Abstract
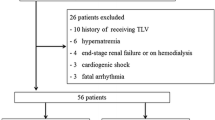
Tolvaptan (TLV) is a newly developed oral vasopressin-2 receptor antagonist that is mostly used for patients with acute decompensated heart failure (ADHF) refractory to conventional diuretic therapy. The aim of this study was to investigate the effects of outpatient TLV dose on cardiac mortality in patients discharged after hospitalization for ADHF. One hundred and five patients with ADHF who had been treated with TLV for the first time during hospitalization were retrospectively divided into three groups based on outpatient TLV use and dose. The non-TLV group comprised patients who were not treated with TLV after discharge (n = 36). Patients who continued TLV after discharge were further classified into two groups: low-dose (LD)-TLV (3.75 mg/day, n = 27) and high-dose (HD)-TLV (7.5 or 15 mg/day, n = 42). The primary endpoint was cardiac mortality. Secondary endpoint included the composite of all-cause mortality or re-hospitalization due to worsening of ADHF. There were no significant differences in demographic variables other than body mass index (p = 0.0026), echocardiographic data, laboratory data other than serum chloride before TLV administration (p = 0.041), serum sodium (p = 0.040) and potassium (p = 0.027) at discharge, and concomitant medications among the three groups. The Kaplan–Meier curve showed that the survival rate was lower in HD-TLV than in non-TLV, whereas LD-TLV showed the highest survival rate among the three groups (p = 0.0001). Multivariable Cox regression analysis of the clinical characteristics used for predicting cardiac mortality revealed that LD-TLV (hazard ratio [HR] 0.16, 95% confidence interval [CI] 0.01–0.93, p = 0.040) and HD-TLV (HR 2.43, 95% CI 1.06–6.26, p = 0.035) were significant predictors after adjustment for predictive covariates. In conclusion, the judgement of the continuation of LD-TLV according to patient hemodynamics and severity of congestion may not cause worsened prognosis.


Similar content being viewed by others
Abbreviations
- ADHF:
-
Acute decompensated heart failure
- TLV:
-
Tolvaptan
- AQP:
-
Aquaporin
- AVP:
-
Arginine vasopressin
- LD:
-
Low-dose
- EF:
-
Ejection fraction
- HD:
-
High-dose
- LV:
-
Left ventricular
- BMI:
-
Body mass index
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
References
McKee PA, Castelli WP, McNamara PM, Kannel WB (1971) The natural history of congestive heart failure: the Framingham study. N Engl J Med 285:1441–1446
McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, Falk V, Filippatos G, Fonseca C, Gomez-Sanchez MA, Jaarsma T, Køber L, Lip GY, Maggioni AP, Parkhomenko A, Pieske BM, Popescu BA, Rønnevik PK, Rutten FH, Schwitter J, Seferovic P, Stepinska J, Trindade PT, Voors AA, Zannad F, Zeiher A, ESC Committee for Practice Guidelines (2012) ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur Heart J 33:1787–1847
Abdel-Qadir HM, Tu JV, Yun L, Austin PC, Newton GE, Lee DS (2010) Diuretic dose and long-term outcomes in elderly patients with heart failure after hospitalization. Am Heart J 160:264–271
Eshaghian S, Horwich TB, Fonarow GC (2006) Relation of loop diuretic dose to mortality in advanced heart failure. Am J Cardiol 97:1759–1764
Harjai KJ, Dinshaw HK, Nunez E, Shah M, Thompson H, Turgut T, Ventura HO (1999) The prognostic implications of outpatient diuretic dose in heart failure. Int J Cardiol 71:219–225
Imamura T, Kinugawa K, Fujino T, Inaba T, Maki H, Hatano M, Yao A, Komuro I (2014) Increased urine aquaporin-2 relative to plasma arginine vasopressin is a novel marker of response to tolvaptan in patients with decompensated heart failure. Circ J 78:2240–2249
Udelson JE, Smith WB, Hendrix GH, Painchaud CA, Ghazzi M, Thomas I, Ghali JK, Selaru P, Chanoine F, Pressler ML, Konstam MA (2001) Acute hemodynamic effects of conivaptan, a dual V(1A) and V(2) vasopressin receptor antagonist, in patients with advanced heart failure. Circulation 104:2417–2423
Gheorghiade M, Konstam MA, Burnett JC Jr, Grinfeld L, Maggioni AP, Swedberg K, Udelson JE, Zannad F, Cook T, Ouyang J, Zimmer C, Orlandi C, Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study With Tolvaptan (EVEREST) Investigators (2007) Short-term clinical effects of tolvaptan, an oral vasopressin antagonist, in patients hospitalized for heart failure: the EVEREST Clinical Status Trials. JAMA 297:1332–1343
Udelson JE, McGrew FA, Flores E, Ibrahim H, Katz S, Koshkarian G, O’Brien T, Kronenberg MW, Zimmer C, Orlandi C, Konstam MA (2007) Multicenter, randomized, double-blind, placebo-controlled study on the effect of oral tolvaptan on left ventricular dilation and function in patients with heart failure and systolic dysfunction. J Am Coll Cardiol 49:2151–2159
Uemura Y, Shibata R, Takemoto K, Uchikawa T, Koyasu M, Ishikawa S, Imai R, Ozaki Y, Watanabe T, Teraoka T, Watarai M, Murohara T (2017) Safety and efficacy of long-term use of tolvaptan in patients with heart failure and chronic kidney disease. Circ J 81:1736–1738
