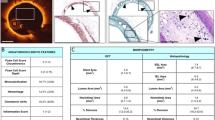
Abstract
Although Nobori®, with a bioresorbable polymer and biolimus A9 abluminal coating, has unique characteristics, few data exist regarding endothelialization early after implantation. Fifteen Nobori® and 14 control bare-metal stents (S-stent™) were implanted in 12 pigs. Histopathology of stented segments, inflammation, and intimal fibrin content was evaluated on the 2nd and 14th day after implantation. On the 2nd day, endothelial cells were morphologically and immunohistologically confirmed on the surface of both stents, although some inflammatory cells might be involved. Stent surface endothelialization evaluated with a scanning electron microscope showed partial cellular coverage in both stents. On the 14th day, neointimal thickness and percentage of the neointimal area were significantly lower in Nobori® than in S-stent™ (51.4 ± 4.5 vs. 76.4 ± 23.6 µm, p < 0.05 and 10.8 ± 2.6 vs. 14.1 ± 4.2%, p < 0.01). No significant differences were found in these parameters on the 2nd day (17.3 ± 14.9 vs. 26.7 ± 13.6 µm and 3.7 ± 3.0 vs. 6.7 ± 3.7%), in inflammatory and intimal fibrin content scores. These results demonstrate that endothelialization could occur early after Nobori® implantation with similar inflammatory reaction to bare-metal stents, probably contributing to low frequency of in-stent thrombosis and restenosis.





Similar content being viewed by others
References
Versaci F, Gaspardone A, Tomai F, Crea F, Chiariello L, Gioffrè PA (1997) A comparison of coronary-artery stenting with angioplasty for isolated stenosis of the proximal left anterior descending coronary artery. N Engl J Med 336:817–822
Grines CL, Cox DA, Stone GW, Garcia E, Mattos LA, Giambartolomei A, Brodie BR, Madonna O, Eijgelshoven M, Lansky AJ, O’Neill WW, Morice MC (1999) Coronary angioplasty with or without stent implantation for acute myocardial infarction. Stent primary angioplasty in myocardial infarction study group. N Engl J Med 341:1949–1956
Uchiyama K, Ino H, Hayashi K, Fujioka K, Takabatake S, Yokawa J, Namura M, Mizuno S, Tatami R, Kanaya H, Nitta Y, Michishita I, Hirase H, Ueda K, Aoyama T, Okeie K, Haraki T, Mori K, Araki T, Minamoto M, Oiwake H, Konno T, Sakata K, Kawashiri M, Yamagishi M (2011) Impact of severe coronary disease associated or not associated with diabetes mellitus on outcome of interventional treatment using stents: results from HERZ (Heart Research Group of Kanazawa) analyses. J Int Med Res 39:549–557
Kastrati A, Dibra A, Mehilli J, Mayer S, Pinieck S, Pache J, Dirschinger J, Schömig A (2006) Predictive factors restenosis after coronary implantation of sirolimus- or paclitaxel-eluting stents. Circulation 113:2293–2300
Serruys PW, Kutryk MJ, Ong AT (2006) Coronary-artery stents. N Engl J Med 354:483–495
Yoshida T, Sakata K, Nitta Y, Taguchi T, Kaku B, Katsuda S, Shimojima M, Gamou T, Nakahashi T, Konno T, Kawashiri MA, Yamagishi M, Hayashi K (2015) Short- and long-term benefits of drug-eluting stents compared to bare metal stents even in treatment for large coronary arteries. Heart Vessels 31:635–642
Kawashiri MA, Sakata K, Uchiyama K, Konno T, Namura M, Mizuno S, Tatami R, Kanaya H, Nitta Y, Michishita I, Hirase H, Ueda K, Aoyama T, Okeie K, Haraki T, Mori K, Araki T, Minamoto M, Oiwake H, Ino H, Hayashi K, Yamagishi M (2014) Impact of lesion morphology and associated procedures for left main coronary stenting on angiographic outcome after intervention: sub-analysis of Heart Research Group of Kanazawa, HERZ, Study. Cardiovasc Interv Ther 29:117–122
Kobayashi N, Ito Y, Hirano K, Yamawaki M, Araki M, Sakai T, Takimura H, Sakamoto Y, Mori S, Tsutsumi M, Takama T, Takafuji H, Maruyama T, Honda Y, Tokuda T, Makino K, Shirai S, Muramatsu T (2016) Comparison of first- and second-generation drug-eluting stent efficacies for treating left main and/or three-vessel disease: a propensity matched study. Heart Vessels 31:1930–1942
