Abstract
Purpose
Brainstem abscess is a rare condition accounting for merely 1% of brain abscesses incidence in the pediatric population. This study aimed to present a single patient with a pontine abscess and review the literature to highlight clinical features, diagnosis, and management of brainstem abscess.
Methods
The PubMed database was screened for English-language articles concerning pediatric brainstem abscess. We, therefore, identified 22 publications, which concisely depict 23 cases. Our study reports on the 24th pediatric patient diagnosed with that entity. All included reports were analyzed in terms of clinical presentation, diagnosis, management, and outcomes of described patients.
Results
There was slight women predominance (15:9), with a mean age of occurrence 6.4 years, ranging from 7 months to 16 years. Pons was the most common location of brainstem abscess, occurring in 75% of patients. Clinically, they mostly presented with cranial nerves palsy (79.2%), hemiparesis (66.7%), and pyramidal signs (45.8%). The classic triad of symptoms, including fever, headache, and the focal neurologic deficit was present in 20.8% of patients. Positive pus cultures were obtained in 61.1%. Streptococci and Staphylococci were the most frequently identified pus microorganisms. Outcomes were satisfactory, with a 79.2% rate of general improvement.
Conclusions
Neurosurgical aspiration is a safe and beneficial therapeutic method. It should always be considered and should promptly be performed when the conservative treatment is not successful and clinical deterioration occurs. Prognosis in pediatric brainstem abscess is generally favorable. Most patients recover with minor neurologic deficits or improve completely.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Brain abscess is defined as a collection of pus in the brain parenchyma. It appears typically in a devitalized area or affected by poor microcirculation [1]. Its incidence is estimated between 0.4 and 0.9 per 100,000 people per year [2]. Nathoo et al. report that 42% of brain abscesses appear in the pediatric population [3]. Their typical location includes frontal, temporal, parietal lobes, and cerebellum. Merely 1% of all brain abscesses are found within the brainstem [4, 5]. To date, 23 childhood brainstem abscesses were described in the literature, and we report on the 24th case [5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26]. Typical triad of symptoms, including fever, headache, and the focal neurologic deficit, is being found in merely up to 28% of patients diagnosed with brain abscess [27]. Clinical presentation varies depending on abscess location in the brain. In up to 35% of patients, the primary source of infection cannot be determined; therefore, they are classified as cryptogenic [4, 28, 29]. Streptococci and Staphylococci are the most frequently isolated microorganisms, followed by anaerobes and Gram-negative enteric bacteria [30, 31]. We report on a single pediatric patient diagnosed with a pontine abscess caused by Streptococcus intermedius of undetermined origin. Furthermore, we reviewed English-language literature to discuss the diagnosis and management of brainstem abscess in children.
Historical background
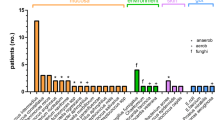
The first pediatric patient diagnosed with brainstem abscess was presented in 1974 by Danziger et al. [8]. To date, 23 cases of that entity were described in world literature, and we present the 24th pediatric patient. The mean age of occurrence in cerebral abscesses ranges between 4 and 7 years [28]. Considering brainstem abscesses, the mean age of occurrence during childhood was 6.4 years, ranging from 7 months to 16 years. Pontine location was the most common, found in eighteen patients (75%); ten children had a midbrain location (41.7%), whereas only four patients (16.7%) were diagnosed with an abscess in the medulla oblongata (Table 1). Abscess involving more than one part of the brainstem was seen in 7 patients (29.2%); thereof, only one patient presented with entire brainstem neuroaxis affected (midbrain, pons, and medulla).
