Abstract
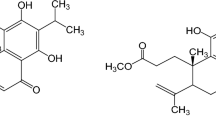
A new isochromanone, cladosporinisochromanone (1), accompanied by 15 known compounds (2–16) were obtained from secondary metabolites produced by marine-derived fungus Cladosporium sp. DLT-5. NMR and HRESIMS spectra elucidation determined the planar structure of 1. Subsequent electronic circular dichroism (ECD) experiment assigned the absolute configuration of 1. Compounds 1, 2, 4–6, and 10 displayed different degrees of neuroprotective activities on human neuroblastoma cells SH-SY5Y. Five compounds (1, 3–5, and 13) emerged resistance to protein tyrosine phosphatase 1B (PTP1B), further kinetic analysis and molecular docking study indicated that the most potent compound 13 (IC50 value of 10.74±0.61 µmol/L) was found as a noncompetitive inhibitor for PTP1B. Surface plasmon resonance (SPR) and molecular docking studies also demonstrated the interaction between compound 12 and Niemann-Pick C1 Like 1 (NPC1L1), which has been identified as significant therapeutic target for hypercholesteremia. In addition, compounds 3, 6, and 14 showed attractive inhibitory activity against the phytopathogenic fungi: Colletotrichum capsici. Therefore, library of Cladosporium metabolites is enriched and new active uses of known compounds are explored.
Similar content being viewed by others
5 Data Availability Statement
All data generated or analyzed during this study are included in this published article and its supplementary material.
References
Alam M B, Chowdhury N S, Sohrab M H et al. 2020. Cerevisterol alleviates inflammation via suppression of MAPK/NF-κB/AP-1 and activation of the Nrf2/HO-1 signaling cascade. Biomolecules, 10(2): 199, https://doi.org/10.3390/biom10020199.
Bensch K, Braun U, Groenewald J Z et al. 2012. The genus Cladosporium. Studies in Mycology, 72: 1–401, https://doi.org/10.3114/sim0003.
Boonyaketgoson S, Trisuwan K, Bussaban B et al. 2015. Isochromanone derivatives from the endophytic fungus Fusarium sp. PDB51F5. Tetrahedron Letters, 56(36): 5076–5078, https://doi.org/10.1016/j.tetlet.2015.07.048.
Campos F R, Barison A, Daolio C et al. 2005. Complete 1H and 13C NMR assignments of aurasperone A and fonsecinone A, two bis-naphthopyrones produced by Aspergillus aculeatus. Magnetic Resonance in Chemistry, 43(11): 962–965, https://doi.org/10.1002/mrc.1654.
Carroll A R, Copp B R, Davis R A et al. 2022. Marine natural products. Natural Product Reports, 39(6): 1122–1171, https://doi.org/10.1039/d1np00076d.
Cheng A L, Uetani N, Simoncic P D et al. 2002. Attenuation of leptin action and regulation of obesity by protein tyrosine phosphatase 1B. Developmental Cell, 2(4): 497–503, https://doi.org/10.1016/S1534-5807(02)00149-1.
Cox R H, Morris P, Cutler H G et al. 1983. Proton and carbon-13 nuclear magnetic resonance studies of the conformation of cytochalasin H derivatives and plant growth regulating effects of cytochalasins. Journal of Agricultural and Food Chemistry, 31(2): 405–408, https://doi.org/10.1021/jf00116a055.
Dai L T, Yang L, Kong F D et al. 2021. Cytotoxic indole-diterpenoids from the marine-derived fungus Penicillium sp. KFD28. Marine Drugs, 19(11): 613, https://doi.org/10.3390/md19110613.
Dettrakul S, Kittakoop P, Isaka M et al. 2003. Antimycobacterial pimarane diterpenes from the fungus Diaporthe sp. Bioorganic & Medicinal Chemistry Letters, 13(7): 1253–1255, https://doi.org/10.1016/S0960-894X(03)00111-2.
Fan Y Q, Jiang C J, Li P H et al. 2023. A new chloroazaphilone derivative with pro-angiogenesis activity from the hadal trench-derived fungus Chaetomium globosum YP-106. Journal of Oceanology and Limnology, 41(3): 1145–1151, https://doi.org/10.1007/s00343-022-2017-1.
Goldsmith M, Abramovitz L, Peer D. 2014. Precision nanomedicine in neurodegenerative diseases. ACS Nano, 8(3): 1958–1965, https://doi.org/10.1021/nn501292z.
Gu B B, Zhang Y Y, Ding L J et al. 2015. Preparative separation of sulfur-containing diketopiperazines from marine fungus Cladosporium sp. using high-speed counter-current chromatography in stepwise elution mode. Marine Drugs, 13(1): 354–365, https://doi.org/10.3390/md13010354.
