Abstract
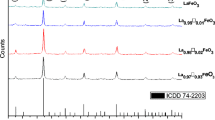
Multiferroics—materials attract considerable attention, due to their fascinating physics properties. The motivating physical properties of GdFe1−xCoxO3 (0.00 ≤ x ≤ 0.25) orthorhombic structure were adjusted in a controlled way by modifying the composition through the synthesis processes. The orthorhombic structure within the space group Pbnm is confirmed through X-ray diffraction patterns. The data reveal that the samples under investigation are G-type antiferromagnetic with a weak ferromagnetic moment. Dzyaloshinskii–Moriya interaction is a crucial source of the observed weak ferromagnetic behavior for the investigated samples. An anisotropy constant for GdFe0.9Co0.1O3 is approximately equal to 4065 emu Oe/gm, which is 9 times greater than that of GdFeO3 samples. The higher values of anisotropic constant K indicate that the prepared samples will be promising and helpful for technological applications at a variety of temperatures. Cobalt doping has a unique advantage as it reduces the switching field distribution and the change of the entropy (ΔS) of the investigated samples. The presence of Gd3+–O2− 2p, Fe3+–O2− 2p and Co–O-2p hybridization stabilizes the ferroelectric distortion causing an improvement in ferroelectric property. The effects of pH value and the contact time on the adsorption progression were studied and optimized to obtain the maximum possible adsorption efficiency of the Gadolinium/cobalt samples. The use of GdFe0.8Co0.2O3 removes 98% of lead from the waste water. The specific advantages of the investigated samples are ease of separation, high adsorption per unit area, low cost as well as recycled with significant efficiency. The investigated samples are recommended to be used as a reusable adsorbent for the highly efficient removal of lead metal ions from aqueous solutions.











Similar content being viewed by others
References
T.W. Kool, Properties of Perovskites and Other Oxides (World Scientific, Singapore, 2010)
M.A. Gilleo, J. Chem. Phys. 24, 6 (1956)
N. Kumar, A. Shukla, N. Kumar, S. Hajra, S. Sahoo, R.N.P. Choudhary, J. Mater. Sci. Mater. Electron. 30, 1919–1926 (2019)
N. Kumar, A. Shukla, Int. J. Modern Phys. B 32, 1840069 (2018)
D. Treves, J. Appl. Phys. 36, 1033 (1965)
N.J.R. Hayes, A.P. Grosvenor, J. Phys.: Condens. Matter 23, 465502 (2011)
N. Kumar, A. Shukla, R.N.P. Choudhary, J. Mater. Sci.: Mater. Electron. 28, 6673–6684 (2017)
N. Kumar, A. Shukla, R.N.P. Choudhary, J. Alloys Compd. 747, 895–904 (2018)
N. Kumara, A. Shuklaa, R.N.P. Choudhary, Prog. Nat. Sci.: Mater. Int. 28, 308–314 (2018)
C. Madhu, M.B. Bellakki, V. Mannivannan, Indian J. Eng. Mater. Sci. 17, 131 (2010)
P.K. Roy, J. Bera, J. Magn. Magn. Mater. 298, 38 (2006)
H. Harzali, F. Saida, A. Marzouki, A. Megriche, F. Baillon, F. Espitalier, A.A. Mgaidi, J. Magn. Magn. Mater. 419, 50–56 (2016)
M. Kumar, H.S. Dosanjh, H. Singh, J. Environ. Chem. Eng. 6, 6194–6206 (2018)
B.D. Cullity, Elements of X-ray Diffraction (Adison-Wesley Publ. Co., London, 1967)
R.D. Shannon, Acta Crystallogr. Sect. A 32, 751 (1976)
C.S. Vandana, B.H. Rundramadevi, Int. J. Res. Appl. Sci. Eng. Technol. (IJRASET) 5, 1999 (2017)
K. Sultan, M. Ikram, K. Asakan, Vacuum 99, 251 (2014)
E.E. Ateia, D.E. El-Nashar, R. Ramadan, M.F. Shokry, J. Inorg. Organomet. Polym Mater. 30, 1041–1048 (2020)
X. Ge, Y. Liu, X. Liu, Sensors Actuators B: Chem. 79, 171 (2001)
