Abstract
Animals are faced with a fundamental risk-reward trade-off when making decisions about foraging in the presence of predation, yet little is known about how social, reproductive and environmental factors mediate this trade-off. In the marine environment, anemonefishes provide a model system for investigating the determinants of risk–reward trade-offs, because they live in size- and sex-structured groups within protective sea anemones tentacles, yet feed predominantly in the water column where they are at risk of predation. Furthermore, exposure to changing tides means the availability of planktonic food covaries with their risk of predation. Therefore, we examined how tide, sex and status, and the presence of eggs influenced the time that Amphiprion mccullochi spent at different distances from their anemone, a proxy for foraging effort and predation risk. We found that individuals significantly adjusted their time spent far and close to the anemone depending on the tide, status and the presence of eggs, and that these adjustments can be explained in light of threat sensitive behaviour. This study illustrates the relative importance of environmental and social factors on intraspecific variation in foraging and antipredator behaviour and bolsters our understanding of the decision’s individuals make to balance the costs and benefits of foraging over temporal and spatial scales.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Predation is a key selective force in the evolution of animal behaviour, shaping numerous behavioural adaptations relating to reproduction, sociality and foraging in a wide range of taxa (Schneider 1984; Lima and Dill 1990; Sih 1994; Khater et al. 2016). With respect to foraging, animals must weigh up the need to obtain food while minimising risks of predation, a decision-making process that is further complicated by the fact that predation pressure can vary spatially and temporally (Schartel and Schauber 2016; Palmer et al. 2017; Kohl et al. 2018). How individuals might balance this risk–reward trade-off, whereby they attempt to maximise their net energy intake, is explained in optimal foraging theory (OFT). This theory was developed to predict optimal diet, foraging path, patch choice and optimal time allocation to different patches (Pyke et al. 1977). While OFT has been instrumental in guiding our understanding of animal foraging, what is less clear is how external factors, such as variability in predation threat, food availability and social behaviour, interconnect and modulate this trade-off, and how those factors may differentially affect individuals based on their traits (e.g. Lima and Dill 1990; Kie 1999; Clement et al. 2016; Donelan and Trussell 2020).
Environmental, social and reproductive factors are likely to influence the trade-off between foraging and predation (Lima and Dill 1990; Kie 1999; Donelan and Trussell 2020). For example, spatial and temporal variability in temperature and predator abundance influences foraging behaviour in a tropical damselfish, which displays reduced foraging when exposed to predators at high but not low temperatures (Beck et al. 2016). Additionally, social context, including social dominance, group size and composition, may modulate the behaviour of individuals (Lima and Dill 1990). For example, dominant birds exclude more subordinate individuals from access to feeding sites closer to their protective shelters, demonstrating a key effect of social rank (Schneider 1984). Further, reproductive factors, such as relative parental investment, have been shown to influence foraging decisions in relation to predation risk (Lima and Dill 1990; Komdeur and Kats 1999). These examples illustrate that individuals of many taxa can respond to shifting environmental, social and reproductive factors when deciding whether or not to forage. Therefore, it is important to examine the relative roles of multiple factors shaping the risk–reward trade-off to explain the degree of behavioural variation in animals and extend existing theory (Drakeley et al. 2015; Catano et al. 2017).
In shallow-water marine environments, tidal cycles and associated changes in hydrodynamic conditions can strongly influence the behaviours exhibited by organisms (Gibson 1992; Kerford et al. 2008; Eggertsen et al. 2016). Crucially, foraging opportunities and predation risk often covary with tides (Eggertsen et al. 2016; Hansen et al. 2016; Udyawer et al. 2020). During high tide, an increase in food due to tidal influx increases planktivore foraging opportunities by fish; however, predation risk is simultaneously heightened because the substrate, a key protective habitat for many benthic fishes, is more readily accessible to predators in deeper water (Rypel et al. 2007; Armstrong et al. 2016; Hansen et al. 2016). High tide can therefore present rich opportunities for maximising energy intake, but also increases exposure to predation, with the opposite being true at low tide. As such, shallow-water marine environments provide an excellent medium for investigating how this risk–reward trade-off may be modulated in response to changes in environmental, social and reproductive factors (Rypel et al. 2007; Hansen et al. 2016).