Hosotani N, Kumagai K, Nakagawa H et al. 2005. Antimycins A10–A16, seven new antimycin antibiotics produced by Streptomyces spp. SPA-10191 and SPA-8893. The Journal of Antibiotics, 58(7): 460–467, https://doi.org/10.1038/ja.2005.61.
Hu M Q, Yang F, Huang Y W et al. 2021. Structural insights into the mechanism of human NPC1L1-mediated cholesterol uptake. Science Advances, 7(29): eabg3188, https://doi.org/10.1126/sciadv.abg3188.
Jung H A, Islam M N, Lee C M et al. 2013. Kinetics and molecular docking studies of an anti-diabetic complication inhibitor fucosterol from edible brown algae Eisenia bicyclis and Ecklonia stolonifera. Chemico-Biological Interactions, 206(1): 55–62, https://doi.org/10.1016/j.cbi.2013.08.013.
Kim J W, Choi H G, Song J H et al. 2019. Bioactive secondary metabolites from an endophytic fungus Phoma sp. PF2 derived from Artemisia princeps Pamp. The Journal of Antibiotics, 72(3): 174–177, https://doi.org/10.1038/s41429-018-0131-2.
Kobayashi S, Senoo F, Kihara M et al. 1971. Studies on the syntheses of benzoheterocyclic compounds. VI. Diphenide and its methoxyl derivatives. Chemical and Pharmaceutical Bulletin, 19(6): 1262–1267, https://doi.org/10.1248/cpb.19.1262.
Kong F D, Fan P, Zhou L M et al. 2019. Penerpenes A–D, four indole terpenoids with potent protein tyrosine phosphatase inhibitory activity from the marine-derived fungus Penicillium sp. KFD28. Organic Letters, 21(12): 4864–4867, https://doi.org/10.1021/acs.orglett.9b01751.
Li X J, Gao J M, Chen H et al. 2012. Toxins from a symbiotic fungus, Leptographium qinlingensis associated with Dendroctonus armandi and their in vitro toxicities to Pinus armandi seedlings. European Journal of Plant Pathology, 134(2): 239–247, https://doi.org/10.1007/s10658-012-9981-9.
Liu J K, Xie M, Li X Z et al. 2018. Main allelochemicals from the rhizosphere soil of Saussurea Lappa (Decne.) Sch. Bip. and their effects on plants’ antioxidase systems. Molecules, 23(10): 2506, https://doi.org/10.3390/molecules23102506.
Long H L, Qiu X H, Cao L et al. 2021. Discovery of the signal pathways and major bioactive compounds responsible for the anti-hypoxia effect of Chinese cordyceps. Journal of Ethnopharmacology, 277: 114215, https://doi.org/10.1016/j.jep.2021.114215.
Peng J X, Lin T, Wang W et al. 2013. Antiviral alkaloids produced by the mangrove-derived fungus Cladosporium sp. PJX-41. Journal of Natural Products, 76(6): 1133–1140, https://doi.org/10.1021/np400200k.
Pongcharoen W, Rukachaisirikul V, Phongpaichit S et al. 2006. Pimarane diterpene and cytochalasin derivatives from the endophytic fungus Eutypella scoparia PSU-D44. Journal of Natural Products, 69(5): 856–858, https://doi.org/10.1021/np0600649.
Rischer M, Lee S R, Eom H J et al. 2019. Spirocyclic cladosporicin A and cladosporiumins I and J from a Hydractinia-associated Cladosporium sphaerospermum SW67. Organic Chemistry Frontiers, 6(8): 1084–1093, https://doi.org/10.1039/c8qo01104d.
Ruffels J, Griffin M, Dickenson J M. 2004. Activation of ERK1/2, JNK and PKB by hydrogen peroxide in human SH-SY5Y neuroblastoma cells: role of ERK1/2 in H2O2-induced cell death. European Journal of Pharmacology, 483(2–3): 163–173, https://doi.org/10.1016/j.ejphar.2003.10.032.
Sun L, Li D L, Tao M H et al. 2012. Scopararanes C-G: new oxygenated pimarane diterpenes from the marine sediment-derived fungus Eutypella scoparia FS26. Marine Drugs, 10(12): 539–550, https://doi.org/10.3390/md10030539.
Tayone W C, Kanamaru S, Honma M et al. 2011. Absolute stereochemistry of novel isochromanone derivatives from Leptosphaeria sp. KTC 727. Bioscience, Biotechnology, and Biochemistry, 75(12): 2390–2393, https://doi.org/10.1271/bbb.110621.
