Abstract
Purpose
Infant medulloblastoma (MB) is a malignant neuroepithelial embryonal tumor of the cerebellum, believed to derive from precursor granule cells with stem or progenitor cells appearance, and caused by a change in expression profile of genes related to the development. This work aims to study the expression profile of these genes in MB tumors, correlating with clinicopathological characteristics.
Methods
We quantified, by qPCR in 40 MB tumor samples, the expression of genes in HH (PTCH1, PTCH2, and GLI1), WNT (APC, CTNNB1, WIF1, and DKK2), and NOTCH pathways (NOTCH2 and HES1), which have a crucial role in development, and genes as MYCC, MYCN, and TERT, correlating this findings to patient’s clinicopathological characteristics.
Results
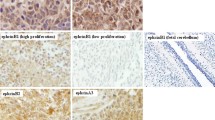
Considering the universal RNA as our control sample, and considering the median of gene expression in the control samples as our cutoff, we observed that HES1 gene showed decreased expression compared to control (p = 0.0059), but patients with HES1 overexpression were directly related to a shorter survival (p = 0.0165). Individuals with higher GLI1 gene expression had significant shorter survival (p = 0.0469), and high expression was prevalent in patients up to 5 years old (p = 0.0479). Patients showing high PTCH2 expression were related to worse survival (p = 0.0426), and it was correlated with GLI1 high expression (p = 0.0094). We also observed a concomitant overexpression of WIF1 and DKK2 genes in a subgroup of MB samples (n = 11, p = 0.0118).
Conclusions
Our results suggest the presence of activated developmental signaling pathways in MB, which are important for cell proliferation and maintenance, and that may be targeted for novel therapeutic options.




Similar content being viewed by others
References
Artavanis-Tsakonas S, Muskavitch MAT (2010) Notch: the past, the present and the future. Current Topics in Developmental Biology, vol. 92
Baek JH, Hatakeyama J, Sakamoto S, Ohtsuka T, Kageyama R (2006) Persistent and high levels of Hes1 expression regulate boundary formation in the developing central nervous system. Development 133(13):2467–2476, Epub 2006 May 25
Bell E, Chen L, Liu T, Marshall GM, Lunec J, Tweddle DA (2010) MYCN oncoprotein targets and their therapeutic potential. Cancer Lett 293(2):144–157
Ehebauer M, Hayward P, Martinez-Arias A (2006) Notch signaling pathway. Sci STKE
Gilbertson RJ, Ellison DW (2008) The origins of medulloblastoma subtypes. Annu Rev Pathol Mech Dis 3:341–365
Guessous F, Li Y, Abounader R (2008) Signaling pathways in medulloblastoma. J Cell Physiol 217:577–583
Kageyama R, Masamizu Y, Niwa Y (2007) Oscillator mechanism of notch pathway in the segmentation clock. Dev Dyn 236(6):1403–1409
Kobayashi T, Mizuno H, Imayoshi I, Furusawa C, Shirahige K, Kageyama R (2009) The cyclic gene Hes1 contributes to diverse differentiation responses of embryonic stem cells. Genes Dev 23:1870–1875
Kool M, Koster J, Bunt J, Hasselt NE, Lakeman A et al (2008) Integrated genomics identifies five medulloblastoma subtypes with distinct genetic profiles, pathway signatures and clinicopathological features. PLoS ONE 3(8):e3088
Lambiv WL, Vassallo I, Delorenzi M, Shay T, Diserens AC, Misra A, Feuerstein B, Murat A, Migliavacca E, Hamou MF, Sciuscio D, Burger R, Domany E, Stupp R, Hegi ME (2011) The Wnt inhibitory factor 1 (WIF1) is targeted in glioblastoma and has a tumor suppressing function potentially by induction of senescence. Neuro-Oncology 13(7):736–747
Lee Y, Miller HL, Russell HR, Boyd K, Curran T, McKinnon PJ (2006) Patched2 modulates tumorigenesis in patched1 heterozygous mice. Cancer Res 66(14):6964–6971
Lutz W, Leon J, Eilers M (2002) Contributions of MYC to tumorigenesis. Biochem Biophys Acta 1602:61–71
Machold R, Hayashi S, Rutlin M et al (2003) Sonic hedgehog is required for progenitor cell maintenance in telencephalic stem cell niches. Neuron 39:937–950
Manoranjan B, Venugopal C, McFarlane N, Dobe BW, Dunn SE, Scheinemann K, Singh S (2012) Medulloblastoma stem cells: where development and cancer cross pathways. Pediatric Research 71(4):516–522
Northcott PA, Jones DTW, Kool M, Robinson GW, Gilbertson RJ, Cho YJ, Pomeroy SL, Korshunov A, Lichter P, Taylor MD, Pfister SM (2012) Medulloblastomics: the end of the beginning. Nat Rev Cancer 12
