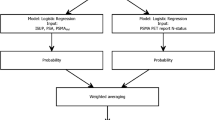
Abstract
Objective
To test the ability of high-performance machine learning (ML) models employing clinical, radiological, and radiomic variables to improve non-invasive prediction of the pathological status of prostate cancer (PCa) in a large, single-institution cohort.
Methods
Patients who underwent multiparametric MRI and prostatectomy in our institution in 2015–2018 were considered; a total of 949 patients were included. Gradient-boosted decision tree models were separately trained using clinical features alone and in combination with radiological reporting and/or prostate radiomic features to predict pathological T, pathological N, ISUP score, and their change from preclinical assessment. Model behavior was analyzed in terms of performance, feature importance, Shapley additive explanation (SHAP) values, and mean absolute error (MAE). The best model was compared against a naïve model mimicking clinical workflow.
Results
The model including all variables was the best performing (AUC values ranging from 0.73 to 0.96 for the six endpoints). Radiomic features brought a small yet measurable boost in performance, with the SHAP values indicating that their contribution can be critical to successful prediction of endpoints for individual patients. MAEs were lower for low-risk patients, suggesting that the models find them easier to classify. The best model outperformed (p ≤ 0.0001) clinical baseline, resulting in significantly fewer false negative predictions and overall was less prone to under-staging.
Conclusions
Our results highlight the potential benefit of integrative ML models for pathological status prediction in PCa. Additional studies regarding clinical integration of such models can provide valuable information for personalizing therapy offering a tool to improve non-invasive prediction of pathological status.
Clinical relevance statement
The best machine learning model was less prone to under-staging of the disease. The improved accuracy of our pathological prediction models could constitute an asset to the clinical workflow by providing clinicians with accurate pathological predictions prior to treatment.
Key Points
• Currently, the most common strategies for pre-surgical stratification of prostate cancer (PCa) patients have shown to have suboptimal performances.
• The addition of radiological features to the clinical features gave a considerable boost in model performance. Our best model outperforms the naïve model, avoiding under-staging and resulting in a critical advantage in the clinic.
•Machine learning models incorporating clinical, radiological, and radiomics features significantly improved accuracy of pathological prediction in prostate cancer, possibly constituting an asset to the clinical workflow.






Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- AI:
-
Artificial intelligence
- AUC:
-
Area under the curve
- cN:
-
Clinical N stage
- cT:
-
Clinical T stage
- EPE:
-
Extraprostatic extension
- GBDT:
-
Gradient-boosted decision tree
- GS:
-
Gleason Score
- ISUP :
-
International Society of Urological Pathology
- MAE:
-
Mean absolute error
- MCC:
-
Matthews correlation coefficient
- ML:
-
Machine learning
- mp-MRI:
-
Multiparametric magnetic resonance imaging
- PCa:
-
Prostate cancer
- PI-RADS:
-
Prostate Imaging Reporting and Data System
- pN:
-
Pathological N stage
- Post-op:
-
Post-operative
- PSA:
-
Prostate-specific antigen
- pT:
-
Pathological T stage
- RP:
-
Radical prostatectomy
- RT:
-
Radiation therapy
- SHAP:
-
SHapley Additive exPlanations
References
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Sung H, Ferlay J, Siegel RL et al (2021) Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Hamdy FC, Donovan JL, Lane JA et al (2016) 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med 375:1415–1424. https://doi.org/10.1056/NEJMoa1606220
Mottet N, van den Bergh RCN, Briers E et al (2021) EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol 79:243–262. https://doi.org/10.1016/j.eururo.2020.09.042
Schaeffer E, An Y, Barocas D et al (2022) NCCN Guidelines Version 1.2023 Prostate Cancer. In: Available from: https://www.nccn.org/home/. Accessed 1 Mar 2023
Rodrigues G, Warde P, Pickles T et al (2012) Pre-treatment risk stratification of prostate cancer patients: a critical review. Can Urol Assoc J 6:121–127. https://doi.org/10.5489/cuaj.11085
Stolzenbach LF, Rosiello G, Pecoraro A et al (2020) Prostate cancer grade and stage misclassification in active surveillance candidates: Black versus White patients. J Natl Compr Canc Netw 18:1492–1499. https://doi.org/10.6004/jnccn.2020.7580
Yang DD, Mahal BA, Muralidhar V et al (2019) Risk of upgrading and upstaging among 10 000 patients with Gleason 3+4 favorable intermediate-risk prostate cancer. Eur Urol Focus 5:69–76. https://doi.org/10.1016/j.euf.2017.05.011
Daskivich TJ, Wood LN, Skarecky D et al (2017) Limitations of the National Comprehensive Cancer Network® (NCCN®) Guidelines for Prediction of Limited Life Expectancy in Men with Prostate Cancer. J Urol 197:356–362. https://doi.org/10.1016/j.juro.2016.08.096
Martin NE, Chen M-H, Zhang D et al (2017) Unfavorable intermediate-risk prostate cancer and the odds of upgrading to gleason 8 or higher at prostatectomy. Clin Genitourin Cancer 15:237–241. https://doi.org/10.1016/j.clgc.2016.06.001
Sorce G, Flammia RS, Hoeh B et al (2022) Grade and stage misclassification in intermediate unfavorable-risk prostate cancer radiotherapy candidates. Prostate 82:1040–1050. https://doi.org/10.1002/pros.24349