On coral reefs, anemonefishes (family Pomacentridae) form an obligate symbiosis with sea anemones, yet generally feed in the water column (Fautin and Allen 1992). When residing in their host anemone, they gain protection from predation in the anemone’s nematocyst-laden tentacles, whereas when feeding on planktonic food sources, they potentially become more vulnerable. Anemonefishes are therefore expected to face the classic trade-off between foraging to maximise food intake while balancing the costs of being predated. Threat-sensitive behavioural adjustments in response to altered environmental, social or reproductive contexts could thus be reflected by the time that individuals spend feeding at different distances from the anemone, as has been demonstrated in other taxa (e.g. Schneider 1984; Lima and Dill 1990). The hypothesis being that anemonefishes spending more time closer to their anemones affords greater protection from predators, but reduces opportunities to forage on plankton throughout the water column.
In addition, anemonefishes form social groups that are organised into size-based dominance hierarchies, wherein the largest individual is the female (rank one), the second largest the male (rank two) and all other individuals are smaller non-breeding subordinates (rank three onwards) (Buston 2003a; Buston and Cant 2006). Smaller individuals have been shown to suffer higher mortality (Buston 2003b), presumably because their small size increases probability of death during a predatory encounter, which would therefore potentially modulate the risk–reward trade-off from foraging. Furthermore, anemonefishes lay eggs close to the base of the anemone that are mainly cared for by the male (Fautin and Allen 1992; Barbasch and Buston 2018). While males would presumably have reduced predation risks relative to smaller non-breeders when foraging in the water column, a male’s ability to forage far from the anemone would be reduced even at high tide when food resources are more abundant owing to the need for parental care. On the other hand, female and subordinate anemonefish, being relatively emancipated from parental duties (Buston and Elith 2011; Barbasch and Buston 2018), may be able to forage further from the anemone even when eggs are present, potentially taking greater advantage of elevated food availability at high tides.
To examine how environmental, social and reproductive factors influence the risk–reward trade-off, we compared the time that Amphiprion mccullochi Whitley 1929 spent foraging at different distances from its sea anemone habitat. Specifically, we evaluated the effects of tidal cycles (environmental factor) on foraging-related behaviours, and how the effects of tides might be modulated by social (rank and hence body size) and reproductive (presence of eggs) factors, we compared time spent foraging at different distances from the anemone. We predicted that individuals would be capable of assessing risk and reward, and that the amount of time that individuals spent foraging at different distances from the anemone would be vary in response to the tested variables.
Materials and methods
This study was conducted at Far Flats, Lord Howe Island, New South Wales, Australia (31°31′46″S, 159°04′29″E) between 20 and 28 January 2019. Using SCUBA, nine social groups of A. mccullochi were haphazardly located between 0.2 and 2.5 m depth. Groups contained two breeding adults (breeder female and male) plus three to six non-breeding subordinates (mean and SE group size = 6.1 ± 0.3). All fish resided in clusters of the host sea anemone, Entacmaea quadricolor.
Behavioural observations
Each group was filmed for 20 min each day by three divers across three consecutive days at both low tide (days 1–3, tidal height 0.4–1 m) and high tide (days 7–9, tidal height 1.5–2.5 m). On each occasion, filming was only done in the afternoon to control for any potential diurnal effects on behaviour (Catano et al. 2017). Groups were filmed using GoPro cameras (models: 3 + , 5 and 7) on the same settings (960 resolution, 60 frames s−1 and wide field of view) that were mounted on weighted stainless-steel tripods 50 cm above the substrate. To achieve the same field of view, marked weights were left on the substrate near each group and the tripods were directly placed above the marker during filming. To ensure that filming commenced at the same time before the peak of the tide each day (either low or high), the divers placed video cameras at all groups approximately 10–15 min before the tidal peak. Once video recording commenced, the divers moved at least 10 m away and returned after filming was completed to minimise disturbance.
