Abstract
Due to the rapid microbial degradation, ethylene (ethene; C2H4) detection in most soils is difficult. The knowledge about factors favoring the occurrence of C2H4 is mainly based on laboratory experiments which are not necessarily representative for real field conditions in forest soils. We report results from a total of 24 measuring plots in southwest Germany and more than 50,000 gas samples, including long-term forest monitoring sites and other study sites involving liming, artificial soil compaction, and temporary waterlogging. Many of the patterns and influencing factors identified in the laboratory were confirmed. In well-aerated forest soils, C2H4 was detected in 1.3% of the gas samples. C2H4 detection was higher at oxygen (O2) concentrations below 10% in the soil. The effect was positively correlated with bulk density in compacted soils and CO2, CH4, and N2O concentrations—factors that indicate reduced microbial activity and thus also facilitate the detection of C2H4. It could be detected 3.5 times more often in spruce than in beech stands, indicating that plant species plays a role and was not found to follow any distinct spatial or temporal pattern with the exception of seemingly random spatio-temporal clusters of root-born C2H4 near large roots of spruce trees. The long observation period and large dataset of this field study allowed valuable insights into C2H4 occurrence in forest soils under natural conditions supporting the theory that the plant hormone C2H4 is produced as a warning for anaerobic soil areas that are limiting root growth.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most of the studies on soil gases in forest soils focus on soil respiration and other greenhouse gases. However, other trace gases can also provide indicators for important soil ecological processes and point to stress-related climate consequences (Conrad 1996). In plant physiology, the gaseous plant hormone C2H4 plays an important role throughout the whole life cycle in plants, from seedling to mature plants (Bakshi et al. 2015; Sisler and Yang 1984). Being associated with the ripening process (Reid 1995), C2H4 can induce abscission (enhanced dropping of leaves, flowers, and fruits) or senescence (accelerated aging of the plant). Plants produce C2H4 (Sisler and Yang 1984), whereas microorganisms and fungi can produce and degrade it (Fukuda et al. 1993; Primrose 1979). If C2H4 concentrations in the soil gas phase exceed certain thresholds, plants will be negatively influenced. Even though threshold values depend on plant species, C2H4 concentrations >0.01 μmol mol−1 were identified to already have effects on plant growth, whereas C2H4 concentrations >10 μmol mol−1 cause severe plant damages (Primrose 1979; Smith and Russell 1969). C2H4 induces distinct changes in plant growth and morphology known as “triple response” (Binder 2020; Bleecker and Kende 2000; Zhong et al. 2014). These responses collectively enable plants to adapt to stressful conditions caused by mechanical obstacles or anaerobic soil atmosphere (Abeles et al. 1992).
In soils, the occurrence and concentration of C2H4 are affected by several physical and chemical soil properties. Smith and Dowdell (1974) list soil temperature, oxygen (O2) availability, and soil water content (SWC) as most important factors, which alter the production and diffusion of C2H4 in and out of soil. These factors and additionally soil organic matter content (SOM) and soil pH (Otani and Ae 1993), change the rhizosphere microbial composition influencing C2H4 production dynamics. Roots can produce C2H4 under low-oxygen conditions, such as waterlogged or flooded soil since oxygen deprivation activates specific enzymes involved in C2H4 biosynthesis, leading to increased ethylene production (Drew 1997) and C2H4 degradation is inhibited (Smith and Restall 1971). This response helps plants cope with oxygen-deficient conditions. Physical damage or mechanical stress to roots, such as compaction, root displacement, and contact with stones can also stimulate C2H4 production (Mattoo et al. 1991). This also results in a positive correlation of soil bulk density (BD) to C2H4 detection (Danish et al. 2020; Smith and Restall 1971). In response to mechanical stress, C2H4 production helps coordinate root growth and morphology, allowing the plant to navigate and adapt to its environment. C2H4 production varies throughout the different developmental stages of roots (Negi et al. 2008). Further positive correlations with C2H4 occurrence were found to SWC (Dowdell et al. 1972), soil temperature (Xu and Inubushi 2009), and CO2 concentration (Cornforth 1975). However, when related to SWC, increased C2H4 production was also observed during drought stress contributing to the regulation of plant growth (Sharp et al. 2000). On the contrary, other effects like tree type (Elsgaard 2001), OM content (Cornforth 1975; Goodlass and Smith 1978), and soil pH (Lindberg et al. 1979; Zechmeister-Boltenstern and Smith 1998) on C2H4 are not consistent between studies.
The C2H4 is a particularly reactive gas, normally consumed directly in the vicinity of its production and for this reason concentrations are generally low and accumulation of C2H4 occurs mostly in hot spots, making detection particularly difficult. Apart from incubation experiments, the detection of C2H4 in the field depends not only on the amount produced but also on the conditions that favor the accumulation of the trace gas in the soil and delay its degradation.
Dowdell et al. (1972) conducted a field study to investigate C2H4 occurrence in natural forest soils. Hunt et al. (1981), Meek et al. (1983), Otani and Ae (1993), and Smith and Dowdell (1974) conducted field studies with additional treatments of soil samples in the laboratory. Yet, a comprehensive field investigation of C2H4 occurrence in soil has not been conducted. One reason for the paucity of (field) studies is the rare and accidental occurrence of C2H4 in non-compacted, well-aerated soils which mainly act as a net sink of C2H4 (Abeles et al. 1971; Arshad and Frankenberger 1990; Hendrickson 1989). The rare and accidental occurrence of C2H4 challenges the concept of short-term (<4 years) studies and requires long-term data series to get reliable results of C2H4 occurrence and contribute to a better understanding of C2H4 influencing factors in forest soils.
Our study will report on field observations of C2H4 occurrence and quantity in soil gas phase at forest sites. Our objectives were to quantify the occurrence of C2H4 in well-aerated forest soils with different tree species, artificially compacted soil, waterlogged forest sites, limed soils and to identify the influencing factors. The investigated soils provide properties which have so far been identified primarily in laboratory experiments as potential factors influencing C2H4 production, C2H4 degradation and transport in the soil ecosystem.
Material and methods
Study sites and plots
Data from 24 study plots set up at 14 sites in southwest Germany (Fig. 1) were used, originating from long-term soil gas monitoring (n = 13), and project studies (n = 11) on liming and soil compaction including in total 50,922 analyzed soil gas samples. The large number of plots allows us to investigate the effect of various soils, treatments, and stands on ethylene occurrence (Fig. 2).
