Abstract
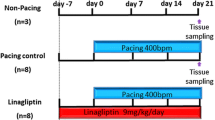
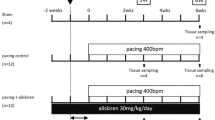
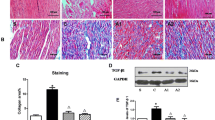
Coagulation factor Xa activates the protease-activated receptor 2 (PAR2) and causes tissue fibrosis; however, the effects of Xa inhibitor edoxaban on atrial fibrosis and atrial fibrillation (AF) have not been investigated. We examined the effect of edoxaban on the progression of atrial fibrosis in a canine congestive heart failure (CHF) model. Beagle dogs were assigned to sham, placebo, and edoxaban groups (n = 6/group). Dogs of the placebo or edoxaban groups received 19 days of medication with daily oral placebo or edoxaban, respectively, followed by 14 days of ventricular tachypacing. Dogs of the sham group had no medication or pacing. Ventricular tachypacing prolonged AF duration in dogs of the placebo group (159 ± 41 s, p < 0.01 vs. sham); however, this effect was suppressed by edoxaban treatment. Compared with the sham group, tachypacing alone also significantly increased the atrial fibrotic area (2.9 ± 0.1% vs. 7.8 ± 0.4%, p < 0.01), PAR2 expression (1.0 ± 0.1 vs. 1.8 ± 0.3, p < 0.05), and atrial fibronectin expression (1.0 ± 0.2 vs. 2.0 ± 0.2, p < 0.01). These responses were suppressed by edoxaban treatment (area 5.9 ± 0.4%, p < 0.01; PAR2 1.1 ± 0.1, p < 0.05; fibronectin 1.2 ± 0.2, p < 0.05 vs. placebo). Edoxaban showed suppressive effects on atrial remodeling, AF progression, and excessive expressions of PAR2 and fibronectin in a canine CHF model. The suppression of the Xa/PAR2 pathway might be a potential pharmacological target of edoxaban.






Similar content being viewed by others
References
Iwasaki YK, Nishida K, Kato T, Nattel S (2011) Atrial fibrillation pathophysiology: implications for management. Circulation 124:2264–2274
Nishida K, Nattel S (2014) Atrial fibrillation compendium: historical context and detailed translational perspective on an important clinical problem. Circ Res 114:1447–1452
Nattel S, Burestein B, Dobrev D (2008) Atrial remodeling and atrial fibrillation: mechanisms and implications. Circ Arrhythm Electrophysiol 1:62–73
Burstein B, Comtois P, Michael G, Nishida K, Villeneuve L, Yeh YH, Nattel S (2009) Changes in connexin expression and the atrial fibrillation substrate in congestive heart failure. Circ Res 105:1213–1222
Nishida K, Qi XY, Wakili R, Comtois P, Chaetier D, Harada M, IwasakiYK Romeo P, Maguy A, Dobrev D, Michael G, Talajic M, Nattel S (2011) Mechanisms of atrial tachyarrhythmia associated with coronary artery occlusion in a chronic canine model. Circulation 123:137–146
Burstein B, Nattel S (2008) Atrial fibrosis: mechanisms and clinical relevance in atrial fibrillation. J Am Coll Cardiol 51:802–809
Borisoff J, Spronk HM, ten Cate H (2011) The hemostatic system as a modulator of atherosclerosis. N Engl J Med 364:1746–1760
Spronk HM, de Jong AM, Crijns HJ, Schotten U, Van Gelder IC, Ten Cate H (2014) Pleiotropic effects of factor Xa and thrombin: what to expect from novel anticoagulants. Cardiovasc Res 101:344–351
Bukowska A, Zacharias I, Weinert S, Skopp K, Hartmann C, Huth C, Goette A (2013) Coagulation factor Xa induces an inflammatory signaling by activation of protease-activated receptors in human atrial tissue. Eur J Pharmacol 718:114–123
Borensztajn K, Peppelenbosch MP, Spek CA (2008) Factor Xa: at the crossroads between coagulation and signaling in physiology and disease. Trends Mol Med 14:429–440
Antoniak S, Sparkenbaugh EM, Tencati M, Rojas M, Mackman N, Pawlinski R (2013) Protease activated receptor-2 contributes to heart failure. PLoS ONE 8:e81733
Borenstajin K, Stiekema J, Nijmeijer S, Reitsma PH, Peppelenbosch MP, Spek CA (2008) Factor Xa stimulates proinflammatory and profibrotic responses in fibroblasts via protease-activated receptor-2 activation. Am J Pathol 172:309–320
Kondo H, Abe I, Fukui A, Saito S, Miyoshi M, Aoki K, Shinohara T, Teshima Y, Yufu K, Takahashi N (2018) Possible role of rivaroxaban in attenuating pressure-overload-induced atrial fibrosis and fibrillation. J Cardiol 71:310–319
Bode MF, Auriemma AC, Grove SP, Hisada Y, Rennie A, Bode WD, Vora R, Subramaniam S, Cooley B, Andrade-Gordon P, Antoniak S, Mackman N (2018) The factor Xa inhibitor rivaroxaban reduces cardiac dysfunction in a mouse model of myocardial infarction. Thromb Res 167:128–134