Footage was viewed using VLC media player and behavioural traits for each focal fish scored by LA. Of the 20 min of video footage, the first 2 min was disregarded to allow the fish to acclimatise to the camera, and the following 10 min was analysed (following Wong et al. 2017). When individuals could not be observed in the field of view and were not within the habitat, additional footage was analysed to try and achieve 10 min of observed behaviour per focal fish. However, if fish were unable to be scored for this duration because they were out of view, the scored behaviours for a given time observed were scaled up to represent 10 min. This occurred during six of the 54 observations and for each there were at least 300 s of recorded behaviour.
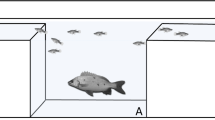
Wherever possible, the behaviour of the breeder female and male (i.e. rank 1 and 2, respectively), and the two smallest non-breeding subordinates were scored per group. Specifically, we scored (i) time spent within habitat (s), (ii) close to habitat and (iii) far from habitat, with time spent within these three zones used as proxies for threat-sensitive foraging behaviour (Fig. 1). Within habitat was defined when a fish was in contact with an anemone or was sheltering within the associated substratum, as sometimes it was not possible to differentiate from the videos whether a fish was in its anemone or just in the associated substrate (Fig. 1a). Time spent close was defined as when an individual was within two total body lengths of an anemone or associated substratum (Fig. 1b). Time spent far from habitat was scored as the time spent greater than two total body lengths away from an anemone or associated substratum (Fig. 1c). The presence of anemonefish eggs was also noted in situ, as well as in each video. In total, five groups had eggs at some point during the study, and one group had eggs for the entire time (Online Resource Tables S1 and S2).
Anemonefish were scored as time spent (a) within habitat; when in contact with an anemone or within the associated substratum (b) close; when within two total body lengths of an anemone or associated substratum and (c) far from habitat; when greater than two total body lengths away from an anemone or associated substratum
Statistical analysis
Time spent in the three habitat zones was studied in response to the effects of three factors of interest: tide, breeding status and egg presence by fitting linear models. Time in each zone is subject to a sum constraint so that more time in one zone must be compensated by less time in the others. This constraint was removed by transformation to log-ratio scale as follows, where in habitat, close to habitat and far from habitat represent time (s) in each zone:
Therefore, a negative close to habitat log-ratio means that less time was spent close than within the habitat, and vice versa for positive log ratio. Similarly, a negative far from habitat log-ratio means that less time was spent far than within the habitat, and vice versa for positive log-ratio. Aitchison (1986) showed that these constructed variables can be studied independently and statistical inference is independent of baseline choice. Furthermore, inverse transformation of estimates back to proportion of time in each zone will obey the sum constraint. Positive values of the transformed variables indicate more time in the alternate zone than in habitat, while negative values indicate more time in habitat. All observed times were incremented by 1 s to accommodate two observations of habitat = 0.
Linear models were fitted to describe each transformed variable in response to tide (high, low), breeding status (male, female, non-breeder), egg presence (yes, no) and all interactions. Group size (number of fish) was added as a covariate and later excluded from the models owing to non-significant effects (most probably due to the limited range of group sizes (five to eight individuals). The models included random effects to accommodate the nested sampling structure of days within groups and the repeat measures on each fish.
An analysis of variance was constructed from each model and terms failing to account for significant variation (F-ratio exceed 5% critical value) were excluded. Estimates of the expected log-ratio for each group of fish as classified by breeding status, egg presence and tide with 95% confidence intervals were obtained from the final models and post hoc statistical comparison between specific pairs of estimates made by Tukey’s test. The estimates were inverse transformed to proportion of time spent in each zone and presented in a ternary plot to aid data visualisation.