In the pediatric population, factors predisposing to brain abscess development were found in 56–86% of patients. Amidst underlying infectious risk factors are sinusitis, periorbital and orbital cellulitis, otitis media, meningitis, and mastoiditis [4, 27, 32, 33]. No studies considering pediatric brainstem abscesses risk factors were conducted due to low incidence accounting for less than 1% of cerebral abscesses [5]. Nevertheless, our study shows that among infectious conditions, otogenic etiology was the most frequently appearing, followed by dental source and meningitis. Sinusitis was not identified in any patient. It can be easily explained since most of the sinusitis-related abscesses localize in the frontal lobes, which is also the most commonly affected cerebral location for abscesses [27]. Brainstem abscesses tend to spread through a hematogenous route from distant locations such as the lungs or heart, alternatively from primary otogenic infection [32]. Among underlying non-infectious primary predisposing conditions are congenital heart disease, immunodeficiency, and neurosurgical intervention [33]. Six brain abscesses (25%) were associated with underlying congenital heart disease, being also the most commonly identified etiology. The majority of abscesses located in the brainstem are defined as cryptogenic, with no infection source found on diagnostics. To date, nine out of 24 cases, accounting for 37.5%, were described as cryptogenic (Table 2). Literature estimates that incidence at about 15–35% of brain abscesses [34, 35].
Clinical presentation
Classic brain abscess-related triad of symptoms including headache, fever, and focal neurologic deficit appears in merely 9–28% of pediatric patients [27]. According to the 24 described brainstem abscesses, that symptomatology occurred in 5 patients (20.8%). Typically, headache is the most common symptom and fever being the second accompanying brain abscess [29]. In our series, the headache was present in 12 (50%) and fever in 13 patients (54.2%). Neurological deficits depend on abscess location. Brainstem abscesses tend to reveal cranial nerve palsy and disturbances in afferent and efferent long tracts, causing typically hemiparesis [36]. Cranial nerve palsy was diagnosed in 19 patients (79.2%) and hemiparesis in 16 patients (66.7%). The most commonly affected nerves were facial (VII) and abducens (VI). However, all cranial nerve palsies were identified in pediatric brainstem abscess patients, except for the hypoglossal nerve (XII). Altered level of consciousness was found out in 10 patients (41.7%). All these patients presented with somnolence; accordingly, there was no incidence of coma at all. Typically, 30–50% of pediatric patients diagnosed with brain abscess present with seizures [37]. Notwithstanding, considering brainstem abscesses, an analysis revealed only one patient with a medical history of abscess-related seizures, accounting for just 4.2% of the entire group (Table 2).
Diagnosis
Neuroimaging
Brain imaging techniques, including MRI or CT scans, are crucial in a diagnostic process. Therefore, they should always be performed when suspicion of brain abscess exists. Since the introduction of the CT scan in 1974, the overall brain abscess–related mortality decreased from 40 to 20% [31]. Computed tomography allows localizing and identifying a number of abscesses, assessing cerebritis area, and mass effect; however, we cannot discriminate between abscess and tumor. In contrast, MRI allows making a differential diagnosis with different lesions and is associated with less toxicity and better resolution [32, 38]. MRI supplied by diffusion-weighted imaging (DWI) allows differentiating abscess from cystic or necrotic lesions. It appears as a hyperintense mass in DWI and hypointense in apparent diffusion coefficient (ADC) as a result of restricted diffusion through the abscess capsule. Sensitivity and specificity for DWI in differentiating brain abscess from other cystic lesions are estimated at 96% [39]. Therefore, many authors state that in the case of brain abscess suspicion, the MRI scan should be the imaging of choice [32, 33, 38]. CT or MRI abscess features depend on the infection phase and etiology of infection [40]. Typically, it appears hypointense on CT and T1WI MRI and hyperintense on T2WI.
Both, contrast CT- and gadolinium-enhanced MRI, reveal characteristic, regular ring enhancement, which is present at the periphery of the lesion. Usually, significant cerebritis is visible in the surrounding of the abscess, on both CT and MRI scans. Magnetic resonance spectroscopy (MRS), which enables the discovery of lesion’s metabolic profile, seems to be a very promising sequence in the diagnostic process of brain abscess. A combination of DWI with MRS can improve the specificity and sensitivity of abscess diagnosis from 61% and 62%, respectively, to 100% for both parameters [41]. Diffusion tensor imaging (DTI) can provide essential measures, significantly differentiating abscesses from abscess-mimicking glioblastomas or metastases [42]. Those modalities can be particularly helpful in establishing a diagnosis of an abscess when a clinical presentation does not clearly indicate an abscess. However, the differential diagnosis should always include high-grade glioma, whose appearance can be misleading even in DWI [43]. To date, 11 brainstem abscesses were diagnosed based on CT-scan features and eight basing on MRI scan. The remaining five abscesses were visualized via pneumoencephalography just before the CT scan introduction in 1974.