The plots have a mean annual temperature between 7.4 and 9.6 °C and a mean annual precipitation total of 700 to 1385 mm. They are located at altitudes ranging from 172 to 816 m a.s.l. The acid topsoil (0-5 cm) had pH (KCl) values ranging from 2.9 to 4.1 and in humus from 2.6 to 5.0. Sites on carbonate rock or with clay texture are not included in the study. Each site has at least one plot area with one adjustment of multiple passive soil gas samplers.
Long-term environmental monitoring plots (LTEMPs)
Soil gas data of 13 plots at 6 different sites (Table 1) originate from long-term environmental monitoring plots (LTEMP) which are part of the International Cooperation Program Forests (ICP Forests). Five of the six sites have an adjacent beech plot (Fagus sylvatica L.) to the spruce plot (Picea abies L.), and two of the sites have additional liming plots. More detailed information on the long-term soil gas monitoring concept is presented by Maier et al. (2020). During establishment, permanent soil gas sampling positions were mapped with neighboring roots and trees, allowing for subsequent identification of root-influenced gas samplers. In addition to the general site characteristics (Table 1), monitoring data on soil temperature, soil water content, as well as climate and soil gas diffusivity are available. The discretization of the SWC was modeled with LWF Brook 90 (Schmidt-Walter et al. 2020), and the soil temperature was modeled in Hydrus 1D (Maier et al. 2020; Simunek et al. 2005)
Project studies: soil compaction studies
Soil compaction studies were conducted at SM (Horn et al. 2007) and MU (Schäffer 2012) to investigate the effect of artificial soil compaction on soil functions. In these studies, the soil was compacted by typical forestry operations (e.g., operation of heavy forestry machinery). Soil gas was sampled within, and next to, wheel tracks as well as in nearby, non-compacted control areas as reference (Fig. 7). In WW and FO, the soil compacted by heavy machinery was afforested.
The soil at the ST site (Schack-Kirchner and Hildebrand 1998) was also compacted by heavy machinery. However, after 4 years, when soil gas was sampled, surficial signs of deformation were no longer detected. Additionally, temporary waterlogging and O2 deficiency were evident in the appearance of distinct hydromorphic features, typical for a Stagnosol, which is why the ST site was also assigned to the compaction study. General soil gas data (e.g., greenhouse gas fluxes) were already discussed in former publications (Schack-Kirchner and Hildebrand 1998; Schäffer 2012; Schäffer and Von Wilpert 2004), but C2H4 was largely omitted.
Project studies: liming trials
The study sites of the liming trials (BW, HW, HO) had previously been subject to intensive analysis after liming, which showed increased soil pH values and enhanced base saturation in limed soils (Jansone et al. 2020) compared to a corresponding unlimed reference plot nearby. Soil gas data should reveal a changing diffusivity of the soil due to liming.
Sampling and analyzing soil gas samples
The measurement results were obtained from all plots via the analysis of soil gas samples that were collected at regular intervals. To collect soil gas, passive soil gas samplers were used according to methods described by Schack-Kirchner et al. (1993) (Fig. 8). An artificially perforated steel tube was placed in the soil, acting as a macropore and, thus, allowing diffusive gas transport via a cannula into a gas vial (4 ml) initially filled with helium. Although all gas samples were collected and analyzed in the same way, there is no uniform experimental design for the plots as our study is only an evaluation of existing data sets. As a consequence, the definition of the measurement depths was done according to the objectives of the study at that time. In well-aerated forest soils (LTEMP and liming trials), the focus was on microbial active topsoil, so gas samplers were only installed in humus (+3/+2 cm) and at depths of 0 cm (at the humus/mineral soil interface), −5 cm, and −10 cm. At each depth level, there are at least three (though typically five) replicates resulting in about 20 soil gas samplers on each of the plots. For the compaction study plots, the focus shifted to deeper soil layers. In order to capture the totality of soil changes in these plots, gas samplers were located at −15 cm, −20 cm, −25 cm, −30 cm, and −35 cm (Table 2). The exact position and number of gas samplers here can be seen in Fig. 7.
As gas exchange between soil pore space and the gas vial is passive, the gas composition in the vials represents the average soil gas atmosphere of the most recent hours; however, it is possible for the vials to still contain components of the soil gas atmosphere of the previous day if high SWC slowed down the gas exchange. Similarly, if the cannula is clogged or there is water in the perforated steel tube of the samplers, the gas exchange with the soil gas phase cannot be completed. During the subsequent determination of the soil gas composition, a remaining helium content is then determined. These vials were then eliminated from further evaluation.
The gas vials were collected in a regular and at least 4-week cycle, and the soil gas composition (N2, O2, Ar, CO2, CH4, C2H4, N2O) was immediately analyzed in the laboratory with a gas chromatograph (GC) Clarus 680 GC (Perkin Elmer, Waltham, MA, US). In the GC, C2H4 was separated by a CP-SilicaPLOT column (Agilent Technologies) and determined by a flame ionization detector (FID). The respective GC system that was used did change over the years; however, the configuration of the GC system remained the same (more details in Maier et al. (2020)). The limit of quantification (LOQ) for C2H4 measurements was 0.12 μmol mol−1 (10 × standard deviation (SD) of C2H4 scattering of the calibration gas), and the limit of detection (LOD) was 0.036 μmolmol−1 (3 × SD). LOD and LOQ values were validated by mixing and testing the signal to noise ratio of an artificial air mix, with C2H4 concentrations two times higher than the LOQ.
Statistical analysis
The C2H4 concentration dataset is left-censored and contains a high proportion of true-zero measurements (96%). Since a proper approach could not be found in related studies dealing with true-zero and left-censored data simultaneously (Brown 2008; Guideline 2005; Proctor 2008), an in-house procedure was developed based on Blackwood (1991), Helsel (2011), and Zuur et al. (2009), who developed their methodology mainly for the evaluation of the most prominently censored datasets from survival studies. Using the in-house methodology, 22% of values >0 μmol mol−1 were below LOD and 58% were below LOQ. Concentrations below LOD or LOQ were not excluded from analysis or substituted by artificial values in order to avoid artificial assumptions or bias that could lead to incorrect results or blur information contained in the measurements (Brown 2008; Helsel 2011). Statistical analyses were conducted using the open-access software R (version 4.0.3; R Development Core Team ), with a significance level set to 5%.
