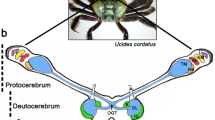
Abstract
As in vertebrates, brains play key roles in rhythmic regulation, neuronal maintenance, differentiation and function, and control of the release of hormones in arthropods. But the structure and functional domains of the brain are still not very clear in crustaceans. In the present study, we reveal the structural details of the brain in the redclaw crayfish using hematoxylin-eosin staining and microscopic examination, firstly. The brain of crayfish is consist of three main parts, namely, protocerebrum, deutocerebrum, and tritocerebrum, including some tracts and commissures, briefly. Secondly, at least 9 kinds of brain cells were identified on the basis of topology and cell shapes, as well as antibody labeling. We also provide morphological details of most cell types, which were previously un-described. In general, four types of glia and three types of neurosecretory cells were described except cluster 9/11 and cluster 10 cells. Glia were categorized into another three main kinds: (1) surface glia; (2) cortex glia; and (3) neuropile glia in addition to astrocytes identified by GFAP labelling. And neurosecretory cells were categorized into I, II and III types based on morphological observation. Finally, cluster 9/11 and 10 cells derived from the brain of crayfish, could be used for primary culture about 7–9 d under the optimized conditions. There results provide a resource for improving the knowledge of the still incompletely defined neuroendocrinology of this species. Using the crayfish as an animal model, we are easy to carry out further research in manipulating their endocrine system, exploring cellular and synaptic mechanisms so much as larval production on a small scale, such as in a cell or tissue.
Similar content being viewed by others
References
Álvarez R A, Villalobos M G P, Rosete G C, Sosa L R, Aréchiga H. 2005. Dopaminergic modulation of neurosecretory cells in the crayfish. Cellular and Molecular Neurobiology, 25 (2): 345–370.
Apitz H, Salecker I. 2014. A challenge of numbers and diversity: neurogenesis in the Drosophila optic lobe. Journal of Neurogenetics, 28 (3–4): 233–249.
Beltz B S, Sandeman D C. 2003. Regulation of life–long neurogenesis in the decapod crustacean brain. Arthropod Structure & Development, 32 (1): 39–60.
Benton J L, Beltz B S. 2002. Patterns of Neurogenesis in the midbrain of embryonic lobsters differ from proliferation in the insect and the crustacean ventral nerve cord. Journal of Neurobiology, 53 (1): 57–67.
Benton J L, Chaves da Silva P G, Sandeman D C, Beltz B S. 2013. First–generation neuronal precursors in the crayfish brain are not self–renewing. International Journal of Developmental Neuroscience, 31 (7): 657–666.
Benton J L, Kery R, Li J J, Noonin C, Söderhäll I, Beltz B S. 2014. Cells from the immune system generate adult–born neurons in crayfish. Developmental Cell, 30 (3): 322–333.
Christie A E. 2011. Crustacean neuroendocrine systems and their signaling agents. Cell and Tissue Research, 345 (1): 41–67.
Cooke I M, Sullivan R E. 1982. Hormones and neurosecretion. In: Bliss D E, Atwood H L, Sandeman D C eds. The Biology of Crustacea. Academic Press, New York. p.205–290.
de la Paz Rodríguez–Muñoz M, Escamilla–Chimal E G. 2015. Glial fibrillary acidic protein (GFAP) shows circadian oscillations in crayfish Procambarus clarkii putative pacemakers. Chronobiology International, 32: 1 135–1 144.
Fernández–Hernández I, Rhiner C, Moreno E. 2013. Adult Neurogenesis in Drosophila. Cell Reports, 3 (6): 1 857–1 865.
Ghanawi J, Saoud I P. 2012. Molting, reproductive biology, and hatchery management of redclaw crayfish Cherax quadricarinatus (von Martens 1868). Aquaculture, 358–359: 183–195.
Ghosal K, Gupta M, Killian K A. 2009. Agonistic behavior enhances adult neurogenesis in male Acheta domesticus crickets. Journal of Experimental Biology, 212 (13): 2 045–2 056.
Hansen A, Schmidt M. 2004. Influence of season and environment on adult neurogenesis in the central olfactory pathway of the shore crab, Carcinus maenas. Brain Research, 1025 (1–2): 85–97.
Hartenstein V. 2006. Blood cells and blood cell development in the animal kingdom. Annual Review of Cell and Developmental Biology, 22: 677–712.
Hartenstein V. 2011. Morphological diversity and development of glia in Drosophila. Glia, 59 (9): 1 237–1 252.
Hartenstein V. 2014. From blood to brain: the neurogenic niche of the crayfish brain. Developmental Cell, 30 (3): 253–254.
Ito K, Shinomiya K, Ito M, Armstrong J D, Boyan G, Hartenstein V, Harzsch S, Heisenberg M, Homberg U, Jenett A, Keshishian H, Restifo L L, Rössler W, Simpson J H, Strausfeld N J, Strauss R, Vosshall L B 2014. A systematic nomenclature for the insect brain. Neuron, 81 (4): 755–765.
Jemaa M, Morin N, Cavelier P, Cau J, Strub J M, Delsert C. 2014. Adult somatic progenitor cells and hematopoiesis in oysters. Journal of Experimental Biology, 217 (17): 3 067–3 077.
Jiravanichpaisal P, Söderhäll K, Söderhäll I. 2006. Characterization of white spot syndrome virus replication in in vitro–cultured haematopoietic stem cells of freshwater crayfish, Pacifastacus leniusculus. The Journal of General Virology, 87 (4): 847–854.