Laboratory findings and microbiology
Evaluation of blood CRP level, erythrocyte sedimentation rate (ESR), and leukocyte count could promptly indicate if pathologic mass in the brain has an infectious character. Nevertheless, inflammatory parameters can be within normal ranges in up to 30–40% of patients; therefore, blood tests cannot rule out abscess [44]. Although they do not provide any additional value in the diagnosis of brain abscess, they should routinely be included in the primal diagnostic process.
Lumbar puncture should be performed occasionally and with clear indications because of the potential risk of brain herniation. The elevated opening pressure is observed in 70% of patients [45]. Some authors state that lumbar puncture is contraindicated in suspicion of brain abscess [6]. Moreover, CSF cultures in children are usually negative unless abscess ruptured into the ventricular system, or meningitis coexists [29, 36]. It is worth mentioning that abscesses rupture promptly result in elevated intracranial pressure; therefore, lumbar puncture should be contraindicated in those cases, too [45]. Papilledema was observed on admission in two (8.3%) pediatric patients with brainstem abscess, indicating intracranial hypertension.
Furthermore, three additional patients had hydrocephalus (12.5%), which was present generally in four children (16.7%). Depicted analysis indicates that in 20.8% of children with brainstem abscess diagnosis, a lumbar puncture was initially contraindicated. CSF obtained via lumbar puncture may show mononuclear pleocytosis, protein elevation, and normal glucose levels. However, these parameters can be within normal limits in up to 16% of patients [31].
Identification of causative pathogens is crucial for the implementation of targeted antimicrobial treatment. Pathogen type depends on infection source, patient age, and underlying comorbidities [37]. They are isolated in about two-thirds of patients who did not undergo antimicrobial therapy before material collection. In contrast, patients who underwent antibiotic treatment preoperatively showed positive pus cultures in merely 35% [46]. According to previous studies, CSF cultures or blood cultures are rarely positive comparing with pus microbiology. Blood cultures are positive in 2.8–28.6%; however, studies regarding this issue are inconsistent. CSF utility is also very limited, ranging from 2.8 to 44% of positive cultures [32].
Nevertheless, meta-analysis provided by Brouwer et al. indicated that 28% of CSF cultures and 24% of blood cultures are positive [31]. In 24 described brainstem abscesses, neither CSF nor blood was helpful in the identification of microorganisms in any patient, comparing to 61.1% of positive pus cultures. Those discrepancies could be caused by a considerably lower number of performed CSF and blood microbiological tests comparing to the number of pus tests. Laboratory findings of CSF, blood, and pus are described in Table 3.
Most abscesses are monomicrobial, although about 25% can be polymicrobial, which is said to be associated mainly with otogenic infection source [29, 31]. Our analysis revealed only one positive polymicrobial abscess, however unrelated to otitis. The distribution of microorganisms is generally equal in adults and children [31]. The majority of authors state that Gram-positive cocci (Streptococci and Staphylococci) and anaerobes are the most frequently isolated pus microorganisms [27, 47]. Brouwer et al. indicated a higher incidence of Gram-negative enteric bacteria than anaerobes.
Nevertheless, these differences were not considerable; therefore, both groups of microorganisms should be considered to have comparable incidence [31]. In brainstem abscesses, Gram-positive cocci (Streptococci and Staphylococci) dominated, encompassing 58.3% of identified pathogens, followed by 33.3% of anaerobes and 8.3% of Gram-negative non-enteric bacteria (Table 3).
The identification of infection source seems to be crucial in the implementation of efficient therapy. In that way, we can presume which group of bacteria is likely to be present in the brain abscess.
That knowledge allows applying the most accurate empiric antibiotic therapy, therefore increasing the probability of successful treatment without the necessity of neurosurgical abscess drainage. Streptococci being the most frequently appearing bacteria spread through hematogenous route from distant infection sources such as endocarditis or other cardiac-related diseases [37]. Moreover, Streptococci, with a particular impact on Streptococcus intermedius, form the physiological flora of the oral cavity and upper respiratory tract. Thus, cerebral abscesses, including Streptococci, are likely to originate from continuous infection from otitis, sinusitis, or dental source [48]. It is said that cyanotic congenital heart disease is a significant risk factor for S. intermedius abscess development due to the formation of microinfarcts and the increase of blood-brain barrier permeability. Cardiogenic etiology accounts for about 25–46% of S. intermedius–related brain abscesses [30].