For an initial assessment, the total data set was divided into multiple classes. Firstly, data was classified as “C2H4 detection” (C2H4 >0 μmol mol−1) or “no C2H4 detection” (C2H4 = 0 μmol mol−1), which allowed for the determination of promoting and inhibiting factors on C2H4 occurrence. To check for significant differences, the non-parametric Wilcoxon test (also known as Mann–Whitney-U-test) was conducted. For non-parametric Spearman correlation analysis, C2H4 values >0 μmol mol−1 and <LOD were set to 0.02 μmol mol−1 and values ≥LOD and <LOQ to 0.07 μmol mol−1. As suggested by Helsel (2011)values between different measurement limits were set to equal numbers respectively enables to treat data as the same ranks with a degradation of values <LOD (lower) and <LOQ (higher).
The dataset was further divided into five classes: “0 μmol mol−1,” “>0 μmol mol−1 <LOD,” “≥ LOD < LOQ,” “≥ LOQ < 1 μmol mol−1,” and “>1 μmol mol−1”. To identify environmental influencing factors such as air temperature or precipitation amount, a Kruskal–Wallis test, as a non-parametric correspondent to an ANOVA, was conducted. If a significant difference was detected, Dunn’s test with Bonferroni correction was run to identify groups responsible for difference.
The maximum likelihood estimate (MLE) approach was used to conduct summary statistics, which performed well for our data when excluding zero values and with a censored data share between 50 and 80% (Helsel 2011). In order to apply the MLE approach, the data were tested for lognormal distribution. The implementation of MLE in R utilizes the NADA packages developed to handle censored data. As evaluation with NADA allows the use of only one reporting limit, LOD was selected, and concentrations ≥LOD were, thus, treated as absolute values, knowing that values between LOD and LOQ cannot be determined correctly in concentrations. For checking differences of C2H4 concentrations between groups, a censored data adapted t-test for 2-factor variables and a censored data adjusted ANOVA for > 2-factor variables were conducted. The equivalent of both tests in R is censored regression. If there are no more censored data in partial data sets (e.g., ST), the MLE approach or censored regression was no longer necessary.
To identify correlations to other soil gases for the overall data set, a classification approach of logistic regression was used (Nwanganga and Chapple 2020). The graphical result is an s-curve, representing the range of an event’s occurrence probability between 0 and 1 (Nwanganga and Chapple 2020). For quantifying the effect of a predictor variable, an odds ratio is used. It represents the probability that an event occurs relative to the probability of the event’s non-occurrence (Peng et al. 2002). The higher the odds ratio, the higher the influence of a predictor variable. The model is validated by a chi-squared <0.05 and R2. With test and training data sets, and accuracy of predicted probabilities, it can further be stated whether the fitted model leads to reasonable results (Nwanganga and Chapple 2020).
Results
Long-term environmental monitoring plots
LTEMPs data represent 80% of the samples. C2H4 was detected at least once on every LTMEP. In proportion to the total number of gas samples, C2H4 was detected most frequently at ESS (2.55%) and least frequently at ESB (0.07%) (Table 6). Analysis of all 13 LTEMPs combined shows an average C2H4 detection frequency of 1.3%. The range of measured C2H4 concentrations is from <LOD (0.01 μmol mol−1) to 23.9 μmol mol−1, with a mean value of 0.78 μmol mol−1. Six out of 13 plots showed median C2H4 concentrations <LOQ.
Censored regression showed a significant depth gradient in mineral soil, with decreasing C2H4 concentration as depth increases (Table 3). However, when root-induced C2H4 release (mainly registered in the humus layer at ESs) is excluded, the differences between C2H4 concentrations in the mineral soil and humus layer were no longer significant (“(excl.)” line in Table 3).
During the establishment of the plots in 1998, field maps were made in which the position of samplers and also strong roots in the near environment (<1 m distance) were registered. At these three plots (ESS, COS, ASS), episodes of clustered ethylene detection were observed at the gas samplers located closest to the roots (Fig. 9). Therefore, root influence is clearly influencing C2H4 hot spots and is the most important factor influencing spatial patterns.
When analyzing the temporal evolution of C2H4 occurrence (Fig. 9), most years show no, or minimal C2H4 occurrence frequency but, overall, single intensive periods of C2H4 release. The dataset divided into classes was compared to different environmental parameter values (e.g., air temperature, SWC, precipitation) by conducting a Kruskal–Wallis test. The analysis was site specific. Overall, an influence of the climatic parameters on the ethylene occurrence showed no significance when considering the total time series. Trends are recognizable for some short-term periods, but when considering different locations at the same time, or the full time series, the trends are no longer present or contrary. Consequently, general conclusions could not be drawn regarding temperature or moisture being the main influencing factors for the temporal variability of many other soil gases.
To identify the tree-species effect, only the period of time from 2010 onward, when samples were taken on adjacent LTEMPs of different forest stands, was considered. At spruce sites, C2H4 was detected in 1.17% of the samples, with simultaneously elevated concentrations (Fig. 3), which represents a significant difference (Spearman’s rho: −0.110; p < 0.05) compared to the samples of beech sites, which had a C2H4 detection frequency of only 0.33% as well as lower C2H4 concentrations. This effect is also visible in a pairwise comparison of neighboring spruce and beech plots at the same site (Fig. 10). Low frequencies of occurrence on some plots, however, permitted statistical analysis of the pairwise comparison.
For a quantitative analysis of the effect of soil texture on C2H4 occurrence, the LTEMPs dataset was classified according to the sites’ main soil texture group (Table 1). The frequency of C2H4 occurrence was slightly higher in sandy soils (1.36% compared to 1.04% in silty soils). Median and mean C2H4 concentrations were higher in silty soils (median: 0.14 μmol mol−1) than in sandy soils (median: 0.05 μmol mol−1) and the difference was found to be significant (p < 0.05; censored regression). It must be noted that no clayey and less-sandy soils were included. Total pore space (TPS), air-filled pore space (AFPS), and bulk density (BD) showed no significant effect when examining LTEMPs’ soils.
