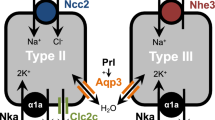
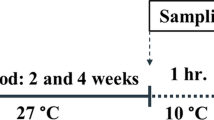
Abstract
The maintenance of ion and pH homeostasis despite changes in ambient temperature is crucial for ectothermic organisms. Thermal sensitivity of Na+/K+ ATPase mRNA expression, protein expression and activity was determined in gills of North Sea cod (NC) and Northeastern Arctic cod (NEAC), acclimated for 6 weeks at 4 and 10 °C and compared to field samples of North Sea cod (sNC), acclimatized to early spring (4 °C) and summer (18 °C) conditions. The same analyses were conducted in gills of the confamiliar whiting, acclimated at 4 and 10 °C. Branchial Na+/K+ ATPase capacities remained uncompensated at functional and protein levels in NC and NEAC at both acclimation temperatures. Na+/K+ ATPase mRNA expression in NEAC acclimated at 10 °C was about twofold higher compared to NC, indicating some population-specific differentiation at this level. Lower Na+/K+ ATPase capacities in gills of warm-acclimatized sNC at common assay temperatures indicate thermal compensation between seasonal extremes, and post-translational modifications contributed to this mitigation at high assay temperature. Together, cod compensates Na+/K+ ATPase capacities on the warm edge of the thermal window and below 4 °C, respectively. In contrast, whiting Na+/K+ ATPase capacities were cold compensated at 4 °C, supported by 1.5-fold higher mRNA and protein expression. Besides, capacities were lower in whiting compared to NC and NEAC at optimum temperature, which may be advantageous in terms of reduced maintenance cost, but at temperatures ≤4 °C, compensation may represent an energy trade-off to maintain homeostasis. The species-specific response of gadid Na+/K+ ATPase indicates certain threshold temperatures beyond which compensation of the pump is elicited, possibly related to the different biogeography of these species.






Similar content being viewed by others
References
Allen JC, Schwartz A (1969) A possible biochemical explanation for the insensitivity of the rat to cardiac glycosides. J Pharmacol Exp Ther 168:42–46
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Burton RF (1986) Ionic regulation in fish: the influence of acclimation temperature on plasma composition and apparent set points. Comp Biochem Physiol A Mol Integr Physiol 85:23–28. doi:10.1016/0300-9629(86)90456-1
Deigweiher K, Koschnick N, Pörtner H-O, Lucassen M (2008) Acclimation of ion regulatory capacities in gills of marine fish under environmental hypercapnia. Am J Physiol Regul Integr Comp Physiol 295:R1660–R1670. doi:10.1152/ajpregu.90403.2008
Finstad B, Staurnes M, Reite OB (1988) Effect of low temperature on sea-water tolerance in rainbow trout, Salmo gairdneri. Aquaculture 72:319–328. doi:10.1016/0044-8486(88)90220-7
Flik G, Verbost PM (1993) Calcium transport in fish gills and intestine. J Exp Biol 184:17–29
Gibbs A, Somero GN (1990) Na+-K+-adenosine triphosphatase activities in gills of marine teleost fishes: changes with depth, size and locomotory activity level. Mar Biol 106:315–321. doi:10.1007/BF01344307
Guynn S, Dowd F, Petzel D (2002) Characterization of gill Na/K-ATPase activity and ouabain binding in Antarctic and New Zealand nototheniid fishes. Comp Biochem Physiol A Mol Integr Physiol 131:363–374. doi:10.1016/S1095-6433(01)00488-3
Harden Jones FR, Scholes P (1974) The effect of low temperature on cod, Gadus morhua. J Cons int Expor Mer 35:258–271. doi:10.1093/icesjms/35.3.258
Hazel JR (1995) Thermal adaptation in biological membranes: is homeoviscous adaptation the explanation? Annu Rev Physiol 57:19–42. doi:10.1146/annurev.ph.57.030195.000315
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. doi:10.1038/227680a0
Lannig G, Eckerle LG, Serendero I et al (2003) Temperature adaptation in eurythermal cod (Gadus morhua): a comparison of mitochondrial enzyme capacities in boreal and Arctic populations. Mar Biol. doi:10.1007/s00227-002-0967-6
Lin H, Randall DJ (1991) Evidence for the presence of an electrogenic proton pump on the trout gill epithelium. J Exp Biol 161:119–134
Lucassen M, Koschnick N, Eckerle LG, Pörtner H-O (2006) Mitochondrial mechanisms of cold adaptation in cod (Gadus morhua L.) populations from different climatic zones. J Exp Biol 209:2462–2471. doi:10.1242/jeb.02268
McCarty LS, Houston AH (1977) Na+:K+- and HCO−-stimulated ATPase activities in the gills and kidneys of thermally acclimated rainbow trout, Salmo gairdneri. Can J Zool 55:704–712. doi:10.1139/z77-092
