Abstract
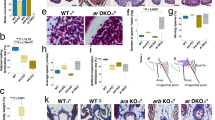
Steroid hormones play numerous important and diverse roles in the differentiation and development of vertebrate primary and secondary reproductive characteristics. However, the exact role of androgen receptors—which bind circulating androgens—in this regulatory pathway is unclear. Teleost fishes further complicate this question by having two paralogs of the androgen receptor (ARα and ARβ) resulting from a duplication of their ancestral genome. We investigated the functional role of these two ARs on adult testes mass, by eliminating receptor function of one or both paralogs using CRISPR/Cas9 genome edited Astatotilapia burtoni, an African cichlid fish. Fish with two or more functional AR alleles were more likely to be male compared to fish with one or fewer, suggesting that the two paralogs may play redundant roles in the A. burtoni sex determination system. We replicated previous work showing that fish lacking functional ARβ possess testes smaller than wild-type fish, while fish lacking ARα possess testes larger than wild-type fish. However, we found novel evidence supporting a complex relationship between the two AR paralogs in the regulation of testes mass. For instance, the effects of ARα mutation on testes mass are eliminated in homozygous ARβ mutants but the reverse is not true. These results suggest a dynamic relationship between these two AR paralogs where ARβ functions may be permissive to ARα functions in the control of testes mass. This mechanism may contribute to the robust physiological plasticity displayed by A. burtoni and other social teleost fishes.





Similar content being viewed by others
References
Alward BA, Laud VA, Skalnik CJ et al (2020) Modular genetic control of social status in a cichlid fish. Proc Natl Acad Sci 117:28167–28174
Baker IJ, Solar II, Donaldson EM (1988) Masculinization of chinook salmon (Oncorhynchus tshawytscha) by immersion treatments using 17α-methyltestosterone around the time of hatching. Aquaculture 72:359–367. https://doi.org/10.1016/0044-8486(88)90224-4
Baroillera J-F, Guiguenb Y, Fostierb A (1999) Endocrine and environmental aspects of sex differentiation in fis. Cell Mol Life Sci 55:910–931. https://doi.org/10.1007/s00167-018-5125-x
Blazquez M, Felip A, Zanuy S et al (2001) Critical period of androgen-inducible sex differentiation in a teleost fish, the European sea bass. J Fish Biol 58:342–358. https://doi.org/10.1006/jfbi.2000.1453
Brawand D, Wagner CE, Li YI et al (2014) The genomic substrate for adaptive radiation in African cichlid fish. Nature 513:375–381. https://doi.org/10.1038/nature13726
Crowder CM, Lassiter CS, Gorelick DA (2018) Nuclear androgen receptor regulates testes organization and oocyte maturation in Zebrafish. Endocrinology 159:980–993. https://doi.org/10.1210/en.2017-00617
De Gendt K, Swinnen JV, Saunders PTK et al (2004) A Sertoli cell-selective knockout of the androgen receptor causes spermatogenic arrest in meiosis. Proc Natl Acad Sci USA 101:1327–1332. https://doi.org/10.1073/pnas.0308114100
Douard V, Brunet F, Boussau B et al (2008) The fate of the duplicated androgen receptor in fishes: a late neofunctionalization event? BMC Evol Biol 8:336. https://doi.org/10.1186/1471-2148-8-336
Feist G, Yeoh CG, Fitzpatrick MS, Schreck CB (1995) The production of functional sex-reversed male rainbow trout with 17α-methyltestosterone and 11 β-hydroxyandrostenedione. Aquaculture 131:145–152. https://doi.org/10.1016/0044-8486(94)00336-M
Fernald RD (2012) Social control of the brain. Annu Rev Neurosci 35:135–151
Fernald RD, Hirata NR (1977) Field study of Haplochromis burtoni: quantitative behavioural observations. Anim Behav 25:964–975. https://doi.org/10.1016/0003-3472(77)90048-3
Fujii J, Kodama M, Oike A et al (2014) Involvement of androgen receptor in sex determination in an amphibian species. PLoS ONE. https://doi.org/10.1371/journal.pone.0093655
Gale WL, Fitzpatrick MS, Lucero M et al (1999) Masculinization of Nile tilapia (Oreochromis niloticus) by immersion in androgens. Aquaculture 178:349–357. https://doi.org/10.1016/S0044-8486(99)00136-2
Glasauer SMK, Neuhauss SCF (2014) Whole-genome duplication in teleost fishes and its evolutionary consequences. Mol Genet Genom 289:1045–1060. https://doi.org/10.1007/s00438-014-0889-2
Harbott LK, Burmeister SS, White RB et al (2007) Androgen receptors in a cichlid fish, Astatotilapia burtoni: structure, localization, and expression levels. J Comp Neurol 504:57–73. https://doi.org/10.1002/cne.21435
Hossain MS, Larsson A, Scherbak N et al (2008) Zebrafish androgen receptor: isolation. Mol Biochem 369:361–369. https://doi.org/10.1095/biolreprod.107.062018
Ikeuchi T, Todo T, Kobayashi T, Nagahama Y (1999) cDNA cloning of a novel androgen receptor subtype. J Biol Chem 274:25205–25209. https://doi.org/10.1074/jbc.274.36.25205
Juntti SA, Tollkuhn J, Wu MV et al (2010) The androgen receptor governs the execution, but not programming, of male sexual and territorial behaviors. Neuron 66:260–272. https://doi.org/10.1016/j.neuron.2010.03.024
Kavumpurath S, Pandian TJ (1994) Masculinization of fighting fish, Betta splendens Regan, using synthetic or natural androgens. Aquac Res 25:373–381. https://doi.org/10.1111/j.1365-2109.1994.tb00702.x