Liming studies
The effect of liming was studied based on LTEMPs, including limed and non-limed reference plots in HD and OC. An influence of soil pH or carbonate content could not be identified for either the humus or the mineral soil. C2H4 occurred slightly less (not significant) on limed plots (1.30%) than on non-limed plots (1.41%) when the areas of both sites are evaluated together. The difference between C2H4 concentrations caused by liming was significant in censored regression analysis (p < 0.05), with C2H4 concentrations being slightly lower on limed plots (median: 0.04 μmol mol−1; mean: 0.13 μmol mol−1) compared to non-limed plots (median: 0.06 μmol mol−1; mean: 0.40 μmol mol−1). The three sites of the liming study (BW, HO, and HW), with a duration of 1.5 years, did not show sufficient C2H4 detection for statistical analysis, since C2H4 could only be measured in 5 of almost 2000 samples.
Compaction studies
Considering all five compaction study sites together (SM, MU, WW, FO, ST), C2H4 detection frequency was 11.24%—almost ten times higher than the detection frequency at the LTEMPs (Table 6). To investigate the compaction effect, the individual gas samplers were divided into “compacted” and “non-compacted” according to their position and the bulk density of soil samples collected there (Table 4).
C2H4 shows significantly higher (p < 0.001; censored regression) occurrence (Fig. 11) and concentrations in compacted soils (14.75%, median: 0.41 μmol mol−1) than in non-compacted soils nearby (6.95%, median: 0.12 μmol mol−1) (Fig. 4). There is also a positive correlation between C2H4 occurrence and bulk density (p < 0.05; Wilcoxon test), with samples showing C2H4 concentrations > 0 μmol mol−1 having higher BD values (1.53 g cm−3) than samples without C2H4 detection (1.44 g cm−3).
C2H4 concentration (μmol mol−1, logarithmic transformation–log10) in non-compacted and artificially compacted soils (left) of the compaction study sites. Data of FO were excluded since information of compaction status is missing. LOD and LOQ are represented by the dashed lines and mean, median, and SD were calculated with the MLE approach. O2 (middle) and CO2 (right) concentrations (mol mol−1) in samples with and without C2H4 detection. Differences in concentration are significant. The notches of the boxplots representing the 95% confidence interval in samples with C2H4 detection extend beyond the 25th percentile due to small sample size (n = 17). It shows the uncertainty of the true median
SM and MU showed the highest C2H4 occurrence right after the artificial compaction of the forest soils in 2001 (20.37%)/2002 (27.53%) and in 2009 (0.91%), respectively. In the following years, the number of C2H4 measurements decreased to zero. At WW and FO, the analysis of temporal patterns was difficult due to short measurement time as well as large temporal gaps (Table 1). C2H4 occurred preferentially in the summer months from April to August.
To examine the depth gradient in compacted soils, ethylene results were pooled by study site and depth (Table 3), considering only gas samplers in compacted soils (Table 4). As the WW site was only sampled at the −30 cm depth level, these data are for comparison only. The heaviest machine in the soil compaction study at SM caused the most frequent C2H4 detection (Table 3) and the highest C2H4 concentrations underneath the tracks. This effect also emerged within the SM plots, where different driving operations were carried out, resulting in an effect of machine weight and track position on ethylene detection frequency (Table 5 and Fig. 12).
Contrary to the results of LTEMPs, all compacted plots where soil gas was sampled at different soil depths (SM, MU, and FO in Table 3) show rising C2H4 occurrence frequencies with increasing depth. C2H4 concentrations in mineral soil (median: 0.09 μmol mol−1; mean: 0.39 μmol mol−1) differ significantly from C2H4 concentrations in humus (median: 0.03 μmol mol−1; mean: 0.06 μmol mol−1; p < 0.001; censored regression). However, there are large variabilities in C2H4 occurrence frequencies and concentrations between the study sites (Table 3). When examining a depth gradient, it must be noted that the sampling depths are not uniform.
Temporary waterlogged site
Because of the predominant Stagnosol soil type, and the fact that the temporary water influence still overlaps the effects of compaction, the ST site was analyzed separately from the other compaction studies. This fact is also reflected in the highest C2H4 detection frequency for ST, with a C2H4 detection frequency of 26.98% (Table 6), which was also the highest of all 24 monitoring plots. All C2H4 measurements exceeded the LOD (Fig. 4), and therefore, the MLE approach was not applied.
C2H4 correlated with O2 (Spearman’s rho: −0.345; p < 0.05) and CO2 (Spearman’s rho: 0.284; p < 0.05). Both gases differ significantly in samples with and without C2H4 detection (p < 0.05; Wilcoxon test). Anaerobic soils with low oxygen concentrations and correspondingly elevated CO2 levels showed higher occurrence and concentrations of C2H4 (Fig. 4).
Combined analysis of all plots
Using the dataset of all 24 study plots, we could show that C2H4 was positively correlated (p < 0.001; Wilcoxon test) with CO2, CH4, and N2O. Concentrations of the latter gas species are not only different in samples with and without C2H4 detection but also changes with rising C2H4 concentrations if only samples with > 0 μmol mol−1 C2H4 are considered (Fig. 5) when tested with the Spearman correlation. Quantitatively, a higher C2H4 concentration is detected in samples of higher CO2, CH4, and N2O concentrations. Above a CO2 concentration of 0.3 mol mol−1, the C2H4 detection frequency seems to rise (Fig. 5). There is a significant positive correlation (p < 0.001) between CO2 and C2H4 detection frequency if all data are included (Spearman’s rho: 0.111) and if only C2H4 concentrations > 0 μmol mol−1 are included (Spearman’s rho: 0.391). Additionally, there is a 1.18 times higher chance of detecting C2H4 if CO2 concentration rises by 1 mol mol−1 (odds ratio). With logistic regression, it is shown that CO2 concentration must be >21.05 mol mol−1 to detect C2H4 by a probability >50%, which is hardly reached in our upland forest soils. The Chi2 for the test model was <0.05 and R2 was 0.2. Furthermore, the correlation of CH4 and N2O concentrations to C2H4 detection frequency was significantly positive (p < 0.001) for the whole dataset (Spearman’s rho: 0.172 (CH4) and 0.077 (N2O)) and for C2H4 measurements >0 μmol mol−1 (Spearman’s rho: 0.172 (CH4) and 0.105 (N2O)). The analysis of all monitoring plots combined was only conducted for the gas composition, as plot specific factors, such as compaction or soil type, overlay the singular influence of individual site parameters.