Imano H, Kato R, Tanikawa S, Yoshimura F, Nomura A, Ijiri Y, Yamaguchi T, Izumi Y, Yoshiyama M, Hayashi T (2018) Factor Xa inhibition by rivaroxaban attenuates cardiac remodeling due to intermittent hypoxia. J Pharmacol Sci 137:274–282
Nishida K, Michael G, Dobrev D, Nattel S (2010) Animal models for atrial fibrillation: clinical insights and scientific opportunities. Europace 12:160–172
Hanna N, Cardin S, Leung TK, Nattel S (2004) Differences in atrial versus ventricular remodeling in dogs with ventricular tachypacing-induced congestive heart failure. Cardiovasc Res 63:236–244
Furugohri T, Isobe K, Honda Y, Matsumoto C, Sugiyama N, Nagahara T, Morishima Y, Shibano T (2008) Du-176b, a potent and orally active factor Xa inhibitor: in vitro and in vivo pharmacological profiles. J Thromb Haemost 6:1542–1549
Cardin S, Libby E, Pelletier P, Bouter SL, Takeshita A, Meur NL, Leger J, Demolombe S, Ponton A, Glass L, Nattel S (2007) Contrasting gene expression profiles in two canine models of atrial fibrillation. Circ Res 100:425–433
Cardin S, Pelletier P, Libby E, Bouter SL, Xiao L, Kaab S, Demolombe S, Glass L, Nattel S (2008) Marked differences between atrial and ventricular gene-expression remodeling in dogs with experimental heart failure. J Mol Cell Cardiol 45:821–831
Lee KW, Everett T, Rahmutula D, Guerra JM, Wilson ED, Ding C, Olgin JE (2006) Pirfenidone prevents the development of a vulnerable substrate for atrial fibrillation in a canine model of heart failure. Circulation 114:1703–1712
Bukowska A, Zachrias I, Weinert S, Skopp K, Hartmann C, Huth C, Goette A (2013) Coagulation factor Xa induces an inflammatory signaling by activation of protease-activated receptors in human atrial tissue. Eur J Pharmacol 718:114–123
Maze SS, Kotler MN, Parry WR (1989) Flow characteristics in the dilated left ventricular with thrombus: qualitative and quantitative Doppler analysis. J Am Coll Cardiol 13:873–881
Yamamoto K, Ikeda U, Furuhashi K, Irokawa M, Nakayama T, Shimada K (1995) The coagulation system is activated in idiopathic cardiomyopathy. J Am Coll Cardiol 25:1634–1640
Gustavsson CG, Persson SU, Larsson H, Persson S (1994) Changed blood rheology in patients with idiopathic dilated cardiomyopathy. Angiology 45:107–111
Hashikata T, Tojo M, Namba S, Kitasato L, Kameda R, Murakami M, Niwano H, Shimohama T, Tojo T, Ako J (2015) Rivaroxaban inhibits fibrotic progression in vitro. Int Heart J 56:544–550
Ritchie E, Saka M, MacKenzie C, Drummond R, Wheeler-Jones C, Kanke T, Plevin R (2007) Cytokine upregulation of proteinase-activated-receptors 2 and 4 expression mediated by p38 MAP kinase and inhibitory kappa B kinase β in human endothelial cells. Br J Pharmacol 150:1044–1054
Qi XY, Yeh YH, Xiao L, Burstein B, Maguy A, Chaetier D, Villeneuve LR, Brundel BJ, Dobrev D, Nattel S (2008) Cellular signaling underlying atrial tachycardia remodeling of L-type calcium current. Circ Res 103:845–854
Nattel S, Li D (2000) Ionic remodeling in the heart: pathophysiological significance and new therapeutic opportunities for atrial fibrillation. Circ Res 87:440–447
Allessie M, Ausma J, Schotten U (2002) Electrical, contractile and structural remodeling during atrial fibrillation. Cardiovasc Res 54:230–246
Li D, Fareh S, Leung TK, Nattel S (1999) Promotion of atrial fibrillation by heart failure in dogs: atrial remodeling of a different sort. Circulation 100:87–95
Cha TJ, Ehrlich JR, Zhang L, Shi Y-F, Tardif J-C, Leung T-K, Nattel S (2004) Dissociation between ironic remodeling and ability to sustain atrial fibrillation during recovery from experimental congestive heart failure. Circulation 109:412–418
Goette A, Schon N, Kirchhof P, Breithardt G, Fetsch T, Hausler KG, Klein HU, Steinbeck G, Wegscheider K, Meinertz T (2012) Angiotensin II-antagonist in paroxysmal atrial fibrillation (ANTIPAF) trial. Circ Arrythm Electrophysiol 5:43–51
Rahimi K, Emberson J, McGale P, Majoni W, Merhi A, Asselbergs FW, Krane V, Macfarlane PW (2011) Effect of statin on atrial fibrillation: collaborative meta-analysis of published and unpublished evidence from randomized controlled trials. BMJ. https://doi.org/10.1136/bmj.d1250
Acknowledgments
The authors thank Kiyomi Note for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tsujino, Y., Sakamoto, T., Kinoshita, K. et al. Edoxaban suppresses the progression of atrial fibrosis and atrial fibrillation in a canine congestive heart failure model. Heart Vessels 34, 1381–1388 (2019). https://doi.org/10.1007/s00380-019-01377-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-019-01377-2