The data analysis was conducted in the R environment (R Core Team 2021) with particular use of the packages lme4 (Bates et al. 2015) and emmeans (Lenth 2021) for modelling and estimation.
Results
Time spent close relative to within habitat
A total of eight breeder females, six breeder males and 13 non-breeding subordinates were filmed and scored from nine groups of A. mccullochi (Online Resource Tables S1 and S2).
When examining the ratio of time spent close to habitat relative to within the habitat (i.e. mean log habitat: close), statistically important effects of breeding status, egg presence and the interaction between breeding status and tide were detected on log-preference for time in the close zone (Table 1). These effects were largely driven by all group members spending more time in the habitat when eggs were present versus absent (Figs. 2 and 3; Online Resource Table S3). Additionally, non-breeders in particular spent more time close to the habitat relative to within the habitat at high tide compared to low tide (Figs. 2 and 3; Online Resource Table S3). A likelihood ratio test for the random group variance parameter showed it was not significantly different from zero (p = 0.2).
Time spent far relative to within the habitat
When examining the ratio of time spent far relative to within the habitat (i.e. mean log habitat: far), statistically important effects of breeding status, egg presence and tide were detected on log-preference for time in the far zone (Table 1; Figs. 2 and 4). All individuals spent less time far at low tide compared to high tide, breeder females and males spent more time far than non-breeders, and all group members spent less time far when eggs were present compared to absent (Figs. 2 and 4; Online Resource Table S4). A likelihood ratio test for the random group variance parameter indicated the term was statistically important (p = 0.05) (Table 2).
Discussion
The need to reduce predation risk while maximising foraging efficiency is a central determinant of behaviour in most animals, and to this end, many studies have assessed foraging decisions of animals under predation threat (e.g. Sih 1982; Lima and Dill 1990; Olson et al. 2015). However, relatively few studies have examined the simultaneous effects of social, reproductive and environmental factors (but see Hansen et al. 2016; Catano et al. 2017), and thus our understanding of the factors governing variation in the risk–reward trade-off is limited, especially in the field. Here we show that anemonefish social rank, the presence of eggs and changes in tide altered the time they spent at different distances to the shelter of their host sea anemone. This suggests that risk–reward trade-offs can be altered in response to the interplay of social, reproductive and environmental factors in the marine environment.
In anemonefish groups, breeder females and males are the two largest group members and as a result experience the lowest risks of mortality compared to non-breeders (Buston 2003a). Not surprisingly, we found that breeder females and males spent more time far from the anemone (rather than within it) than non-breeders. Other studies examining foraging in social reef fishes have also found that larger, higher-ranked dominants typically forage further from protective coral habitat than subordinates, and are therefore the first to encounter prey arriving with the current (Coates 1980; Forrester 1991). From the breeder female’s perspective, fecundity is typically size-limited (Wootton 1979), and female reproductive success is related to growth and size (Buston and Elith 2011). Therefore, breeder females would benefit more from foraging further to grow and produce more eggs. Given that the breeder males are the second largest group member, they also experience relatively low predation risk. Hence they can afford to spend more time far relative to within the habitat than their smaller non-breeders. However, males spent slightly less time far away from the habitat than females, which may be related to the fact that male reproductive success is unrelated to male size (Buston and Elith 2011), reducing the benefits to males of riskier foraging. Non-breeders, being the smallest fish within the group and therefore experiencing highest risks of mortality (Buston 2003b), spent the least time far from the habitat. By staying close to and seeking refuge within their anemone, non-breeders reduce predation risk, as demonstrated in many other small-bodied coral reef fishes (Hixon and Beets 1993; Holbrook and Schmitt 2002). Although consistent sheltering may restrict non-breeder growth, these subordinates must regulate their growth to remain smaller than their immediate dominants anyway (Buston 2003a; Buston and Cant 2006). As such, social growth regulation coupled with an elevated risk of predation could account for why non-breeders spent the least time far from the habitat.