C2H4 detection frequency depending on the CO2 (a, mol mol−1) and CH4 (c, μmol mol−1) and N2O (d, μmol mol−1) concentration in soil gas divided into classes and the graphical visualization of the logistic regression of C2H4 occurrence with CO2 concentration (b, mol mol-1). Data of all 24 study plots were considered
Discussion
Temporal and spatial occurrences
C2H4 field measurements showed that C2H4 could potentially be measured at all sites if the observation time is sufficiently long. This confirms the assumption that C2H4 is always produced in soil even under aerobic conditions (Jäckel et al. 2004). Furthermore, it suggests that the local C2H4 degradation rate is far higher than the C2H4 production rate in soils with high O2 concentration, so much so that C2H4 emitted by any source will be directly consumed in the vicinity of the source (Hendrickson 1989). Well-aerated soils, therefore, serve primarily as a sink for C2H4 (Arshad and Frankenberger 1990). Our study confirms that C2H4 occurrence is characterized by a high spatial variability, affirming the results of the only pure field study on C2H4 conducted by Dowdell et al. (1972). The high C2H4 concentrations observed in the laboratory study by Smith and Russell (1969) are far from the mean C2H4 occurrence in 1.3% of the gas samples observed in well-aerated forest soils of our study. Our findings on factors influencing ethylene occurrence are summarized in the diagram Fig. 6. Significantly, more C2H4 can be observed in forest ecosystems with soil compaction, and this is further increased by the influence of waterlogging in soils. Concerning the depth distribution of C2H4, no clear pattern could be identified. While the C2H4 occurrence at the LTEMPs decreased with soil depth, the compaction study sites showed a higher ethylene occurrence frequency in deeper soil layers. The only clear spatial pattern of C2H4 was the enhanced C2H4 detection around roots, owing to C2H4 release by forest tree roots.
The episodes of increased ethylene detection in the near vicinity of woody roots, which often occurred many years after installation of the samplers, do not have to be caused by root-related C2H4 production. Increasing radial growth of the roots can compact the soil, too, resulting in increased C2H4 release due to local soil compaction. Fixed measurement installation positions prevent a clear understanding about the current root penetration of the surrounding forest soil.
Oxygen effect and the correlation with other gas compositions
Our analysis confirms that low oxygen concentration correlated negatively with C2H4 occurrence at the temporary waterlogged Stagnosol site (Fig. 6), with an average O2 concentration in soils with C2H4 detection of about 6%. The oxygen effect, could not be observed in the total data set, as 80% of the gas samples originate from well-aerated forest soils with O2 concentration >18%. Anaerobic conditions are considered to be the most important factor influencing the increased C2H4 production in soils (Elsgaard 2001; Primrose 1979; Xu and Inubushi 2009; Zechmeister-Boltenstern and Nikodim 1999). It is assumed that the accumulation of C2H4 results from the suppression of the aerobic process of microbial decomposition of C2H4 (Arshad and Frankenberger 1990; Cornforth 1975; Smith and Restall 1971). The O2 concentration must be very low to have an influence on C2H4 (Hunt et al. 1981; Meek et al. 1983; Smith and Restall 1971).
Besides the oxygen effect, we found a significant positive correlation of C2H4 with CO2, CH4, and N2O (Fig. 6). The positive correlations with these trace gases indirectly confirm the findings of low O2 concentrations leading to C2H4 accumulation, as the increase in CO2, CH4, and N2O indicates a reduced gas exchange (Pandey et al. 2021). Consequently, as the gas exchange is limited, this also leads to an accumulation of C2H4. High CO2 concentrations of more than 1 to 2% indicate poor soil aeration, which are typically detected in compacted soils (Schack-Kirchner and Hildebrand 1998; Schäffer 2012), whereas, in our field study, the effect of a more frequent C2H4 detection could already be observed at much smaller CO2 concentrations. C2H4 was preferentially detected in the subsoil of the compaction study plots, an outcome we interpret again as an effect due to low O2 concentrations.
Our observation of methane being positively correlated with C2H4 is in line with results of Jäckel et al. (2004) and Xu et al. (2008), who explained this by the inhibition of methane-oxidizing bacteria in an environment of C2H4 accumulation. Furthermore, methanogenic microbes are more likely found in anaerobic soils (Conrad 1996), being favorable for C2H4 accumulation. Similarly, N2O concentrations are positively correlated with C2H4 occurrence and concentration, as N2O is formed under suboxic conditions during denitrification (Bremner and Blackmer 1978), which are favorable conditions for C2H4 evolution as well.
Compaction effect
Although LTEMPs did not show any effect of BD on C2H4 occurrence, data of the compaction study sites clearly did (Fig. 6). The rather low BD in the topsoil of the LTMEPs prevents the accumulation of ethylene; thus, the effect of BD does not appear on these plots. Soil compaction leads to a twofold increase in the frequency of C2H4 occurrence (7% to 15%) and significantly higher C2H4 concentrations compared to reference plots (Fig. 11). This effect can be explained by a reduction of the gas diffusion in soils as TPS, and more importantly, AFPS are reduced (Pandey et al. 2021). Considering the study results of Pandey et al. (2021) on plant growth, this could confirm the theory that the release of the plant hormone C2H4 is a warning signal to prevent root growth into compacted soil areas.
Tree-type effect
Due to the common observation time of more than 10 years, a clear tree-type effect, with 3.5 times more frequent C2H4 detection under spruce than under beech, could be observed. Studies on C2H4 in coniferous and deciduous forest soils already indicated that C2H4 degradation is faster in deciduous forest soils (Zechmeister-Boltenstern and Smith 1998), whereas coniferous forest soils show a higher probability of C2H4 accumulation, especially under wet conditions (Rigler and Zechmeister-Boltenstern 1999; Sexstone and Mains 1990), which lead to contrary results on the tree type effect (Fig. 6).
Reasons for the observed tree species effect could be the combination of gas measurements being only down to 10 cm depth and differences in rooting. Whereas spruce consists of a shallow root system, beech is a deep-rooted tree (Schmid and Kazda 2005). Spruce stands therefore show an enhanced detected root release of C2H4 in the upper soil areas, as root density decreases exponentially from the upper to the lower soil layers (Schmid and Kazda 2001). This is validated by the fact that only spruce plots show patterns of C2H4 root release, which could be identified as the main source of C2H4 in soil. Roots are thus the most important factor influencing spatial heterogeneity of C2H4 in soil, followed by microbial C2H4 production (Hendrickson 1989).