Joner M, Finn AV, Farb A, Mont EK, Kolodgie FD, Ladich E, Kutys R, Skorija K, Gold HK, Virmani R (2006) Pathology of drug-eluting stents in humans: delayed healing and late thrombotic risk. J Am Coll Cardiol 48:193–202
Cook S, Ladich E, Nakazawa G, Eshtehardi P, Neidhart M, Vogel R, Togni M, Wenaweser P, Billinger M, Seiler C, Gay S, Meier B, Pichler WJ, Jüni P, Virmani R, Windecker S (2009) Correlation of intravascular ultrasound findings with histopathological analysis of thrombus aspirates in patients with very late drug-eluting stent thrombosis. Circulation 120:391–399
Sakata K, Namura M, Takagi T, Tama N, Inoki I, Terai H, Horita Y, Ikeda M, Yamagishi M (2015) Repeated occurrence of slow flow phenomenon during and late after sirolimus-eluting stent implantation. Heart Vessels 30:406–409
Kurosawa T, Kotani J, Matsuyama TA, Ishibashi-Ueda H (2015) A case of active peri-stent inflammation after sirolimus-eluting stent implantation. Heart Vessels 30:270–273
Finn AV, Joner M, Nakazawa G, Kolodgie F, Newell J, John MC, Gold HK, Virmani R (2007) Pathological correlates of late drug-eluting stent thrombosis: strut coverage as a marker of endothelialization. Circulation 115:2435–2441
Pfisterer M, Brunner-La Rocca HP, Buser PT, Rickenbacher P, Hunziker P, Mueller C, Jeger R, Bader F, Osswald S, Kaiser C, Investigators BASKET-LATE (2006) Late clinical events after clopidogrel discontinuation may limit the benefit of drug-eluting stents. J Am Coll Cardiol 48:2584–2591
Virmani R, Guagliumi G, Farb A, Musumeci G, Grieco N, Motta T, Mihalcsik L, Tespili M, Valsecchi O, Kolodgie FD (2004) Localized hypersensitivity and late coronary thrombosis secondary to a sirolimus-eluting stent: should we be cautious? Circulation 109:701–705
Nebeker JR, Virmani R, Bennett CL, Hoffman JM, Samore MH, Alvarez J, Davidson CJ, McKoy JM, Raisch DW, Whisenant BK, Yarnold PR, Belknap SM, West DP, Gage JE, Morse RE, Gligoric G, Davidson L, Feldman MD (2006) Hypersensitivity cases associated with drug-eluting coronary stents: a review of available cases from the Research on Adverse Drug Events and reports (RADAR) project. J Am Coll Cardiol 47:175–181
Chevalier B, Silber S, Park SJ, Garcia E, Schuler G, Suryapranata H, Koolen J, Hauptmann KE, Wijns W, Morice MC, Carrie D, van Es GA, Nagai H, Detiege D, Paunovic D, Serruys PW, NOBORI 1 Clinical Investigators (2009) Randomized comparison of the Nobori Biolimus A9-eluting coronary stent with the Taxus Liberte paclitaxel-eluting coronary stent in patients with stenosis in native coronary arteries: the NOBORI 1 trial—phase 2. Circ Cardiovasc Interv 2:188–195
Nishimiya K, Matsumoto Y, Shindo T, Hanawa K, Hasebe Y, Tsuburaya R, Shiroto T, Takahashi J, Ito K, Ishibashi-Ueda H, Yasuda S, Shimokawa H (2015) Association of adventitial vasa vasorum and inflammation with coronary hyperconstriction after drug-eluting stent implantation in pigs in vivo. Circ J 79:1787–1798
Serruys PW, Farooq V, Kalesan B, de Vries T, Buszman P, Linke A, Ischinger T, Klauss V, Eberli F, Wijns W, Morice MC, Di Mario C, Corti R, Antoni D, Sohn HY, Eerdmans P, Rademaker-Havinga T, van Es GA, Meier B, Jüni P, Windecker S (2013) Improved safety and reduction in stent thrombosis associated with biodegradable polymer-based biolimus-eluting stents versus durable polymer-based sirolimus-eluting stents in patients with coronary artery disease: final 5-year report of the LEADERS (Limus Eluted From A Durable Versus ERodable Stent Coating) randomized, noninferiority trial. JACC Cardiovasc Interv 6:777–789
Natsuaki M, Kozuma K, Morimoto T, Kadota K, Muramatsu T, Nakagawa Y, Akasaka T, Igarashi K, Tanabe K, Morino Y, Ishikawa T, Nishikawa H, Awata M, Abe M, Okada H, Takatsu Y, Ogata N, Kimura K, Urasawa K, Tarutani Y, Shiode N, Kimura T (2013) Biodegradable polymer biolimus-eluting stent versus durable polymer everolimus-eluting stent: a randomized, controlled, noninferiority trial. J Am Coll Cardiol 62:181–190