Kikuchi K, Hamaguchi S (2013) Novel sex-determining genes in fish and sex chromosome evolution. Dev Dyn 242:339–353. https://doi.org/10.1002/dvdy.23927
Lorin T, Salzburger W, Böhne A (2015) Evolutionary fate of the androgen receptor-signaling pathway in ray-finned fishes with a special focus on cichlids. G3 Genes. Genomes Genet 5:2275–2283. https://doi.org/10.1534/g3.115.020685
Marjani M, Jamili S, Ghavam Mostafavi P, Ramin M (2012) Influence of 17-alpha methyl testosterone on masculinization and growth in Tilapia (Oreochromis mossambicus). J Fish Aquat Sci 4:71–74. https://doi.org/10.3923/jfas.2009.71.74
Maruska KP, Fernald RD (2011a) Plasticity of the reproductive axis caused by social status change in an african cichlid fish: II. Testicular gene expression and spermatogenesis. Endocrinology 152:291–302. https://doi.org/10.1210/en.2010-0876
Maruska KP, Fernald RD (2011b) Social regulation of gene expression in the hypothalamic-pituitary-gonadal axis. Physiology 26:412–423. https://doi.org/10.1152/physiol.00032.2011
Maruska KP, Becker L, Neboori A, Fernald RD (2013) Social descent with territory loss causes rapid behavioral, endocrine and transcriptional changes in the brain. J Exp Biol 216:3656–3666. https://doi.org/10.1242/jeb.088617
Matsumoto T, Shiina H, Kawano H et al (2008) Androgen receptor functions in male and female physiology. J Steroid Biochem Mol Biol 109:236–241. https://doi.org/10.1016/j.jsbmb.2008.03.023
Moser JC, Reeve JD, Bento JMS et al (2004) Eye size and behaviour of day- and night-flying leafcutting ant alates. J Zool 264:69–75. https://doi.org/10.1017/S0952836904005527
Ogino Y, Katoh H, Yamada G (2004) Androgen dependent development of a modified anal fin, gonopodium, as a model to understand the mechanism of secondary sexual character expression in vertebrates. FEBS Lett 575:119–126. https://doi.org/10.1016/j.febslet.2004.08.046
Ogino Y, Katoh H, Kuraku S, Yamada G (2009) Evolutionary history and functional characterization of androgen receptor genes in jawed vertebrates. Endocrinology 150:5415–5427. https://doi.org/10.1210/en.2009-0523
Ogino Y, Kuraku S, Ishibashi H et al (2016) Neofunctionalization of androgen receptor by Gain-of-Function mutations in teleost fish lineage. Mol Biol Evol 33:228–244. https://doi.org/10.1093/molbev/msv218
Orn S, Holbech H, Madsen TH et al (2003) Gonad development and vitellogenin production in zebrafish (Danio rerio) exposed to ethinylestradiol and methyltestosterone. Aquat Toxicol 65:397–411. https://doi.org/10.1016/s0166-445x(03)00177-2
Pandian TJ, Sheela SG (1995) Hormonal induction of sex reversal in fish. Aquaculture 138:1–22
Roberts NB, Juntti SA, Coyle KP et al (2016) Polygenic sex determination in the cichlid fish Astatotilapia burtoni. BMC Genom. https://doi.org/10.1186/s12864-016-3177-1
Stevenson TJ, Alward BA, Ebling FJ, Fernald RD, Kelly A, Ophir AG (2018) The value of comparative animal research: Krogh’s principle facilitates scientific discoveries. Policy insights from the behavioral and brain sciences 5(1):118–125. https://doi.org/10.1177/2372732217745097
Takeo J, Yamashita S (1999) Two distinct isoforms of cDNA encoding rainbow trout androgen receptors. J Biol Chem 274:5674–5680. https://doi.org/10.1074/jbc.274.9.5674
Walters KA, Simanainen U, Handelsman DJ (2010) Molecular insights into androgen actions in male and female reproductive function from androgen receptor knockout models. Hum Reprod Update 16:543–558. https://doi.org/10.1093/humupd/dmq003
Wisniewsky AB, Migeon CJ, Meyer-Bahlburg HFL et al (2000) Complete androgen insensitivity syndrome: Long-term medical, surgical, and psychosexual outcome. J Clin Endocrinol Metab 85:2664–2669. https://doi.org/10.1210/jcem.85.8.6742
Acknowledgements
We thank Vibhav Laud (Stanford University) for assistance dissecting fish. We also thank Humayd Mirza and Rohail Siddiqi (University of Houston) for assistance with PCRs and genotyping. Lastly, we thank Mariana Lopez and Lillian Jackson (University of Houston) for providing feedback on an earlier version of this manuscript.
Funding
This work was supported by an Arnold O. Beckman Fellowship to B.A.A., a University of Houston–National Research University Fund grant R0503962 to B.A.A., and NIH grants NS034950, MH101373, and MH096220 to R.D.F.
Author information
Authors and Affiliations
Contributions
Conceptualization—BAA, APH, RDF; methodology—BAA, APH; investigation—BAA, APH; writing—original draft, APH; writing—review and editing, BAA, APH; supervision—BAA.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing or financial interests.
Availability of data and material
Data and code used are available at https://github.com/AlwardLab.
Ethics approval
All experimental procedures were approved by the Stanford University Administrative Panel for Laboratory Animal Care (Protocol #9882).
Additional information
Communicated by B. Pelster.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hoadley, A.P., Fernald, R.D. & Alward, B.A. Control of testes mass by androgen receptor paralogs in a cichlid. J Comp Physiol B 192, 107–114 (2022). https://doi.org/10.1007/s00360-021-01417-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-021-01417-2