Abstract
Purpose
To explore the immune phenotype of peripheral blood mononuclear cells (PBMC) in patients with high-risk non-muscle invasive bladder cancer (NMIBC).
Methods
We prospectively collected blood samples from patients with high-risk NMIBC treated at our institution. PBMC were analyzed by flow cytometry to determine the frequency of T cells and NK cells and the expression of immunoregulatory molecules (Tim-3, TIGIT and PD-1). PBMC from healthy donors (HD) were included for comparison, and associations with response to BCG were investigated.
Results
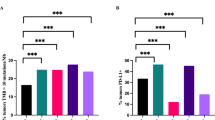
A total of 38 patients were included, 19 BCG responders and 19 BCG refractory. Compared to 16 PBMC from HD, the frequency of total NK cells was significantly higher in patients with NMIBC [15.2% (IQR: 11.4, 22.2) vs. 5.72% (IQR: 4.84, 9.79); p = 0.05], whereas the frequency of T cells was not statistically different. Both Tim-3 and TIGIT expressions were significantly higher in NMIBC compared to HD, particularly in NK cells [13.8% (11.0; 22.4) vs. 5.56% (4.20; 10.2) and 34.9% (18.9; 53.5) vs. 1.82% (0.63; 5.16), respectively; p < 0.001]. Overall, the expression of PD-1 in all cell types was low in both NMIBC patients and HD. The immune phenotype was not significantly different before and after initiation of BCG. However, the proportion of CD8+ T cells before BCG was significantly higher in responders.
Conclusion
The immune phenotype of PBMC from patients with high-risk NMIBC was significantly different from HD, regardless of the presence of disease or the initiation of BCG. Peripheral CD8+ T cells could play a role in response to BCG.

Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2017) Cancer statistics, 2017. CA Cancer J Clin 67(1):7–30
Chang SS, Bochner BH, Chou R, Dreicer R, Kamat AM, Lerner SP et al (2017) Treatment of non-metastatic muscle-invasive bladder cancer: AUA/ASCO/ASTRO/SUO guideline. J Urol sept 198(3):552–559
Babjuk M, Böhle A, Burger M, Capoun O, Cohen D, Compérat EM et al (2017) EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder: update 2016. Eur Urol mars 71(3):447–461
Yeung C, Dinh T, Lee J (2014) The health economics of bladder cancer: an updated review of the published literature. PharmacoEconomics 32(11):1093–1104
Couzin-Frankel J (2013) Breakthrough of the year 2013. Cancer immunotherapy. Science 342(6165):1432–1433
Redelman-Sidi G, Glickman MS, Bochner BH (2014) The mechanism of action of BCG therapy for bladder cancer—a current perspective. Nat Rev Urol mars 11(3):153–162
Brierley J, Gospodarowicz MK, Wittekind C (2017) TNM classification of malignant tumours, 8th edn. John Wiley & Sons Inc., Chichester, Hoboken
Eble JN, Sauter G, Epstein JI, Sesterhenn IA (2004) Pathology and genetics of tumours of the urinary system and male genital organs. IARC Press, Lyon, p 359 (World Health Organization classification of tumours)
Coley WB (1893) The treatment of malignant tumors by repeated inoculations of erysipelas. With a report of ten original cases. Am J Med Sci 105:487–511
Lamm DL, Thor DE, Harris SC, Reyna JA, Stogdill VD, Radwin HM (1980) Bacillus Calmette-Guerin immunotherapy of superficial bladder cancer. J Urol juill 124(1):38–40
D’Andrea D, Moschini M, Gust K, Abufaraj M, Özsoy M, Mathieu R et al (2017) prognostic role of neutrophil-to-lymphocyte ratio in primary non-muscle-invasive bladder cancer. Clin Genitourin Cancer 15(5):e755–e764
Spitzer MH, Carmi Y, Reticker-Flynn NE, Kwek SS, Madhireddy D, Martins MM et al (2017) Systemic immunity is required for effective cancer immunotherapy. Cell 168(3):487–502.e15
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 22 mars 12(4):252–264
Francisco LM, Salinas VH, Brown KE, Vanguri VK, Freeman GJ, Kuchroo VK et al (2009) PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J Exp Med 206(13):3015–3029
Pichler R, Heidegger I, Fritz J, Danzl M, Sprung S, Zelger B et al (2017) PD-L1 expression in bladder cancer and metastasis and its influence on oncologic outcome after cystectomy. Oncotarget 8(40):66849–66864
Yu X, Harden K, Gonzalez LC, Francesco M, Chiang E, Irving B et al (2009) The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol 10(1):48–57