Kress T, Harzsch S, Dircksen H. 2016. Neuroanatomy of the optic ganglia and central brain of the water flea Daphnia magna (Crustacea, Cladocera). Cell and Tissue Research, 363 (3): 649–677.
Li X, Erclik T, Bertet C, Chen Z Q, Voutev R, Venkatesh S, Morante J, Celik A, Desplan C. 2013. Temporal patterning of Drosophila medulla neuroblasts controls neural fates. Nature, 498 (7455): 456–462.
Lindsey B W, Tropepe V. 2006. A comparative framework for understanding the biological principles of adult neurogenesis. Progress in Neurobiology, 80 (6): 281–307.
Manfrin C, Tom M, De Moro G, Gerdol M, Giulianini P G, Pallavicini A. 2015. The eyestalk transcriptome of red swamp crayfish Procambarus clarkii. Gene, 557 (1): 28–34.
Maynard D M. 1962. Organization of neuropil. American Zoologist, 2 (1): 79–96.
Saetan J, Senarai T, Tamtin M, Weerachatyanukul W, Chavadej J, Hanna P J, Parhar I, Sobhon P, Sretarugsa P. 2013. Histological organization of the central nervous system and distribution of a gonadotropin–releasing hormonelike peptide in the blue crab, Portunus pelagicus. Cell and Tissue Research, 353 (3): 493–510.
Sandeman D, Sandeman R, Derby C, Schmidt M. 1992. Morphology of the brain of crayfish, crabs, and spiny lobsters: a common nomenclature for homologous structures. The Biological Bulletin, 183 (2): 304–326.
Saver M A, Wilkens J L, Syed N I. 1999. In situ and in vitro identification and characterization of cardiac ganglion neurons in the crab, Carcinus maenas. Journal of Neurophysiology, 81 (6): 2 964–2 976.
Schmidt M, Derby C D. 2011. Cytoarchitecture and ultrastructure of neural stem cell niches and neurogenic complexes maintaining adult neurogenesis in the olfactory midbrain of spiny lobsters, Panulirus argus. The Journal of Comparative Neurology, 519 (12): 2 283–2 319.
Schmidt M, Harzsch S. 1999. Comparative analysis of neurogenesis in the central olfactory pathway of adult decapod crustaceans by in vivo BrdU labeling. The Biological Bulletin, 196 (2): 127–136.
Sintoni S, Benton J L, Beltz B S, Hansson B S, Harzsch S. 2012. Neurogenesis in the central olfactory pathway of adult decapod crustaceans: development of the neurogenic niche in the brains of procambarid crayfish. Neural Development, 7 (1): 1.
Smarandache–Wellmann C R. 2016. Arthropod neurons and nervous system. Current Biology, 26 (20): R960–R965.
Stegner M E J, Richter S. 2011. Morphology of the brain in Hutchinsoniella macracantha (Cephalocarida, Crustacea). Arthropod Structure & Development, 40 (3): 221–243.
Stegner M E J, Stemme T, Iliffe T M, Richter S, Wirkner C S. 2015. The brain in three crustaceans from cavernous darkness. BMC Neuroscience, 16: 19.
Sullivan J M, Beltz B S. 2005. Newborn cells in the adult crayfish brain differentiate into distinct neuronal types. Journal of Neurobiology, 65 (2): 157–170.
Tinikul Y, Poljaroen J, Kornthong N, Chotwiwatthanakun C, Anuracpreeda P, Poomtong T, Hanna P J, Sobhon P. 2011. Distribution and changes of serotonin and dopamine levels in the central nervous system and ovary of the Pacific white shrimp, Litopenaeus vannamei, during ovarian maturation cycle. Cell and Tissue Research, 345 (1): 103–124.
Xu Y, Ye H H, Ma J, Huang H Y, Wang G Z. 2010. Primary culture and characteristic morphologies of neurons from the cerebral ganglion of the mud crab, Scylla paramamosain. In Vitro Cellular & Developmental Biology–Animal, 46 (8): 708–717.
Ye H, Wang J X, Zhang Z C, Jia C X, Schmerberg C, Catherman A D, Thomas P M, Kelleher N L, Li L J. 2015. Defining the Neuropeptidome of the spiny lobster Panulirus interruptus brain using a multidimensional mass spectrometry–based platform. Journal of Proteome Research, 14 (11): 4 776–4 791.
Zhang Y Z, Buchberger A, Muthuvel G, Li L J. 2015. Expression and distribution of neuropeptides in the nervous system of the crab Carcinus maenas and their roles in environmental stress. Proteomics, 15 (23–24): 3 969–3 979.
Zhang Y, Allodi S, Sandeman D C, Beltz B S. 2009. Adult neurogenesis in the crayfish brain: proliferation, migration, and possible origin of precursor cells. Developmental Neurobiology, 69 (7): 415–436.
Acknowledgement
The authors are grateful to Prof. LIU Haipeng for cell culture training. We would like to give sincere thanks to Dr. GAO Yi and Dr. WANG Quanchao who helped to take some pictures in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No. 41376165), the National Natural Science Foundation of China- Israel Science Foundation (NSFC-ISF) (No. 31461143007), and the Scientific and Technological Innovation Project financially supported by Qingdao National Laboratory for Marine Science and Technology (No. 2015ASKJ02)
Rights and permissions
About this article
Cite this article
Duan, H., Jin, S., Li, F. et al. Neuroanatomy and morphological diversity of brain cells from adult crayfish Cherax quadricarinatus. J. Ocean. Limnol. 36, 2368–2378 (2018). https://doi.org/10.1007/s00343-019-7145-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-019-7145-x