Moreover, according to our analysis, cardiac diseases were the most common etiologic factors for brainstem abscess in the pediatric population, forming 25% (Table 2). Staphylococci, the second most common pathogens, typically occur in abscesses resulting from trauma, neurosurgical procedure, or also in association with endocarditis [33]. Anaerobes can be found when pyogenic pulmonary disease, dental infections, or sinusitis are identified. Analysis of brainstem abscesses shows that hematogenous spread from cardiogenic infection source was positive for Peptostreptococci twice and Streptococci once. Otitis resulted in Fusobacterium isolation, meningitis was associated with Staphylococcus, and finally, dental source resulted in Haemophilus paraprophilus–positive cultures. The available literature, including our patient, suggests that 38.9% of brainstem lesions are sterile. The majority of studies suggest the overall 30% incidence of sterile pediatric brain abscesses [27, 29].
Management
A limited number of pediatric patients diagnosed with brainstem abscess preclude the establishment of treatment guidelines. Successful management should include the identification of the infection source and its intensive therapy. Theoretically, neurosurgical aspiration is superior to antibiotics only. It allows assessing microbiology of the lesion, additionally reduces the number of pathogens and disrupts abscess capsule. Better penetration of antibiotics through an impaired capsule leads to an increase in their concentration, improving the efficacy and length of antimicrobial treatment. Furthermore, neurosurgical intervention enables immediate relief of mass effect by reducing abscess volume [49]. Due to the high incidence of a focal neurologic deficit on admission (87.5%), surgical aspiration aiming to reduce mass effect exerted on brainstem structures should always be considered to gain an immediate improvement in the patient’s neurologic condition. Moreover, the location close to the ventricular system poses a threat of intraventricular rupture with subsequent fatal complications [2]. According to recommendations proposed by Arlotti et al., patients who present with GCS > 12, small, at least 2.5-cm abscess diameter, multiple abscesses, or with culture obtained from other sources (CSF/blood) should be considered for antibiotic therapy only [50]. Jamjoom suggested that children in stable clinical condition and with the availability of neuroimaging control should be treated conservatively [16]. Furthermore, the majority of authors state that surgical intervention should be performed as soon as clinical deterioration occurs or features of abscess enlargement manifest on CT scan [5, 16, 51]. Most children with brainstem abscess were treated with a standard craniotomy (14 patients, 58.3%), six (25%) were given antibiotics only, and the remaining four patients (16.7%) underwent stereotactic abscess aspiration (Table 4). No surgery-related complications were observed in both standard craniotomy and stereotactic aspiration. Due to satisfactory outcomes, safety, and immediate improvement of the patient’s clinical state, neurosurgical intervention should always be considered.
There is no evidence supporting the dominance of one surgical technique in the treatment of brainstem abscess in children. On the other hand, stereotactic or neuronavigation-assisted surgery is said to be safer and more beneficial than the open craniotomy, even when the diagnosis of brainstem abscess is not entirely established preoperatively [22]. These minimally invasive techniques allow both to assess the character of the lesion via biopsy and to perform aspiration of the pus when an abscess is confirmed. Still, these are no risk-free methods because bleeding from the abscess capsule can always occur. Further studies need to be conducted to assess the most efficient surgical technique in these patients. Empirical antibiotics should be implemented as soon as a diagnosis of a cerebral abscess is established and should include broad-spectrum intravenously administered antibiotics with good penetration through the blood-brain barrier [47]. Antibiotics are advised for 6–8 weeks of treatment time; however, theoretically, they should be administered until the abscess cavity disappears on brain MRI. Empirical therapy should cover Gram-positive, Gram-negative bacteria, and anaerobes; therefore, it is indicated to use broad-spectrum cephalosporins with metronidazole [44]. Therapy should be modified after obtaining a positive culture, according to causative microorganisms and antibiograms.