The tree species effect was significant using the entire LTEPM dataset (Fig. 3) but also evident in a pairwise comparison of neighboring beech and spruce plots (Fig. 10). However, the observed root density at our plots was surprisingly the same and partly even lower on spruce sites compared to beech sites. An increased rooting of the spruce sites in the topsoil could therefore not be proven (Fig. 10). However, measuring root densities by counting root tips in soil profiles includes a high inherent uncertainty.
More obvious is a microbial component/contribution of the tree species effect. Arbuscular mycorrhizal fungi (AMF) communities are different for tree species (Rożek et al. 2020). AMF on spruce roots could be specialized in C2H4 production, whereas AMF on beech roots adapted to C2H4 degradation. Yet this was not tested, nor is there clear evidence of this in the literature. Finally, it was shown that soils under beech and spruce differ in CO2 respiration rate (Berger et al. 2010), resulting in the assumption that beech sites have higher C2H4 oxidation rates than spruce sites. In combination with the observed higher SOM-content, identified as influential on the C2H4 occurrence among others by Goodlass and Smith (1978), beech sites may provide a more microbial activity in soil (Achilles et al. 2021), leading to rapid degradation of C2H4 by microorganisms in the soil and complicating detection. The different characteristics of soils under beech and spruce were no object in our study.
Additional physical and chemical soil parameters
For pH, SOM-content, and liming, no effect on C2H4 could be detected (Fig. 6). This coincides with the result of the study conducted by Lindberg et al. (1979). Findings of laboratory studies in pH having a negative correlation with C2H4 concentrations (Arshad and Frankenberger Jr 1991; Zechmeister-Boltenstern and Smith 1998; Zechmeister-Boltenstern and Nikodim 1999) were not confirmed in our study. This is probably because all our sites are acidic, and soil pH values do not differ substantially between the monitoring plots. Furthermore, there are probably other relevant site characteristics having an impact on C2H4 that possibly mask the effect of soil pH. Unfortunately, the analysis of the data of our liming plots did not yield reliable results, since the overall occurrence of CH4 was too low, or the monitoring period was too short.
Analysis of air and soil temperature effects on C2H4 at the LTEMPs led to ambiguous results. Reported heat activation by microorganisms producing C2H4 (Lynch 1975; Sexstone and Mains 1990; Smith and Cook 1974; Smith and Dowdell 1974; Smith and Restall 1971; Xu and Inubushi 2009) could only be partially detected due to a partial preferential occurrence in the summer months in the data of the project studies. Typical repeated temporal or even seasonal pattern could not be detected. The impact of precipitation and SWC on C2H4, by contrast, seems to confirm the findings of other studies (Dowdell et al. 1972; Hunt et al. 1981; Lindberg et al. 1979; Otani and Ae 1993; Zechmeister-Boltenstern and Nikodim 1999). C2H4 measurements >0 μmol mol−1 were predominately observed after rainfall, which can be explained by a reduced O2 concentration in wet soils; thus, lower decomposing rates of C2H4 (Sawada et al. 1985; Zechmeister-Boltenstern and Smith 1998), as well as a reduced gas diffusion in the air-filled soil pore space (Lynch 1975).
Since sandy soils showed a higher C2H4 detection frequency than silty soils, but C2H4 concentrations were higher in silty soils, there is a contrary effect of soil texture on C2H4. Results of the study of Van Cleemput et al. (1983), stating that C2H4 preferentially occurs in sandy soils, can thus only be partly validated. It must be noted that clayey soils were not present in our study.
Conclusion
The analysis of our extensive dataset (more than 50,000 from 1994 to 20201 on 24 plots) showed that the detection of C2H4 is extremely infrequent in forest soils, and, except for sampling gas next to main roots, it does not exhibit distinct temporal and spatial patterns. The investigation of the temporal and spatial distributions did not show any systematic or seasonal patterns. Therefore, a direct influence of environmental parameters, such as soil temperature and moisture, could not be observed. Nevertheless, C2H4 does not occur randomly in soils. A cluster structure with hot moments (C2H4-rich years) and hot spots (C2H4-rich plots) is evident, though thus far it cannot be explained. The most important soil-related and exogenous factors influencing C2H4 occurrence are soil compaction and high water contents, leading to low O2 and elevated CO2 concentrations in the soil. Both factors inhibit microbial activity, allowing accumulation of produced C2H4 in anaerobic soils. Furthermore, the tree species had a clear effect on C2H4. Soils under spruce show an increased C2H4 occurrence than soils under beech. A reason for this might be the different root system or higher microbial activity in beech stands, which leads to faster degradation of C2H4 and reduces the C2H4 detection frequency. In well-aerated upland forest soils, in turn, high oxygen levels usually prevail, and an unhindered gas exchange can take place. This leads to the rapid diffusion and degradation of C2H4 with low probability of C2H4 accumulation in soil. Furthermore, the study presents a statistical approach for the challenging evaluation of left-censored data sets with a high number of zero measurements, which could provide reliable results about the factors influencing C2H4 occurrence even in well-aerated forest soils. Although the scope of our study on ethylene occurrence is unique, the importance of long term monitoring programs must be emphasized. Each additional measurement facilitates the statistical analysis and identification of natural influencing factors.
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article or its supplementary materials.