Okabe T, Yakushiji T, Igawa W, Ono M, Kido T, Ebara S, Yamashita K, Yamamoto MH, Saito S, Hoshimoto K, Amemiya K, Isomura N, Araki H, Ochiai M (2015) The efficacy of tolvaptan in congestive heart failure patients with and without hypoalbuminemia: a pilot study. Cardiovasc Ther 33:275–281
Shimizu K, Doi K, Imamura T, Noiri E, Yahagi N, Nangaku M, Kinugawa K (2015) Ratio of urine and blood urea nitrogen concentration predicts the response of tolvaptan in congestive heart failure. Nephrology (Carlton) 20:405–412
Okayama D, Suzuki T, Shiga T, Minami Y, Tsuruoka S, Hagiwara N (2015) Blood urea nitrogen/creatinine ratio and response to tolvaptan in patients with decompensated heart failure: a retrospective analysis. Am J Cardiovasc Drugs 15:289–293
Iwatani H, Kawabata H, Sakaguchi Y, Yamamoto R, Hamano T, Rakugi H, Isaka Y (2015) Urine osmolarity predicts the body weight-reduction response to tolvaptan in chronic kidney disease patients: a retrospective, observational study. Nephron 130:8–12
Toda H, Nakamura K, Nakahama M, Wada T, Watanabe A, Hashimoto K, Terasaka R, Tokioka K, Nishii N, Miyoshi T, Kohno K, Kawai Y, Miyaji K, Koide Y, Tachibana M, Yoshioka R, Ito H, Investigators Tolvaptan Registry (2016) Clinical characteristics of responders to treatment with tolvaptan in patients with acute decompensated heart failure: IMPORTANCE of preserved kidney size. J Cardiol 67:177–183
JCS Joint Working Group (2013) Guidelines for treatment of acute heart failure (JCS 2011). Circ J 77:2157–2201
Meta-analysis Global Group in Chronic Heart Failure (MAGGIC) (2012) The survival of patients with heart failure with preserved or reduced left ventricular ejection fraction: an individual patient data meta-analysis. Eur Heart J 33:1750–1757
Nakano Y, Mizuno T, Niwa T, Mukai K, Wakabayashi H, Watanabe A, Ando H, Takashima H, Murotani K, Waseda K, Amano T (2018) Impact of continuous administration of tolvaptan on preventing medium-term worsening renal function and long-term adverse events in heart failure patients with chronic kidney disease. Int Heart J 59:105–111
Uemura Y, Shibata R, Takemoto K, Uchikawa T, Koyasu M, Ishikawa S, Mitsuda T, Miura A, Imai R, Iwamiya S, Ozaki Y, Kato T, Miura T, Watarai M, Murohara T (2016) Clinical benefit of tolvaptan in patients with acute decompensated heart failure and chronic kidney disease. Heart Vessels 31:1643–1649
Hanatani A, Shibata A, Kitada R, Iwata S, Matsumura Y, Doi A, Sugioka K, Takagi M, Yoshiyama M (2017) Administration of tolvaptan with reduction of loop diuretics ameliorates congestion with improving renal dysfunction in patients with congestive heart failure and renal dysfunction. Heart Vessels 32:287–294
Matsukawa R, Kubota T, Okabe M, Yamamoto Y (2016) Early use of V2 receptor antagonists is associated with a shorter hospital stay and reduction in in-hospital death in patients with decompensated heart failure. Heart Vessels 31:1650–1658
Kimura K, Momose T, Hasegawa T, Morita T, Misawa T, Motoki H, Izawa A, Ikeda U (2016) Early administration of tolvaptan preserves renal function in elderly patients with acute decompensated heart failure. J Cardiol 67:399–405
Ambrosy AP, Pang PS, Khan S, Konstam MA, Fonarow GC, Traver B, Maggioni AP, Cook T, Swedberg K, Burnett JC Jr, Grinfeld L, Udelson JE, Zannad F, Gheorghiade M, Trial Investigators EVEREST (2013) Clinical course and predictive value of congestion during hospitalization in patients admitted for worsening signs and symptoms of heart failure with reduced ejection fraction: findings from the EVEREST trial. Eur Heart J 34:835–843
Jujo K, Saito K, Ishida I, Furuki Y, Kim A, Suzuki Y, Sekiguchi H, Yamaguchi J, Ogawa H, Hagiwara N (2016) Randomized pilot trial comparing tolvaptan with furosemide on renal and neurohumoral effects in acute heart failure. ESC Heart Fail 3:177–188
Kinugawa K, Inomata T, Sato N, Yasuda M, Shimakawa T, Bando K, Fukuta Y (2017) Who needs longer tolvaptan treatment? Int Heart J 58:30–35
Konstam MA, Gheorghiade M, Burnett JC Jr, Grinfeld L, Maggioni AP, Swedberg K, Udelson JE, Zannad F, Cook T, Ouyang J, Zimmer C, Orlandi C, Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study With Tolvaptan (EVEREST) Investigators (2007) Effects of oral tolvaptan in patients hospitalized for worsening heart failure: the EVEREST Outcome Trial. JAMA 297:1319–1331
Shoaf SE, Bricmont P, Dandurand A (2017) Low-dose tolvaptan PK/PD: comparison of patients with hyponatremia due to syndrome of inappropriate antidiuretic hormone secretion to healthy adults. Eur J Clin Pharmacol 73:1399–1408
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
All procedures performed in this study were conducted with the patients’ informed consent, and complied with the national ethical guidelines for medical and health research involving human subjects and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Matsumoto, K., Ehara, S., Nakamura, Y. et al. The effects of tolvaptan dose on cardiac mortality in patients with acute decompensated heart failure after hospital discharge. Heart Vessels 33, 1204–1213 (2018). https://doi.org/10.1007/s00380-018-1177-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-018-1177-6