Prognosis and outcomes
Despite advances in diagnosis and therapy of cerebral abscesses, they remain a serious disease associated with high morbidity [36]. Currently, the mortality rate for brain abscesses is estimated at 5–15% values and has been decreasing to that level since the CT scan was introduced in 1974 [35]. In a recent analysis of brainstem abscesses, mortality was estimated at 12.5%, based on data concerning three pediatric patients diagnosed in 1974, 1975, and 1976. Improvement was noted in 79.1% of patients, with complete resolution of symptoms in 33.3% of patients. Children who did not make a complete neurological recovery comprised 45.8% of the overall group (Table 4). Literature suggests that no neurologic deficits or just minor sequelae are described in 70% of patients with cerebral abscesses [44]. No patient with significant disabilities on follow-up was described. To sum up, the prognosis for brainstem abscess is good, being similar to the entire group of cerebral abscesses.
Exemplary case description
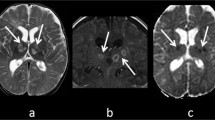
A 5-year-old girl was admitted to a local pediatric department with fever and gait disturbances. The patient was somnolent with negative meningeal signs, nasopharyngeal cavity infection, and labial herpes noticed on examination. Inflammation markers were not elevated. Acyclovir was administered due to suspected herpes encephalitis. On the admission day, significant deterioration occurred with positive meningeal signs, tetraparesis, and consciousness disturbances. Computed tomography (CT) and subsequent MRI revealed hypodense, rim-enhanced mass located in the pons with restriction of diffusion on DWI-weighted imaging (Fig. 1). Inflammation markers were still within the normal range; elevated cytosis was noticed in the CSF test. The patient was given ceftriaxone, vancomycin, and steroids; acyclovir was left off. After that, she improved clinically; however, right-sided paresis was still severe. A slight improvement was observed within a few days of intensive therapy, although the patient remained without verbal contact. No changes in abscess measurements were noticed on the control MRI scan. A few days later, significant deterioration occurred with high fever, tachypnea, anisocoria, saturation < 90%, and pulse variations 50–180/min. Urgent CT scan revealed enlargement of pathologic mass with surrounding edema (Fig. 2). On the next day, the patient was taken to the operating room for navigated abscess puncture. Posterior fossa entry-point through the middle cerebellar peduncle was chosen, and then 10 cm3 of pus was evacuated from the abscess. Postsurgical CT showed a decrease in abscess volume. The patient was still without contact, with severe right-sided paresis and anisocoria.
Antibiotic therapy was continued. Another deterioration with the presence of severe tetraparesis was observed 3 days later. MRI scan revealed a decrease of abscess volume, but the surrounding encephalitis area enlarged (Fig. 3). Microbiological examination showed Streptococcus intermedius cultured from the pus. Meropenem was administered as a targeted antibiotic. Subsequently, a mild improvement was being observed over a few next days. The patient became conscious with still severe right-sided paresis, mild on the left side.
MRI scan performed 1 month after admission showed nearly complete disappearance of the abscess (Fig. 4). Subsequently, she was discharged with the recommendation of intensive rehabilitation. During the next weeks, further clinical improvement was observed. Now, the patient is fully conscious, talking, and walking with only mild right-sided hemiparesis.
Conclusions
Brainstem abscesses are rare findings, with merely 24 pediatric patients being described in the literature so far. Neurologic appearance is typical for pathologic mass occupying the brainstem, presenting with cranial nerve palsy and long tracts impairment, typically in the form of hemiparesis. The combination of MRI spectroscopy with DWI findings would increase the specificity and sensitivity for brain abscess up to 100%. Neurosurgical aspiration is safe and beneficial and should always be considered and performed immediately when conservative therapy is not successful, and clinical deterioration occurs. It is worth mentioning that there is no evidence supporting the dominance of one surgical technique in the treatment of pediatric brainstem abscesses. Prognosis in pediatric brainstem abscess is generally favorable. Most patients recover with minor neurologic deficits or improve completely.