References
Abeles F, Craker L, Forrence L, Leather G (1971) Fate of air pollutants: removal of ethylene, sulfur dioxide, and nitrogen dioxide by soil. Science 173:914–916
Abeles FB, Morgan PW, Saltveit ME Jr (1992) Ethylene in plant biology, 2nd edn. Academic Press, New York
Achilles F, Tischer A, Bernhardt-Römermann M, Heinze M, Reinhardt F, Makeschin F, Michalzik B (2021) European beech leads to more bioactive humus forms but stronger mineral soil acidification as Norway spruce and Scots pine–Results of a repeated site assessment after 63 and 82 years of forest conversion in Central Germany. For Ecol Manag 483:118769
Arshad M, Frankenberger W Jr (1991) Effects of soil properties and trace elements on ethylene production in soils. Soil Sci 151:377–386
Arshad M, Frankenberger W (1990) Production and stability of ethylene in soil. Biol Fertil Soils 10:29–34
Bakshi A, Shemansky JM, Chang C, Binder BM (2015) History of research on the plant hormone ethylene. J. Plant Growth Regul 34:809–827
Berger TW, Inselsbacher E, Zechmeister-Boltenstern S (2010) Carbon dioxide emissions of soils under pure and mixed stands of beech and spruce, affected by decomposing foliage litter mixtures. Soil Biol Biochem 42:986–997
Binder BM (2020) Ethylene signaling in plants. J Biol Chem 295:7710–7725
Blackwood LG (1991) Analyzing censored environmental data using survival analysis: single sample techniques. Environ Monit Assess 18:25–40
Bleecker AB, Kende H (2000) Ethylene: a gaseous signal molecule in plants. Annu Rev Cell Dev Biol 16:1–18
Bremner J, Blackmer AM (1978) Nitrous oxide: emission from soils during nitrification of fertilizer nitrogen. Science 199:295–296
Brown RJ (2008) The use and abuse of limits of detection in environmental analytical chemistry. Sci World J 8:796–801
Conrad R (1996) Soil microorganisms as controllers of atmospheric trace gases (H2, CO, CH4, OCS, N2O, and NO). Microbiol Rev 60:609–640
Cornforth I (1975) The persistence of ethylene in aerobic soils. Plant Soil 42:85–96
Danish S, Zafar-Ul-Hye M, Fahad S, Saud S, Brtnicky M, Hammerschmiedt T, Datta R (2020) Drought stress alleviation by ACC deaminase producing Achromobacter xylosoxidans and Enterobacter cloacae, with and without timber waste biochar in maize. Sustainability 12:6286
Dowdell R, Smith K, Crees R, Restall S (1972) Field studies of ethylene in the soil atmosphere-equipment and preliminary results. Soil Biol. Biochem. 4:325–331
Drew MC (1997) Oxygen deficiency and root metabolism: injury and acclimation under hypoxia and anoxia. Annu Rev Plant Biol 48:223–250
Elsgaard L (2001) Ethylene turn-over in soil, litter and sediment. Soil Biol Biochem 33:249–252
Fukuda H, Ogawa T, Tanase S (1993) Ethylene production by micro-organisms. Adv Microb Physiol 35:275–306
Goodlass G, Smith K (1978) Effect of pH, organic matter content and nitrate on the evolution of ethylene from soils. Soil Biol Biochem 10:193–199
Guideline IHT (2005) Validation of analytical procedures: text and methodology Q2 (R1), current Step 4 version. In: International Conference on Harmonisation, Geneva, Switzerland, p 15
Helsel DR (2011) Statistics for censored environmental data using Minitab and R. John Wiley & Sons, New York
Hendrickson O (1989) Implications of natural ethylene cycling processes for forest soil acetylene reduction assays. Can J Microbiol 35:713–718
Horn R, Vossbrink J, Peth S, Becker S (2007) Impact of modern forest vehicles on soil physical properties. For Ecol Manag 248:56–63
Hunt P, Campbell R, Sojka R, Parsons J (1981) Flooding-induced soil and plant ethylene accumulation and water status response of field-grown tobacco. Plant Soil 59:427–439
IUSS Working Group WRB (2015) World Reference Base for Soil Resources 2014, update 2015 International soil classification system for naming soils and creating legends for soil maps. World Soil Res Rep No. 106. FAO, Rome
Jäckel U, Schnell S, Conrad R (2004) Microbial ethylene production and inhibition of methanotrophic activity in a deciduous forest soil. Soil Biol Biochem 36:835–840
Jansone L, Von Wilpert K, Hartmann P (2020) Natural recovery and liming effects in acidified forest soils in SW Germany. Soil Syst 4:1–35
Lindberg T, Granhall U, Berg B (1979) Ethylene formation in some coniferous forest soils. Soil Biol Biochem 11:637–643
Lynch J (1975) Ethylene in soil. Nature 256:576–577
Maier M, Gartiser V, Schengel A, Lang V (2020) Long term soil gas monitoring as tool to understand soil processes. Appl Sci 10:8653
Mattoo AK, Suttle JC, Press C (1991) The plant hormone ethylene. CRC Press, Boca Raton, Fl
Meek BD, Ehlig CF, Stolzy LH, Graham LE (1983) Furrow and trickle irrigation: effects on soil oxygen and ethylene and tomato yield. Soil Sci Soc Am J 47:631–635
Negi S, Ivanchenko MG, Muday GK (2008) Ethylene regulates lateral root formation and auxin transport in Arabidopsis thaliana. Plant J 55:175–187
Nwanganga F, Chapple M (2020) Logistic regression. In: Nwanganga F, Chapple M (eds) Practical Machine Learning in R. John Wiley & Sons, New York, pp 165–219
Otani T, Ae N (1993) Ethylene and carbon dioxide concentrations of soils as influenced by rhizosphere of crops under field and pot conditions. Plant Soil 150:255–262
Pandey BK, Huang G, Bhosale R, Hartman S, Sturrock CJ, Jose L, Martin OC, Karady M, Voesenek LA, Ljung K (2021) Plant roots sense soil compaction through restricted ethylene diffusion. Science 371:276–280
Peng C-YJ, Lee KL, Ingersoll GM (2002) An introduction to logistic regression analysis and reporting. J Educ Res 96:3–14
Primrose SB (1979) Ethylene and agriculture: the role of the microbe. J Appl Bacteriol 46:1–25
Proctor CH (2008) A simple definition of detection limit. J Agric Biol Environ Stat 13:99–120
Reid MS (1995) Ethylene in plant growth, development, and senescence. In: Davies PJ (ed) Plant Hormones. Springer, pp 486–508
Rigler E, Zechmeister-Boltenstern S (1999) Oxidation of ethylene and methane in forest soils—effect of CO2 and mineral nitrogen. Geoderma 90:147–159
Rożek K, Rola K, Błaszkowski J, Leski T, Zubek S (2020) How do monocultures of fourteen forest tree species affect arbuscular mycorrhizal fungi abundance and species richness and composition in soil? For Ecol Manag 465:118091
Sawada S, Nakahata K, Totsuka T (1985) Fundamental studies on dynamics of ethylene in an ecosystem: III. Degradation capacity of atmospheric ethylene in soils taken from various vegetations. Japanese J Ecol 35:453–459