All individuals spent less time away from the habitat (both far and close) when eggs were present. Therefore, this key reproductive factor has a strong influence on the risk–reward trade-off for all group members. In anemonefishes (and fishes in general; Gross and Sargent 1985), males provide the majority of parental care which involves substantial tending, mouthing, fanning and guarding of eggs (Green and McCormick 2005; Barbasch and Buston 2018). For example, male A. melanopus spend significantly more time (2–4 times) actively tending to the eggs than females (Green and McCormick 2005). As male reproductive success is not correlated to body size (Buston and Elith 2011), males could further trade-off foraging for growth in favour of parental investment when eggs are present. Conversely, when eggs are absent, males could spend more time away from the anemone feeding in preparation for the next round of paternal duties to enhance future breeding success. As such, the reduced time spent far and close to the habitat (relative to within it) when eggs were present was not surprising for males. For breeder females, the finding that they too spent less time away from the habitat relative to within when eggs were present could indicate that some level of maternal care was being provided, which has been demonstrated for A. melanopus and A. percula (Green and McCormick 2005; Barbasch and Buston 2018). Alternatively, females may potentially assist with protecting clutches of eggs and hence may spend less time away from habitat, although this possibility requires further testing.
On the other hand, the reason why non-breeders spent less time away from the habitat when eggs were present is less intuitive, as anemonefish are not known to exhibit alloparental care (Taborsky and Wong 2017). Furthermore, experimental removal of subordinate non-breeders does not enhance breeder survival, growth or reproductive success (Buston 2004). However, removing subordinate non-breeders confounds both the removal of costs and benefits that subordinates may provide to the dominants (Wong and Balshine 2011), hence the lack of change in dominant fitness may mask any benefits that subordinate actions could be providing dominants. Therefore, it is important to quantify the types of behaviours that subordinates engage in when eggs are present versus absent (e.g. massaging and cleaning the anemone, aggressively chasing or biting heterospecific competitors, potential eggs predators or anemone predators), to conclude whether or not subordinates may be directly or indirectly assisting with care. Alternatively, subordinates could simply have spent less time far when eggs were present because they were copying the behaviours of their larger group members, preferring to stay close to them to enhance group cohesiveness and reap the benefits of doing so (Ioannou et al. 2011, 2017; Paijmans et al. 2019). Furthermore, individuals may display consistency in behavioural traits, as has been shown in other anemonefishes (Wong et al. 2013, 2017; Barbasch & Buston 2018) and in A. mccullochi with respect to boldness and aggression (Wong et al. 2017), which may explain variation in behaviour. However, rank and group size were still important determinants of both these behaviours (Wong et al. 2017), suggesting rank is likely to play a key role. In any case, this study is the first to our knowledge to demonstrate altered subordinate behaviour in the presence of eggs, the underlying reasons need further investigation.
In addition to social and reproductive factors, the environmental factor, tidal cycle, was related to the time spent at different distances from the anemone, although more nuanced. At high tide, breeder females, breeder males and non-breeders spent more time far from habitat (relative to within) compared to low tide. Given that high tide is typically associated with greater predation risk (Motro et al. 2005; Rypel et al. 2007), this suggests that all group members took a risk to forage away from shelter at high tide when presumably more planktonic food was available. When considering time spent close to the habitat, there was no substantial change in breeder females and males, but non-breeders increased the amount of time spent close to the habitat at high tide compared to low tide. This suggests that non-breeders, facing higher predation risk, forage at an optimal rate by leaving but staying close to the anemone at high tide. In contrast to the other group members which foraged at an optimal rate by travelling further from the habitat at high tide. Again, this suggests that status, body size and associated predation risk plays a clear role in modulating the risk–reward trade-off experienced by anemonefishes.