Christiansen EH, Jensen LO, Thayssen P, Tilsted HH, Krusell LR, Hansen KN, Kaltoft A, Maeng M, Kristensen SD, Bøtker HE, Terkelsen CJ, Villadsen AB, Ravkilde J, Aarøe J, Madsen M, Thuesen L, Lassen JF (2013) Biolimus-eluting biodegradable polymer-coated stent versus durable polymer-coated sirolimus-eluting stent in unselected patients receiving percutaneous coronary intervention (SORT OUT V): a randomised non-inferiority trial. Lancet 381:661–669
Takabatake S, Hayashi K, Nakanishi C, Hao H, Sakata K, Kawashiri MA, Matsuda T, Yamagishi M (2014) Vascular endothelial growth factor-bound stents: application of in situ capture technology of circulating endothelial progenitor cells in porcine coronary model. J Interv Cardiol 27:63–72
Schwartz RS, Huber KC, Murphy JG, Edwards WD, Camrud AR, Vlietstra RE, Holmes DR (1992) Restenosis and the proportional neointimal response to coronary artery injury: results in a porcine model. J Am Coll Cardiol 19:267–274
Suzuki T, Kopia G, Hayashi S, Bailey LR, Llanos G, Wilensky R, Klugherz BD, Papandreou G, Narayan P, Leon MB, Yeung AC, Tio F, Tsao PS, Falotico R, Carter AJ (2001) Stent-based delivery of sirolimus reduces neointimal formation in a porcine coronary model. Circulation 104:1188–1193
Sakata K, Waseda K, Kume T, Otake H, Nakatani D, Yock PG, Fitzgerald PJ, Honda Y (2012) Impact of diabetes mellitus on vessel response in the drug-eluting stent era: pooled volumetric intravascular ultrasound analyses. Circ Cardiovasc Interv 5:763–771
Pearson JD (1994) Endothelial cell function and thrombosis. Baillieres Clin Haematol 7:441–452
Carter AJ, Laird JR, Farb A, Kufs W, Wortham DC, Virmani R (1994) Morphologic characteristics of lesion formation and time course of smooth muscle cell proliferation in a porcine proliferative restenosis model. J Am Coll Cardiol 24:1398–1405
Taylor AJ, Gorman PD, Kenwood B, Hudak C, Tashko G, Virmani R (2001) A comparison of four stent designs on arterial injury, cellular proliferation, neointima formation, and arterial dimensions in an experimental porcine model. Catheter Cardiovasc Interv 53:420–425
Farb A, Sangiorgi G, Carter AJ, Walley VM, Edwards WD, Schwartz RS, Virmani R (1999) Pathology of acute and chronic coronary stenting in humans. Circulation 99:44–52
Pendyala LK, Matsumoto D, Shinke T, Iwasaki T, Sugimoto R, Hou D, Chen JP, Singh J, King SB 3rd, Chronos N, Li J (2012) Nobori stent shows less vascular inflammation and early recovery of endothelial function compared with Cypher stent. JACC Cardiovasc Interv 5:436–444
Nakazawa G, Shinke T, Ijichi T, Matsumoto D, Otake H, Torii S, Hiranuma N, Ohsue T, Otsuka F, Shite J, Hirata K, Ikari Y (2014) Comparison of vascular response between durable and biodegradable polymer-based drug-eluting stents in a porcine coronary artery model. EuroIntervention 10:717–723
Hiranuma N, Shinke T, Nakazawa G, Otake H, Matsumoto D, Ijichi T, Kawamori H, Nagoshi R, Osue T, Shite J, Hirata K (2014) Optical coherence tomography and histopathology assessment after implantation of first- and second-generation drug-eluting stents in a porcine coronary model. Circ J 78(11):2665–2673
Tagawa S, Matsuda T, Aomizu T, Kuwana M, Ohtake H, Watanabe G, Yamagishi M (2014) Surface-bound vascular endothelial growth factor promotes prolonged activation of endothelial cells: a new technology for capturing endothelial progenitor cells by intravascular stents. J Tissue Sci Eng 5:1000140. doi:10.4172/2157-7552.1000140
Hagiwara H, Hiraishi Y, Terao H, Hirai T, Sakaoka A, Sasaki M, Murota S, Inoue K, Kimura J (2012) Vascular responses to a biodegradable polymer (polylactic acid) based biolimus A9-eluting stent in porcine models. EuroIntervention 8:743–751
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was in part supported by TERUMO.
Conflict of interest
Masakazu Yamagishi receives significant research funding from TERUMO. The other authors have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mori, M., Sakata, K., Nakanishi, C. et al. Early endothelialization associated with a biolimus A9 bioresorbable polymer stent in a porcine coronary model. Heart Vessels 32, 1244–1252 (2017). https://doi.org/10.1007/s00380-017-0992-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-017-0992-5