Schack-Kirchner H, Hildebrand EE (1998) Prozessmonitoring mit Gaskonzentrationsmessungen in Waldböden. In: Hildebrand EE (ed) Der Gashaushalt von Waldböden: Messung, Modellierung und ökologische Bedeutung, Freiburger Bodenkundliche Abhandlungen. Albert-Ludwigs-Univ, Freiburg, Germany, p 149
Schack-Kirchner H, Hildebrand EE, Von Wilpert K (1993) Ein konvektionsfreies Sammelsystem für Bodenluft. Zeitschrift für Pflanzenernährung und Bodenkunde 156:307–310
Schäffer J (2012) Bodenstruktur, Belüftung und Durchwurzelung befahrener Waldböden – Prozessstudien und Monitoring. In: Fakultät für Forst- und Umweltwissenschaften Universität Freiburg, Baden-Württemberg F (Eds) Schriftenreihe Freiburger Forstliche Forschung. Freiburg, Germany, p 148
Schäffer J, Von Wilpert K (2004) Einfluss befahrungsbedingter Bodenstrukturstörungen auf die Wurzelausbildung von Eichen auf durch "Lothar" verursachten Sturmwurfflächen. In: Schriftenreihe der Landesforstverwaltung Baden-Württemberg. Germany, Stuttgart, pp 213–225
Schmid I, Kazda M (2001) Vertical distribution and radial growth of coarse roots in pure and mixed stands of Fagus sylvatica and Picea abies. Can J For Res 31:539–548
Schmid I, Kazda M (2005) Clustered root distribution in mature stands of Fagus sylvatica and Picea abies. Oecologia 144:25–31
Schmidt-Walter P, Trotsiuk V, Meusburger K, Zacios M, Meesenburg H (2020) Advancing simulations of water fluxes, soil moisture and drought stress by using the LWF-Brook90 hydrological model in R. Agric For Meteorol 291:108023
Sexstone A, Mains C (1990) Production of methane and ethylene in organic horizons of spruce forest soils. Soil Biol Biochem 22:135–139
Sharp RE, Lenoble ME, Else MA, Thorne ET, Gherardi F (2000) Endogenous ABA maintains shoot growth in tomato independently of effects on plant water balance: evidence for an interaction with ethylene. J Exp Bot 51:1575–1584
Simunek J, Van Genuchten MT, Sejna M (2005) The HYDRUS-1D software package for simulating the one-dimensional movement of water, heat, and multiple solutes in variably-saturated media. In: User Manual, Version 1.0. Czech Republic, PC Progress. Prague, p 240
Sisler EC, Yang SF (1984) Ethylene, the gaseous plant hormone. Bioscience 34:234–238
Smith A, Cook RJ (1974) Implications of ethylene production by bacteria for biological balance of soil. Nature 252:703–705
Smith K, Dowdell R (1974) Field studies of the soil atmosphere: I. Relationships between ethylene, oxygen, soil moisture content, and temperature. J Soil Sci 25:217–230
Smith K, Restall S (1971) The occurrence of ethylene in anaerobic soil. J Soil Sci 22:430–443
Smith K, Russell RS (1969) Occurrence of ethylene, and its significance, in anaerobic soil. Nature 222:769–771
Van Cleemput O, El-Sebaay AS, Baert L (1983) Evolution of gaseous hydrocarbons from soil: Effect of moisture content and nitrate level. Soil Biol Biochem 15:519–524
Xu X, Inubushi K (2009) Temperature effects on ethylene and methane production from temperate forest soils. Chi Sci Bull 54:1426–1433
Xu X, Yuan B, Wei J (2008) Vertical distribution and interaction of ethylene and methane in temperate volcanic forest soils. Geoderma 145:231–237
Zechmeister-Boltenstern S, Nikodim L (1999) Effect of water tension on ethylene production and consumption in montane and lowland soils in Austria. Eur J Soil Sci 50:425–432
Zechmeister-Boltenstern S, Smith KA (1998) Ethylene production and decomposition in soils. Biol Fertil Soils 26:354–361
Zhong S, Shi H, Xue C, Wei N, Guo H, Deng XW (2014) Ethylene-orchestrated circuitry coordinates a seedling’s response to soil cover and etiolated growth. Proc Natl Acad Sci 111:3913–3920
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Zero-truncated and zero-inflated models for count data. In: Mixed effects models and extensions in ecology with R. Springer, New York, pp 261–293
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was funded by the WKF (Waldklimafond/Forest Climate Fund) and the FNR (Fachagentur Nachwachsende Rohstoffe e.V.), jointly managed by the Federal Ministry for the Environment (BMU) and the Federal Ministry of Food and Agriculture (BMEL)—grant number 2218WK58X4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(CSV 12188 kb)
Appendix
Appendix
Track positions at the different soil compaction study sites with “n” indicating the numbers of soil gas samplers installed at the respective positions. Note that lane does not automatically indicate a compacted soil. At Weisweil, the compaction status is reversed because soil loosening was done on compacted soil within the lane. Soil gas was sampled at: directly on the lane (LA); at the inner track (IT), between both lanes; Wall left/right (WL/WR), 30 cm left of the lane; at the outer track (OT), 50 to 80 cm from the lane; and in forest stock (ST) where no influence of forest machinery on soil was assumed. At Müllheim (MU a distinction could only done between compacted (C) and non-compacted (NC) soil
Cross-section through the passive soil gas samplers installed on all plots according to Schack-Kirchner et al. (1993)
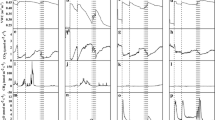
Time series of C2H4 measurements at all LTEMPs combined representing upland forest soils. Y-axes are logarithm-transformed (log10); dashed lines represent the LOD and LOQ. Orange circle: enhanced C2H4 detection frequencies in 2002 and 2003. Black circle: several C2H4 measurements at HDS and ROS on the same day. Blue circle: root-borne C2H4 measurements in humus at ESS
Pairwise comparison of number of ethylene measurements since 2010 of adjacent beech and spruce plots of the same site. The dashed line shows the depth profile of root density determined by root counts at the same depth as the gas collection. It is true that spruce plots generally showed a higher ethylene occurrence in the same observation period. However, a correlation to a simultaneously increased root density in the topsoil under spruce could not be determined
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lang, V., Schneider, V., Puhlmann, H. et al. Spotting ethylene in forest soils—What influences the occurrence of the phytohormone?. Biol Fertil Soils 59, 953–972 (2023). https://doi.org/10.1007/s00374-023-01763-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-023-01763-z