-medulloblastoma: an in vitro and in vivo translational pre-clinical study. Childs Nerv Syst 37, 2163–2175 (2021). https://doi.org/10.1007/s00381-021-05115-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-021-05115-w