References
Moorthy RK, Rajshekhar V (2008) Management of brain abscess: an overview. Neurosurg Focus 24(6):E3. https://doi.org/10.3171/FOC/2008/24/6/E3
Brouwer MC, Tunkel AR, McKhann GM 2nd, van de Beek D (2014) Brain abscess. N Engl J Med 371(5):447–456. https://doi.org/10.1056/NEJMra1301635
Nathoo N, Nadvi SS, Narotam PK, van Dellen JR (2011) Brain abscess: management and outcome analysis of a computed tomography era experience with 973 patients. World Neurosurg 75(5–6):716–617. https://doi.org/10.1016/j.wneu.2010.11.043
Ozsürekci Y, Kara A, Cengiz AB, Celik M, Ozkaya-Parlakay A, Karadağ-Oncel E, Ceyhan M (2012) Brain abscess in childhood: a 28-year experience. Turk J Pediatr 54(2):144–149
Suzer T, Coskun E, Cirak B, Yagci B, Tahta K (2005) Brain stem abscesses in childhood. Childs Nerv Syst 21(1):27–31. https://doi.org/10.1007/s00381-004-1024-1
Ghannane H, Laghmari M, Aniba K, Lmejjati M, Benali SA (2011) Diagnostic and management of pediatric brain stem abscess, a case-based update. Childs Nerv Syst 27(7):1053–1062. https://doi.org/10.1007/s00381-011-1441-x
Kim JH, Jung TY, Jung SH, Lee KH, Kim SK (2015) Pediatric brainstem abscess with hemorrhage mimicking diffuse intrinsic pontine glioma: a case report. Childs Nerv Syst 31(12):2359–2362. https://doi.org/10.1007/s00381-015-2801-8
Danziger J, Allen KL, Bloch S (1974) Brain-stem abscess in childhood. Case report. J Neurosurg 40(3):391–393. https://doi.org/10.3171/jns.1974.40.3.0391
Robert CM Jr, Stern WE, Brown WJ, Greenfield MA, Bentson JR (1975) Brain stem abscess treated surgically. Wtih special note upon the employment of thorium dioxide. Surg Neurol 3(3):153–160
Kozik M, Ozarzewska E (1976) A solitary abscess of the medulla oblongata. Eur Neurol 14(4):302–309. https://doi.org/10.1159/000114752
Messina AV, Guido LJ, Liebeskind AL (1977) Preoperative diagnosis of brain-stem abscess by computerized tomography with survival. Case report. J Neurosurg 47(1):106–108. https://doi.org/10.3171/jns.1977.47.1.0106
Russell JA, Shaw MD (1977) Chronic abscess of the brain stem. J Neurol Neurosurg Psychiatry 40(7):625–629. https://doi.org/10.1136/jnnp.40.7.625
Vaquero J, Cabezudo JM, Leunda G (1980) Nonsurgical resolution of a brain-stem abscess. Case report. J Neurosurg 53(5):726–727. https://doi.org/10.3171/jns.1980.53.5.0726
Kashiwagi S, Abiko S, Aoki H (1987) Brainstem abscess. Surg Neurol 28(1):63–66. https://doi.org/10.1016/0090-3019(87)90208-4
Nauta HJ, Contreras FL, Weiner RL, Crofford MJ (1987) Brain stem abscess managed with computed tomography-guided stereotactic aspiration. Neurosurgery. 20(3):476–480. https://doi.org/10.1227/00006123-198703000-00022
Jamjoom ZA (1992) Solitary brainstem abscess successfully treated by microsurgical aspiration. Br J Neurosurg 6(3):249–253. https://doi.org/10.3109/02688699209002934
Wang HS, Kuo MF, Huang SC (1992) Medical cure of a brainstem abscess and serial brainstem auditory evoked potentials. Dev Med Child Neurol 34(10):911–915. https://doi.org/10.1111/j.1469-8749.1992.tb11389.x
Wabi G (1993) Brain stem abscess. Indian Pediatr 30(4):547–548
Rajshekhar V, Chandy MJ (1994) Successful stereotactic management of a large cardiogenic brain stem abscess. Neurosurgery. 34(2):368–371. https://doi.org/10.1227/00006123-199402000-00025
Imai H, Ono N, Zama A, Tamura M (1995) Diagnosis and treatment of brainstem abscess using magnetic resonance imaging and microsurgical aspiration--case report. Neurol Med Chir (Tokyo) 35(3):160–164. https://doi.org/10.2176/nmc.35.160