The finding that A. mccullochi shows threat-sensitive behaviour in relation to social status, reproductive and environmental factors, is novel and consistent with other studies investigating tidally-mediated behaviour in coral-reef fishes. Most notably, Hansen et al. (2016) reported that a greater proportion of the coral-dwelling damselfish, Dascyllus aruanus, emerged from their coral following a predation scare during high tide compared to during low tide. This suggests that habitat-specialist reef fishes adjust their behaviour in response to increased foraging opportunities at high tide. Additionally, Hansen et al. (2016) demonstrated that emergence times were influenced by group size, with a greater proportion emerging in larger groups, presumably owing to greater protection afforded from predators. Furthermore, fish have been shown to alter their foraging and antipredator behaviour according to a key temporal environmental rhythm, the diel cycle and time of day (Gibson et al. 1996; Metcalfe et al. 1999; Hindell et al. 2000; Catano et al. 2017), whereas in the current study, any behavioural differences due to time of day was controlled for by only observing fish in the afternoon. Therefore, future research on anemonefish foraging and predation behaviour should explore the concurrent effects of diel and tidal cycles, as well as longer term seasonality effects, for a deeper understanding of the interactions between multiple factors on the risk–reward trade-off.
Animals are confronted with a fundamental risk–reward trade-off between the benefits of foraging to increase fitness versus the cost of predation threat (Pyke et al. 1977). To our knowledge, this is the first study to demonstrate anemonefish as compelling models to examine the influence of tides, social and reproductive factors on the trade-off between foraging and antipredator behaviour. Our findings highlight the importance of the potential and interacting role of multiple factors that may ultimately affect the survival and reproductive success. Considering interindividual variation in behaviour would be an important next step, in addition to the role of varying social conflicts described in detail in anemonefishes (e.g. Buston & Cant 2006; Wong et al. 2016) on the risk–reward trade-off. Understanding these relationships can also help us understand the impacts of living in increasingly stressful habitats, such as under conditions of climate change, on optimal foraging behaviour, which in turn can be invaluable for conservation and management of social species.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Aitchison J (1986) The statistical analysis of compositional data. Chapman and Hall, London
Armstrong AO, Armstrong AJ, Jaine FR, Couturier LI, Fiora K, Uribe-Palomino J, Weeks SJ, Townsend KA, Bennett MB, Richardson AJ (2016) Prey density threshold and tidal influence on reef manta ray foraging at an aggregation site on the Great Barrier Reef. PLoS ONE 11:e0153393
Barbasch TA, Buston PM (2018) Plasticity and personality of parental care in the clown anemonefish. Anim Behav 136:65–73
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Beck HJ, Feary DA, Fowler AM, Madin EMP, Booth DJ (2016) Temperate predators and seasonal water temperatures impact feeding of a range expanding tropical fish. Mar Biol 163:70
Buston PM (2003a) Social hierarchies: size and growth modification in clownfish. Nature 424:145–146
Buston PM (2003b) Mortality is associated with social rank in the clown anemonefish (Amphiprion percula). Mar Biol 143:811–815
Buston PM (2004) Does the presence of non-breeders enhance the fitness of breeders? An experimental analysis in the clown anemonefish Amphiprion percula. Behav Ecol Sociobiol 57:27–31
Buston PM, Cant MA (2006) A new perspective on size hierarchies in nature: patterns, causes, and consequences. Oecologia 149:362–372