Stanietsky N, Simic H, Arapovic J, Toporik A, Levy O, Novik A et al (2009) The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc Natl Acad Sci USA 106(42):17858–17863
Johnston RJ, Comps-Agrar L, Hackney J, Yu X, Huseni M, Yang Y et al (2014) The immunoreceptor TIGIT regulates antitumor and antiviral CD8+ T cell effector function. Cancer Cell 26(6):923–937
Fourcade J, Sun Z, Benallaoua M, Guillaume P, Luescher IF, Sander C et al (2010) Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen-specific CD8+ T cell dysfunction in melanoma patients. J Exp Med 207(10):2175–2186
Cai C, Xu Y-F, Wu Z-J, Dong Q, Li M-Y, Olson JC et al (2016) Tim-3 expression represents dysfunctional tumor infiltrating T cells in renal cell carcinoma. World J Urol 34(4):561–567
Wang Y, Sun J, Gao W, Song B, Shao Q, Zhao L et al (2017) Preoperative Tim-3 expression on peripheral NK cells is correlated with pathologic TNM staging in colorectal cancer. Mol Med Rep 15(6):3810–3818
Jie H-B, Srivastava RM, Argiris A, Bauman JE, Kane LP, Ferris RL (2017) Increased PD-1 + and TIM-3 + TILs during cetuximab therapy inversely correlate with response in head and neck cancer patients. Cancer Immunol Res 5(5):408–416
da Silva IP, Gallois A, Jimenez-Baranda S, Khan S, Anderson AC, Kuchroo VK et al (2014) Reversal of NK-cell exhaustion in advanced melanoma by Tim-3 blockade. Cancer Immunol Res 2(5):410–422
Xu L, Huang Y, Tan L, Yu W, Chen D, Lu C et al (2015) Increased Tim-3 expression in peripheral NK cells predicts a poorer prognosis and Tim-3 blockade improves NK cell-mediated cytotoxicity in human lung adenocarcinoma. Int Immunopharmacol 29(2):635–641
Lima L, Dinis-Ribeiro M, Longatto-Filho A, Santos L (2012) Predictive biomarkers of bacillus calmette-guérin immunotherapy response in bladder cancer: where are we now? Adv Urol 2012:232609
Kelley DR, Haaff EO, Becich M, Lage J, Bauer WC, Dresner SM et al (1986) Prognostic value of purified protein derivative skin test and granuloma formation in patients treated with intravesical bacillus Calmette-Guerin. J Urol 135(2):268–271
Taniguchi K, Koga S, Nishikido M, Yamashita S, Sakuragi T, Kanetake H et al (1999) Systemic immune response after intravesical instillation of bacille Calmette-Guérin (BCG) for superficial bladder cancer. Clin Exp Immunol 115(1):131–135
Raj GV, Herr H, Serio AM, Donat SM, Bochner BH, Vickers AJ et al (2007) Treatment paradigm shift may improve survival of patients with high risk superficial bladder cancer. J Urol 177(4):1283–1286 (discussion 1286)
Kamphorst AO, Pillai RN, Yang S, Nasti TH, Akondy RS, Wieland A et al (2017) Proliferation of PD-1 + CD8 T cells in peripheral blood after PD-1-targeted therapy in lung cancer patients. Proc Natl Acad Sci USA 114(19):4993–4998
Siefker-Radtke AO, Apolo AB, Bivalacqua TJ, Spiess PE, Black PC (2018) Immunotherapy with checkpoint blockade in the treatment of urothelial carcinoma. J Urol 199:1129–1142. https://doi.org/10.1016/j.juro.2017.10.041
Funding
These studies were funded by grants from the Cancer Research Institute and from the National Institutes of Health (R01 CA201189).
Author information
Authors and Affiliations
Contributions
FA: project development, data collection, data analysis, manuscript writing. AMF: project development, data collection, data analysis, manuscript editing. HA: data collection, manuscript editing. MDG: project development, data analysis, manuscript editing. NB: project development, data analysis, manuscript editing. JPS: project development, data collection, data analysis, manuscript editing
Corresponding author
Ethics declarations
Conflict of interest
NB is supported by the Parker Institute for Cancer Immunotherapy. AMF is supported by an NIH Immunology Training Grant (T32AI007605).
Research involving human participants
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Audenet, F., Farkas, A.M., Anastos, H. et al. Immune phenotype of peripheral blood mononuclear cells in patients with high-risk non-muscle invasive bladder cancer. World J Urol 36, 1741–1748 (2018). https://doi.org/10.1007/s00345-018-2359-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2359-7