Pandian JD, Moosa NV, Cherian PJ, Radhakrishnan K (2000) Brainstem abscess complicating tetralogy of Fallot successfully treated with antibiotics alone. Neurol India 48(3):272–275
Fuentes S, Bouillot P, Regis J, Lena G, Choux M (2001) Management of brain stem abscess. Br J Neurosurg 15(1):57–62. https://doi.org/10.1080/026886901300004120
Ramdasi RV, Patil MK, Muzumdar DP (2016) Solitary pyogenic brainstem abscess. Neurol India 64(3):572–574. https://doi.org/10.4103/0028-3886.181544
Prasanna Kumar M, Krishnamurthy S, Venkateswaran VS, Mahadevan S, Lalitha M, Sistla S, Nagarajan K (2017) Brainstem micro-abscesses caused by Burkholderia pseudomallei in a 10-month-old infant: a case report. Paediatr Int Child Health 37(3):230–232. https://doi.org/10.1080/20469047.2016.1198560
Yoshikawa TT, Goodman SJ (1974) Brain abscess. West J Med 121(3):207–219
Arzoglou V, D'Angelo L, Koutzoglou M, Di Rocco C (2011) Abscess of the medulla oblongata in a toddler: case report and technical considerations based on magnetic resonance imaging tractography. Neurosurgery. 69(2):E483–E487. https://doi.org/10.1227/NEU.0b013e318218cf37
Shachor-Meyouhas Y, Bar-Joseph G, Guilburd JN, Lorber A, Hadash A, Kassis I (2010) Brain abscess in children - epidemiology, predisposing factors and management in the modern medicine era. Acta Paediatr 99(8):1163–1167. https://doi.org/10.1111/j.1651-2227.2010.01780.x
Acar M, Sutcu M, Akturk H, Muradova A, Torun SH, Salman N, Caliskan M, Izgi N, Somer A (2018) Evaluation of short-term neurological outcomes in children with brain abscesses. Turk Neurosurg 28(1):79–87. https://doi.org/10.5137/1019-5149.JTN.18672-16.1
Sáez-Llorens X (2003) Brain abscess in children. Semin Pediatr Infect Dis 14(2):108–114. https://doi.org/10.1053/spid.2003.127227
Mishra AK, Fournier PE (2013) The role of Streptococcus intermedius in brain abscess. Eur J Clin Microbiol Infect Dis 32(4):477–483. https://doi.org/10.1007/s10096-012-1782-8
Brouwer MC, Coutinho JM, van de Beek D (2014) Clinical characteristics and outcome of brain abscess: systematic review and meta-analysis. Neurology. 82(9):806–813. https://doi.org/10.1212/WNL.0000000000000172
Mameli C, Genoni T, Madia C, Doneda C, Penagini F, Zuccotti G (2019) Brain abscess in pediatric age: a review. Childs Nerv Syst 35(7):1117–1128. https://doi.org/10.1007/s00381-019-04182-4
Felsenstein S, Williams B, Shingadia D, Coxon L, Riordan A, Demetriades AK, Chandler CL, Bassi S, Koutoumanou E, Stapleton S, Sharland M, Bryant PA (2013) Clinical and microbiologic features guiding treatment recommendations for brain abscesses in children. Pediatr Infect Dis J 32(2):129–135. https://doi.org/10.1097/INF.0b013e3182748d6e
Lu CH, Chang WN, Lui CC (2006) Strategies for the management of bacterial brain abscess. J Clin Neurosci 13(10):979–985. https://doi.org/10.1016/j.jocn.2006.01.048
Brook I (2017) Microbiology and treatment of brain abscess. J Clin Neurosci 38:8–12. https://doi.org/10.1016/j.jocn.2016.12.035
Sheehan JP, Jane JA, Ray DK, Goodkin HP (2008) Brain abscess in children. Neurosurg Focus 24(6):E6. https://doi.org/10.3171/FOC/2008/24/6/E6
Bonfield CM, Sharma J, Dobson S (2015) Pediatric intracranial abscesses. J Inf Secur 71(Suppl 1):S42–S46. https://doi.org/10.1016/j.jinf.2015.04.012
Sonneville R, Ruimy R, Benzonana N, Riffaud L, Carsin A, Tadié JM, Piau C, Revest M, Tattevin P, ESCMID Study Group for Infectious Diseases of the Brain (ESGIB) (2017) An update on bacterial brain abscess in immunocompetent patients. Clin Microbiol Infect 23(9):614–620. https://doi.org/10.1016/j.cmi.2017.05.004