S.S. Ata-Allah, A. Hashhash, J. Magn. Magn. Mater. 307, 191 (2006)
V.M. Goldschmidt, Naturwissenchaffen 14, 477 (1926)
E.E. Ateia, A.T. Mohamed, K. Elsayed, J. Magn. Magn. Mater. 452, 169–178 (2018)
E.E. Ateia, A.T. Mohamed, J. Supercond. Novel Magn. 30, 627–633 (2017)
S. Husain, A.O.A. Keelani, W. Khan, Nanostruct. Nanoobjects 15, 17 (2018)
S.S.K. Ma, L. Chen, S. Zhang, J. Lin, S.P. Jiang, Int. J. Hydrog. Energy 38, 13300 (2013)
J. Choi, B. Kim, S.H. Song, J.S. Park, Int. J. Hydrog. Energy 41, 9619 (2016)
Y. Janbutrach, S. Hunpratub, E. Swatsitang, Nanoscale Res. Lett. 9, 498 (2014)
S. Thirumalairajan, K. Girija, V. Ganesh, D. Mangalaraj, C. Viswanathan, N. Ponpandian, Crystal Growth Des. 13, 291 (2013)
M.D. Sturge, E.M. Gyorgy, R.C. LeCraw, J.P. Remeika, Phys. Rev. 180, 413 (1969)
B.J. Rani, M. Ravina, B. Saravanakumar, G. Ravi, V. Ganesh, S. Ravichandran, R. Yuvakkumar, Nanostruct. Nanoobjects 14, 84 (2018)
Recommendations: Pure Appl. Chem. 57 (1985) 603; Recommendations for the Characterization of Porous Solids, IUPAC Commission on Colloid and Surface Chemistry. Pure Appl. Chem. 66 (1994) 1739
P. Paramasivan, P. Venkatesh, J. Supercond. Novel Magn. 29, 2805 (2016)
J. Kanamori, J. Phys. Chem. Solids 10(2–3), 87. Bibcode:1959JPCS…10…87 K. https://doi.org/10.1016/0022-3697(59)90061-7
Y. Wei, H. Gui, Z. Zhao, J. Li, Y. Liu, S. Xin, X. Li, W. Xie, AIP Adv. 4, 127134 (2014)
M.A. Ahmed, S.I. El-Dek, Mater. Sci. Eng. B 128, 30 (2006)
E. Brück, O. Tegus, D.T.C. Thanh, K.H.J. Buschow, J. Magn. Magn. Mater. 310, 2793 (2006)
E.E. Ateia, M.M. Arman, E. Badawy, Appl. Phys. A 125, 499 (2019)
A.A. Azab, E.E. Ateia, S.A. Esmail, Appl. Phys. A 124, 469 (2018)
R. Day, M. Fuller, V.A. Schmidt, Hysteresis properties of titanomagnetites: grain-size and compositional dependence. Phys. Earth Planet. Inter. 13(4), 260–267 (1977)
T. Moriya, Phys. Rev. Lett. 4, 228 (1960)
J.J. Blanco, M. Insausti, I.G. Muro, L. Lezama, T. Rojo, J. Solid State Chem. 179, 623 (2006)
C. Song, Y. You, X. Chen, X. Zhou, Y. Wang, F. Pan, Nanotechnology 29, 112001 (2018)
R.J.M. Van de Veerdonk, X. Wu, D. Weller, IEEE Trans. Magn. 39, 590 (2003)
P. Sharma, P. Saxena, A. Kumar, D. Varshney, J. Alloys Compd. 706, 609 (2017)
N. Kumar, A. Shukla, N. Kumar, S. Sahoo, S. Hajra, R.N.P. Choudhary, Ceram. Int. 44, 21330–21337 (2018)
N. AlokShukla, R.N.P. Choudhary, Phys. Lett. A 381, 2721–2730 (2017)
N. Kumar, A. Shukla, N. Kumar, R.N.P. Choudhary, A. Kumar, RSC Adv. 8, 36939–36950 (2018)
S.K. Pate, B. Kuriachen, N. Kumar, R. Nateriya, Ceram. Int. 44, 6426–6432 (2018)
M.A. Ahmed, S.F. Mansour, M.A. Abdo, Mater. Res. Bull. 48, 1796–1805 (2013)
E.E. Ateia, F.S. Soliman, Mater. Sci. Eng. B 244, 29–37 (2019)
F. Liu, K. Zhou, Q. Chen, A. Wang, W. Chen, J. Alloys Compd. 773, 140 (2019)
E.E. Ateia, R. Ramadan, A.S. Shafaay, Appl. Phys. A 126, 222 (2020)
S. Sobhanardakani, A. Jafari, R. Zandipaka, A. Meidanchi, Process Saf. Environ. Prot. 120, 348–357 (2018)
Acknowledgements
I would like to express my gratitude and appreciation to Ahmed Kamel, physics department, Faculty of Science, Cairo University, for his efforts in ferroelectric measurements.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ateia, E.E., Ramadan, R. & Hussein, B. Studies on multifunctional properties of GdFe1−xCoxO3 multiferroics. Appl. Phys. A 126, 340 (2020). https://doi.org/10.1007/s00339-020-03518-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-020-03518-1