Buston PM, Elith J (2011) Determinants of reproductive success in dominant pairs of clownfish: a boosted regression tree analysis. J Anim Ecol 80:528–538
Catano L, Boswell BM, Burkepile D (2017) Predator identity and time of day interact to shape the risk–reward trade-off for herbivorous coral reef fishes. Oecologia 183:763–773
Clement HS, Tambling CJ, Kerley GIH (2016) Prey morphology and predator sociality drive predator prey preferences. J Mammal 97:919–927
Coates D (1980) Prey-size intake in humbug damselfish, Dascyllus aruanus (Pisces, Pomacentridae) living within social groups. J Anim Ecol 49:335–340
Donelan SC, Trussell GC (2020) Sex-specific differences in the response of prey to predation risk. Funct Ecol 34:1235–1243
Drakeley M, Lapiedra O, Kolbe JJ (2015) Predation risk perception, food density and conspecific cues shape foraging decisions in a tropical lizard. PLoS ONE 10:e0138016
Eggertsen L, Hammar L, Gullström M (2016) Effects of tidal current-induced flow on reef fish behaviour and function on a subtropical rocky reef. Mar Ecol Prog Ser 559:175–192
Fautin DG, Allen GR (1992) Anemonefishes and their host sea anemones. Western Australian Museum, Perth
Forrester GE (1991) Social rank, individual size and group composition as determinants of food consumption by humbug damselfish, Dascyllus aruanus. Anim Behav 42:701–711
Gibson RN (1992) Tidally-synchronised behaviour in marine fishes. Ali MA(eds) Rhythms in fishes. Springer, Boston, pp 63–81
Gibson RN, Robb L, Burrows MT, Ansell AD (1996) Tidal, diel and longer term changes in the distribution of fishes on a Scottish sandy beach. Mar Ecol Prog Ser 130:1–17
Green BS, McCormick MI (2005) O2 replenishment to fish nests: males adjust brood care to ambient conditions and brood development. Behav Ecol 16:389–397
Gross MR, Sargent RC (1985) The evolution of male and female parental care in fishes. Am Zool 25:807–822
Hansen MJ, Morrell LJ, Ward AJW (2016) The effect of temporally variable environmental stimuli and group size on emergence behavior. Behav Ecol 27(3):939–945. https://doi.org/10.1093/beheco/arv237
Hindell JS, Jenkins GP, Keough MJ (2000) Variability in abundances of fishes associated with seagrass habitats in relation to diets of predatory fishes. Mar Biol 136:725–737
Hixon MA, Beets JP (1993) Predation, prey refuges, and the structure of coral-reef fish assemblages. Ecol Monogr 63:77–101
Holbrook SJ, Schmitt RJ (2002) Competition for shelter space causes density-dependent predation mortality in damselfishes. Ecology 83:2855–2868
Ioannou CC, Couzin ID, James R, Croft DP, Krause J (2011) Social organisation and information transfer in schooling fish. Pitcher TJ, Brown C, Laland K, Krause J(eds) Fish cognition and behavior. Blackwell Publishing, New Jersey, pp 217–239
Ioannou CC, Ramnarine IW, Torney CJ (2017) High-predation habitats affect the social dynamics of collective exploration in a shoaling fish. Sci Adv 3:e1602682
Kerford MR, Wirsing AJ, Heithaus MR, Dill LM (2008) Danger on the rise: diurnal tidal state mediates an exchange of food for safety by the bar-bellied sea snake Hydrophis elegans. Mar Ecol Prog Ser 358:289–294
Khater M, Murariu D, Gras R (2016) Predation risk tradeoffs in prey: effects on energy and behaviour. Theor Ecol 9:251–268
Kie JG (1999) Optimal foraging and risk of predation: effects on behavior and social structure in ungulates. J Mammal 80:1114–1129
Komdeur J, Kats RKH (1999) Predation risk affects trade-off between nest guarding and foraging in Seychelles warblers. Behav Ecol 10:648–658
Kohl MT, Stahler DR, Metz MC, Forester JD, Kauffman MJ, White PJ, Smith DW, MacNulty DR (2018) Diel predator activity drives a dynamic landscape of fear. Ecol Monogr 88:638–652
Lenth RV (2021) emmeans: estimated marginal means, aka least-squares means. R package version 1.6.0. https://CRAN.R-project.org/package=emmeans