Wang X N, Radwan M M, Taráwneh A H et al. 2013. Antifungal activity against plant pathogens of metabolites from the endophytic fungus Cladosporium cladosporioides. Journal of Agricultural and Food Chemistry, 61(19): 4551–4555, https://doi.org/10.1021/jf400212y.
Wu W, Dai H Q, Bao L et al. 2011. Isolation and structural elucidation of proline-containing cyclopentapeptides from an endolichenic Xylaria sp. Journal of Natural Products, 74(5): 1303–1308, https://doi.org/10.1021/np100909y.
Xiong H Y, Fei D Q, Zhou J S et al. 2009. Steroids and other constituents from the mushroom Armillaria lueo-virens. Chemistry of Natural Compounds, 45(5): 759–761, https://doi.org/10.1007/s10600-009-9456-1.
Yang L, Kong D X, Xiao N et al. 2022. Antidiabetic lanostane triterpenoids from the fruiting bodies of Ganoderma weberianum. Bioorganic Chemistry, 127: 106025, https://doi.org/10.1016/j.bioorg.2022.106025.
Zhang B, Wu J T, Zheng C J et al. 2021a. Bioactive cyclohexene derivatives from a mangrove-derived fungus Cladosporium sp. JJM22. Fitoterapia, 149: 104823, https://doi.org/10.1016/j.fitote.2020.104823.
Zhang F Z, Li X M, Yang S Q et al. 2019. Thiocladospolides A–D, 12-membered macrolides from the mangrove-derived endophytic fungus Cladosporium cladosporioides MA-299 and structure revision of pandangolide 3. Journal of Natural Products, 82(6): 1535–1541, https://doi.org/10.1021/acs.jnatprod.8b01091.
Zhang F, Zhou L M, Kong F D et al. 2020. Altertoxins with quorum sensing inhibitory activities from the marine-derived fungus Cladosporium sp. KFD33. Marine Drugs, 18(1): 67, https://doi.org/10.20944/preprints201912.0398.v1.
Zhang R S, Liu W J, Zeng J et al. 2022a. Niemann-Pick C1-Like 1 inhibitors for reducing cholesterol absorption. European Journal of Medicinal Chemistry, 230: 114111, https://doi.org/10.1016/j.ejmech.2022.114111.
Zhang R S, Liu W J, Zeng J et al. 2022b. Recent advances in the screening methods of NPC1L1 inhibitors. Biomedicine & Pharmacotherapy, 155: 113732, https://doi.org/10.1016/j.biopha.2022.113732.
Zhang R S, Song Z M, Wang X T et al. 2021b. One-step modification to identify dual-inhibitors targeting both pancreatic triglyceride lipase and Niemann-Pick C1-like 1. European Journal of Medicinal Chemistry, 216: 113358, https://doi.org/10.1016/j.ejmech.2021.113358.
Zhang Z Z, He X Q, Wu G W et al. 2018. Aniline-tetramic acids from the deep-sea-derived fungus Cladosporium sphaerospermum L3P3 cultured with the HDAC inhibitor SAHA. Journal of Natural Products, 81(7): 1651–1657, https://doi.org/10.1021/acs.jnatprod.8b00289.
Zhao B T, Nguyen D H, Le D D et al. 2018. Protein tyrosine phosphatase 1B inhibitors from natural sources. Archives of Pharmacal Research, 41(2): 130–161, https://doi.org/10.1007/s12272-017-0997-8.
Zhou L M, Kong F D, Fan P et al. 2019. Indole-diterpenoids with protein tyrosine phosphatase inhibitory activities from the marine-derived fungus Penicillium sp. KFD28. Journal of Natural Products, 82(9): 2638–2644, https://doi.org/10.1021/acs.jnatprod.9b00620.
Zhou X F, Sun J F, Ma W L et al. 2014. Bioactivities of six sterols isolated from marine invertebrates. Pharmaceutical Biology, 52(2): 187–190, https://doi.org/10.3109/13880209.2013.821664.
Zhu M L, Gao H Q, Wu C M et al. 2015. Lipid-lowering polyketides from a soft coral-derived fungus Cladosporium sp. TZP29. Bioorganic & Medicinal Chemistry Letters, 25(17): 3606–3609, https://doi.org/10.1016/j.bmcl.2015.06.072.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the China Agriculture Research System of MOF and MARA (CARS-21), the Financial Fund of the Ministry of Agriculture and Rural Affairs, China (No. NFZX2021), and the National Natural Science Foundation of China (No. 81973568)
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Dai, L., Xie, Q., Guo, J. et al. Bioactive chemical constituents from the marine-derived fungus Cladosporium sp. DLT-5. J. Ocean. Limnol. (2024). https://doi.org/10.1007/s00343-023-3084-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00343-023-3084-7