Metz JR, van den Burg EH, Wendelaar Bonga SE, Flik G (2003) Regulation of branchial Na+/K+-ATPase in common carp Cyprinus carpio L. acclimated to different temperatures. J Exp Biol 206:2273–2280. doi:10.1242/jeb.00421
Michael K, Kreiss CM, Hu MY et al (2016) Adjustments of molecular key components of branchial ion and pH regulation in Atlantic cod (Gadus morhua) in response to ocean acidification and warming. Comp Biochem Physiol B Biochem Mol Biol 193:33–46. doi:10.1016/j.cbpb.2015.12.006
Nordlie FG (2009) Environmental influences on regulation of blood plasma/serum components in teleost fishes: a review. Rev Fish Biol Fish 19:481–564. doi:10.1007/s11160-009-9131-4
Olsvik PA, Søfteland L, Lie KK (2008) Selection of reference genes for qRT-PCR examination of wild populations of Atlantic cod Gadus morhua. BMC Res Notes 1:47. doi:10.1186/1756-0500-1-47
Packer RK, Garvin JL (1998) Seasonal differences in activity of perch (Perca flavescens) gill Na+/K+ ATPase. Comp Biochem Physiol B: Biochem Mol Biol 120:777–783. doi:10.1016/S0305-0491(98)10078-0
Paxton R, Umminger BL (1983) Altered activities of branchial and renal Na/K-and Mg-ATPases in cold-acclimated goldfish (Carassius auratus). Comp Biochem Physiol B Biochem Mol Biol. doi:10.1016/0305-0491(83)90218-3
Pörtner H-O, Hardewig I, Sartoris FJ, Van Dijk PLM (1998) Energetic aspects of cold adaptation: critical temperatures in metabolic, ionic and acid-base regulation? In: Pörtner H-O, Playle RC (eds) Cold Ocean Physiology. Cambridge University Press, Cambridge, pp 88–120
Pörtner H-O, Berdal B, Blust R et al (2001) Climate induced temperature effects on growth performance, fecundity and recruitment in marine fish: developing a hypothesis for cause and effect relationships in Atlantic cod (Gadus morhua) and common eelpout (Zoarces viviparus). Cont Shelf Res 21:1975–1997. doi:10.1016/S0278-4343(01)00038-3
Pörtner H-O, Lucassen M, Storch D (2005) Metabolic biochemistry: its role in thermal tolerance and in the capacities of physiological and ecological function. In: Physiology of polar fishes. Academic Press, pp 79–154
Righton D, Andersen KH, Neat F et al (2010) Thermal niche of Atlantic cod Gadus morhua: limits, tolerance and optima. Mar Ecol Prog Ser 420:1–13. doi:10.3354/meps08889
Schwarzbaum PJ, Wieser W, Niederstätter H (1991) Contrasting effects of temperature acclimation on mechanisms of ionic regulation in a eurythermic and a stenothermic species of freshwater fish (Rutilus rutilus and Savelinus alpinus). Comp Biochem Physiol A Mol Integr Physiol 98:483–489. doi:10.1016/0300-9629(91)90435-f
Staurnes M, Rainuzzo JR, Sigholt T, Jøgensen L (1994) Acclimation of Atlantic cod (Gadus morhua) to cold water: stress response, osmoregulation, gill lipid composition and gill Na-K-ATPase activity. Comp Biochem Physiol A Mol Integr Physiol 109:413–421. doi:10.1016/0300-9629(94)90145-7
Stuenkel EL, Hillyard SD (1980) Effects of temperature and salinity on gill Na+-K+ ATPase activity in the pupfish, Cyprinodon salinus. Comp Biochem Physiol A Mol Integr Physiol 67:179–182. doi:10.1016/0300-9629(80)90426-0
Umminger BL (1971) Chemical studies of cold death in the Gulf killifish, Fundulus grandis. Comp Biochem Physiol A Mol Integr Physiol 39:625–632. doi:10.1016/0300-9629(71)90184-8
Wodtke E (1981) Temperature adaptation of biological membranes. Compensation of the molar activity of cytochrome c oxidase in the mitochondrial energy-transducing membrane during thermal acclimation of the carp (Cyprinus carpio L.). Biochim Biophys Acta 640:710–720
Acknowledgments
The authors gratefully acknowledge the measurement of Na+/K+ ATPase capacities of the whiting gill samples by Johannes Leiß. This work was funded by the project BIOACID (Biological Impacts of Ocean Acidification, phase 1; FKZ 03F0608B, sub-project 2.3.2) of the German Ministry for Education and Research (BMBF), and is a contribution to the PACES research program (work package 1.6) of the AWI funded by the Helmholtz Association.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Michael, K., Koschnick, N., Pörtner, HO. et al. Response of branchial Na+/K+ ATPase to changes in ambient temperature in Atlantic cod (Gadus morhua) and whiting (Merlangius merlangus). J Comp Physiol B 186, 461–470 (2016). https://doi.org/10.1007/s00360-016-0970-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-016-0970-8