Lima S, Dill L (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640
Metcalfe N, Fraser N, Burns M (1999) Food availability and the nocturnal vs diurnal foraging trade-off in juvenile salmon. J Anim Ecol 68:371–381
Motro R, Ayalon I, Genin A (2005) Near-bottom depletion of zooplankton over coral reefs: III: vertical gradient of predation pressure. Coral Reefs 24:95–98
Olson RS, Haley PB, Dyer FC, Adami C (2015) Exploring the evolution of a trade-off between vigilance and foraging in group-living organisms. R Soc Open Sci. 2:150135
Palmer MS, Fieberg J, Swanson A, Kosmala M, Packer C (2017) A ‘dynamic’ landscape of fear: prey responses to spatiotemporal variations in predation risk across the lunar cycle. Ecol Lett 20:1364–1373
Paijmans KP, Booth DJ, Wong MYL (2019) Towards an ultimate explanation for mixed species shoaling. Fish Fish 20:921–933
Pyke GH, Pulliam HR, Charnov EL (1977) Optimal foraging: A selective review of theory and tests. Q Rev Biol 52:137–154
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rypel AL, Layman CA, Arrington DA (2007) Water depth modifies relative predation risk for a motile fish taxon in bahamian tidal creeks. Estuar Coast 30:518–525
Schartel TE, Schauber EM (2016) Relative preference and localized food affect predator space use and consumption of incidental prey. PLoS ONE 11:e0151483
Schneider KJ (1984) Dominance, predation, and optimal foraging in white-throated sparrow flocks. Ecol 65:1820–1827
Sih A (1982) Foraging strategies and the avoidance of predation by an aquatic insect. Ecol 63:786–796
Sih A (1994) Predation risk and the evolutionary ecology of reproductive behaviour. J Fish Biol 45:111–130
Taborsky M, Wong MYL (2017) Sociality in fishes. Rubenstein D, Abbott P(eds) Comparative social evolution. Cambridge University Press, Cambridge, United Kingdom, pp 354–389
Udyawer V, Goiran C, Chateau O, Shine R (2020) Swim with the tide: Tactics to maximize prey detection by a specialist predator, the greater sea snake (Hydrophis major). PLoS ONE 15:e0239920
Wong MYL, Balshine S (2011) The evolution of cooperative breeding in the African cichlid fish, Neolamprologus pulcher. Biol Rev 86:511–530
Wong MYL, Medina A, Uppaluri C, Arnold S, Seymour JR, Buston PM (2013) Consistent behavioural traits and behavioural syndromes in pairs of the false clown anemonefish Amphiprion ocellaris. J Fish Biol 83:207–213
Wong MYL, Uppaluri C, Medina A, Seymour JR, Buston PM (2016) The four elements of within-group conflict in animal societies: an experimental test using the clown anemonefish, Amphiprion percula. Behav Ecol Sociobiol 70:1467–1475
Wong MYL, Beasley AL, Douglass T, Whalan S, Scott A (2017) Some anemonefish lack personality: a comparative assessment of behavioral variation and repeatability in relation to environmental and social factors. Coral Reefs 36:1307–1316
Wootton RJ (1979) Energy costs of egg production and environmental determinants of fecundity in teleost fishes. In: Miller PJ (ed) Fish phenology. Academic Press, New York, pp 133–159
Acknowledgements
We thank Lord Howe Island Marine Park staff (S. Gudge, K. Maguire and C. Woods) and R. and L. Byrne for their assistance getting to and from the site each day. This work was supported by a Southern Cross University Marine Ecology Research Grant to AS and a Centre for Sustainable Ecosystems grant from the University of Wollongong awarded to MYLW. All procedures were approved by UOW Animal Ethics Committee under protocol number 18/06.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topic Editor Andrew Hoey
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wong, M.Y.L., Aitken, L.R., Beasley, A.L. et al. Environmental, social and reproductive factors influence the risk–reward trade-off in a group-living fish. Coral Reefs 41, 463–471 (2022). https://doi.org/10.1007/s00338-022-02240-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-022-02240-z