Reddy JS, Mishra AM, Behari S, Husain M, Gupta V, Rastogi M, Gupta RK (2006) The role of diffusion-weighted imaging in the differential diagnosis of intracranial cystic mass lesions: a report of 147 lesions. Surg Neurol 66(3):246–251. https://doi.org/10.1016/j.surneu.2006.03.032
Foerster BR, Thurnher MM, Malani PN, Petrou M, Carets-Zumelzu F, Sundgren PC (2007) Intracranial infections: clinical and imaging characteristics. Acta Radiol 48(8):875–893. https://doi.org/10.1080/02841850701477728
Lai PH, Hsu SS, Ding SW, Ko CW, Fu JH, Weng MJ, Yeh LR, Wu MT, Liang HL, Chen CK, Pan HB (2007) Proton magnetic resonance spectroscopy and diffusion-weighted imaging in intracranial cystic mass lesions. Surg Neurol 68(Suppl 1):S25–S36. https://doi.org/10.1016/j.surneu.2007.07.080
Toh CH, Wei KC, Ng SH, Wan YL, Lin CP, Castillo M (2011) Differentiation of brain abscesses from necrotic glioblastomas and cystic metastatic brain tumors with diffusion tensor imaging. AJNR Am J Neuroradiol 32(9):1646–1651. https://doi.org/10.3174/ajnr.A2581
Frassanito P, Tamburrini G, Massimi L, Caldarelli M, Di Rocco C (2015) Ghost tumors of the central nervous system: definition, clinical implications, and proposal of classification. World Neurosurg 84(3):663–670. https://doi.org/10.1016/j.wneu.2015.03.063
Brouwer MC, van de Beek D (2017) Epidemiology, diagnosis, and treatment of brain abscesses. Curr Opin Infect Dis 30(1):129–134. https://doi.org/10.1097/QCO.0000000000000334
Bernardini GL (2004) Diagnosis and management of brain abscess and subdural empyema. Curr Neurol Neurosci Rep 4(6):448–456. https://doi.org/10.1007/s11910-004-0067-8
Tonon E, Scotton PG, Gallucci M, Vaglia A (2006) Brain abscess: clinical aspects of 100 patients. Int J Infect Dis 10(2):103–109. https://doi.org/10.1016/j.ijid.2005.04.003
Chen M, Low DCY, Low SYY, Muzumdar D, Seow WT (2018) Management of brain abscesses: where are we now? Childs Nerv Syst 34(10):1871–1880. https://doi.org/10.1007/s00381-018-3886-7
Yakut N, Kadayifci EK, Karaaslan A, Atici S, Akkoc G, Ocal Demir S, Dagcinar A, Akbulut F, Soysal A, Bakır M (2015) Braın abscess due to Streptococcus intermedius secondary to mastoiditis in a child. Springerplus. 4:809. https://doi.org/10.1186/s40064-015-1608-0
Leys D, Christiaens JL, Derambure P, Hladky JP, Lesoin F, Rousseaux M, Jomin M, Petit H (1990) Management of focal intracranial infections: is medical treatment better than surgery? J Neurol Neurosurg Psychiatry 53(6):472–475. https://doi.org/10.1136/jnnp.53.6.472
Arlotti M, Grossi P, Pea F, Tomei G, Vullo V, De Rosa FG, Di Perri G, Nicastri E, Lauria FN, Carosi G, Moroni M, Ippolito G, GISIG (Gruppo Italiano di Studio sulle Infezioni Gravi) Working Group on Brain Abscesses (2010) Consensus document on controversial issues for the treatment of infections of the central nervous system: bacterial brain abscesses. Int J Infect Dis 14(Suppl 4):S79–S92. https://doi.org/10.1016/j.ijid.2010.05.010
Ruelle A, Zerbi D, Zuccarello M, Andrioli G (1991) Brain stem abscess treated successfully by medical therapy. Neurosurgery. 28(5):742–746. https://doi.org/10.1097/00006123-199105000-00020
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors state that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Antkowiak, Ł., Putz, M. & Mandera, M. Clinical features, microbiology, and management of pediatric brainstem abscess. Childs Nerv Syst 36, 2919–2926 (2020). https://doi.org/10.1007/s00381-020-04835-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-020-04835-9