Park AK, Lee SJ, Phi JH, Wang KC, Kim DG, Cho BK, Haberler C, Fattet S, Dufour C, Puget S, Sainte-Rose C, Bourdeaut F, Grill J, Delattre O, Kim SK, Park WY (2012) Prognostic classification of pediatric medulloblastoma based on chromosome 17p loss, expression of MYCC and MYCN, and Wnt pathway activation. Neuro-Oncology 14(2):203–214
Parsons W, Li M, Zhang X et al (2011) The genetic landscape of the childhood cancer medulloblastoma. Science 331:435
Peris-Bonet R, Martinez-Garcia C, Lacour B (2006) Childhood central nervous system tumours—incidence and survival in Europe (1978–1997): report from Automated Childhood Cancer Information System project. Eur J Cancer 42:2064–2080
Pierfelice T, Alberi L, Gaiano N. (2011) Notch in the vertebrate nervous system: an old dog with new tricks. Neuron 69
Pietsch T, Taylor MD, Rutka JT (2004) Molecular pathogenesis of childhood brain tumors. J Neurooncol 70:203–215
Pizzo PA, Poplack DG (2010) Principles & practice of pediatric oncology. 6a Edição. Lippincott Williams & Wilkinsm, Philadelphiam
Ponzielli R, Katz S, Barsyte-Lovejoy D, Penn LZ (2005) Cancer therapeutics: targeting the dark side of MYC. Eur J Cancer 41(16):2485–2501, Epub 2005 Oct 20
Rahnama F, Toftgård R, Zaphiropoulos PG (2004) Distinct roles of PTCH2 splice variants in hedgehog signalling. Biochem J 378(Pt 2):325–334
Rubinstein LJ (1985) Embryonal central neuroepithelial tumors and their differentiating potential. A cytogenetic view of a complex neuro-oncological problem. J Neurosurg 62:795–805
Ruiz i Altaba A, Sanchez P, Dahmane N (2002) Gli and hedgehog in cancer: tumours, embryos and stem cells. Nat Rev Cancer 2:361–372
Schreck KC, Taylor P, Marchionni L, Gopalakrishnan V, Bar EE, Gaiano N, Eberhart CG (2010) The notch target Hes1 directly modulates Gli1 expression and hedgehog signaling: a potential mechanism of therapeutic resistance. Clin Cancer Res 16(24):6060–6070
Shimojo H, Ohtsuka T, Kageyama R (2008) Oscillations in notch signaling regulate maintenance of neural progenitors. Neuron 58(1):52–64
Swartling FJ, Grimmer MR, Hackett CS, Northcott PA, Fan QW et al (2010) Pleiotropic role for MYCN in medulloblastoma. Genes Dev 24(10):1059–1072
Taylor MD, Northcott PA, Korshunov A, Remke M, Cho YJ, Clifford SC, Eberhart CG, Parsons DW, Rutkowski S, Gajjar A, Ellison DW, Lichter P, Gilbertson RJ, Pomeroy SL, Kool M, Pfister SM. (2012) Molecular subgroups of medulloblastoma: the current consensus. Acta Neuropathol
Varnum-Finney B, Xu L, Brashem-Stein C, Nourigat C, Flowers D, Bakkour S, Pear WS, Bernstein ID (2000) Pluripotent, cytokine-dependent, hematopoietic stem cells are immortalized by constitutive Notch1 signaling. Nat Med 6:1278–1281
Wang H, Mannava S, Grachtchouk V, Zhuang D, Soengas MS, Gudkov AV, Prochownik EV, Nikiforov MA (2008) c-Myc depletion inhibits proliferation of human tumor cells at various stages of the cell cycle. Oncogene 27:1905–1915
Wetmore C, Eberhart DE, Curran T (2001) Loss of p53 but not ARF accelerates medulloblastoma in mice heterozygous for patched. Cancer Res 61:513–516
Wu KJ, Grandori C, Amacker M, Simon-Vermot N, Polack A, Lingner J, Dalla-Favera R (1999) Direct activation of TERT transcription by c-MYC. Nat Genet 21:220–224
Ying S, Häcker G (2003) Apoptosis induced by direct triggering of mitochondrial apoptosis proceeds in the near-absence of some apoptotic markers. Apoptosis 12(11):2003–2011, Epub 2007 Aug 14
Zaphiropoulos PG, Undén AB, Rahnama F, Hollingsworth RE, Toftgård R (1999) PTCH2, a novel human patched gene, undergoing alternative splicing and up-regulated in basal cell carcinomas. Cancer Res 59(4):787–792
Zurawel RH, Chiappa SA, Allen C, Raffel C (1998) Sporadic medulloblastomas contain oncogenic beta-catenin mutations. Cancer Res 58(5):896–899
Acknowledgments
This work was supported by grants from FAPESP (The State of São Paulo Research Foundation: 2011/19629-0; 2011/16221-0) and GRAACC (Grupo de Apoio ao Adolescente e Criança com Câncer).
Ethical standards
Samples from each MB tumor were collected after informed consent was signed by patients/guardians according to the university’s institutional review board (IRB/Federal University of São Paulo no. 0474/11).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mascaro Cordeiro, B., Dias Oliveira, I., de Seixas Alves, M.T. et al. SHH, WNT, and NOTCH pathways in medulloblastoma: when cancer stem cells maintain self-renewal and differentiation properties. Childs Nerv Syst 30, 1165–1172 (2014). https://doi.org/10.1007/s00381-014-2403-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-014-2403-x