Epstein JI, Egevad L, Amin MB et al (2016) The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Definition of Grading Patterns and Proposal for a New Grading System. Am J Surg Pathol 40 244–252. https://doi.org/10.1097/PAS.0000000000000530
Ahmed HU, El-Shater Bosaily A, Brown LC et al (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389:815–822. https://doi.org/10.1016/S0140-6736(16)32401-1
Weinreb JC, Barentsz JO, Choyke PL et al (2016) PI-RADS Prostate Imaging - Reporting and Data System: 2015, Version 2. Eur Urol 69:16–40. https://doi.org/10.1016/j.eururo.2015.08.052
Wibmer AG, Chaim J, Lakhman Y et al (2021) Oncologic outcomes after localized prostate cancer treatment: associations with pretreatment prostate magnetic resonance imaging findings. J Urol 205:1055–1062. https://doi.org/10.1097/JU.0000000000001474
Hassanzadeh E, Glazer DI, Dunne RM et al (2017) Prostate imaging reporting and data system version 2 (PI-RADS v2): a pictorial review. Abdom Radiol (NY) 42:278–289. https://doi.org/10.1007/s00261-016-0871-z
Kasivisvanathan V, Rannikko AS, Borghi M et al (2018) MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med 378:1767–1777. https://doi.org/10.1056/NEJMoa1801993
Algohary A, Viswanath S, Shiradkar R et al (2018) Radiomic features on MRI enable risk categorization of prostate cancer patients on active surveillance: preliminary findings. J Magn Reson Imaging. https://doi.org/10.1002/jmri.25983
Alessi S, Pricolo P, Summers P et al (2019) Low PI-RADS assessment category excludes extraprostatic extension (≥pT3a) of prostate cancer: a histology-validated study including 301 operated patients. Eur Radiol 29:5478–5487. https://doi.org/10.1007/s00330-019-06092-0
Barentsz JO, Richenberg J, Clements R et al (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22:746–757. https://doi.org/10.1007/s00330-011-2377-y
Chaddad A, Kucharczyk MJ, Niazi T (2018) Multimodal radiomic features for the predicting Gleason score of prostate cancer. Cancers (Basel) 10:E249. https://doi.org/10.3390/cancers10080249
Mazzone E, Stabile A, Pellegrino F et al (2021) Positive predictive value of Prostate Imaging Reporting and Data System Version 2 for the detection of clinically significant prostate cancer: a systematic review and meta-analysis. Eur Urol Oncol 4:697–713. https://doi.org/10.1016/j.euo.2020.12.004
Grimm P, Billiet I, Bostwick D et al (2012) Comparative analysis of prostate-specific antigen free survival outcomes for patients with low, intermediate and high risk prostate cancer treatment by radical therapy. Results from the Prostate Cancer Results Study Group: CANCER CONTROL RATES: COMPARISON OF TREATMENT OPTIONS. BJU Int 109:22–29. https://doi.org/10.1111/j.1464-410X.2011.10827.x
Artibani W, Porcaro AB, De Marco V et al (2018) Management of biochemical recurrence after primary curative treatment for prostate cancer: a review. Urol Int 100:251–262. https://doi.org/10.1159/000481438
Kornberg Z, Cooperberg MR, Spratt DE, Feng FY (2018) Genomic biomarkers in prostate cancer. Transl Androl Urol 7:459–471. https://doi.org/10.21037/tau.2018.06.02
Eggener SE, Rumble RB, Beltran H (2020) Molecular biomarkers in localized prostate cancer: ASCO Guideline Summary. JCO Oncol Pract 16:340–343. https://doi.org/10.1200/JOP.19.00752
Gaudreau P-O, Stagg J, Soulières D, Saad F (2016) The present and future of biomarkers in prostate cancer: proteomics, genomics, and immunology advancements. Biomark Cancer 8:15–33. https://doi.org/10.4137/BIC.S31802
Xu M, Fang M, Zou J et al (2019) Using biparametric MRI radiomics signature to differentiate between benign and malignant prostate lesions. Eur J Radiol 114:38–44. https://doi.org/10.1016/j.ejrad.2019.02.032
Zhou Y, Yuan J, Xue C et al (2022) A pilot study of MRI radiomics for high-risk prostate cancer stratification in 1.5 T MR-guided radiotherapy. Magn Reson Med. https://doi.org/10.1002/mrm.29564
Gaudiano C, Mottola M, Bianchi L et al (2022) Beyond multiparametric MRI and towards radiomics to detect prostate cancer: a machine learning model to predict clinically significant lesions. Cancers (Basel) 14:6156. https://doi.org/10.3390/cancers14246156
Woźnicki P, Westhoff N, Huber T et al (2020) Multiparametric MRI for prostate cancer characterization: combined use of radiomics model with PI-RADS and clinical parameters. Cancers (Basel) 12:1767. https://doi.org/10.3390/cancers12071767
Zhang L, Zhe X, Tang M et al (2021) Predicting the grade of prostate cancer based on a biparametric MRI radiomics signature. Contrast Media Mol Imaging 2021:7830909. https://doi.org/10.1155/2021/7830909
Dong D, Tang L, Li Z-Y et al (2019) Development and validation of an individualized nomogram to identify occult peritoneal metastasis in patients with advanced gastric cancer. Ann Oncol 30:431–438. https://doi.org/10.1093/annonc/mdz001
Lambin P, Rios-Velazquez E, Leijenaar R et al (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446. https://doi.org/10.1016/j.ejca.2011.11.036
Bourbonne V, Vallières M, Lucia F et al (2019) MRI-derived radiomics to guide post-operative management for high-risk prostate cancer. Front Oncol 9:807. https://doi.org/10.3389/fonc.2019.00807
Abdollahi H, Mofid B, Shiri I et al (2019) Machine learning-based radiomic models to predict intensity-modulated radiation therapy response, Gleason score and stage in prostate cancer. Radiol Med 124:555–567. https://doi.org/10.1007/s11547-018-0966-4
Jia Y, Quan S, Ren J et al (2022) MRI radiomics predicts progression-free survival in prostate cancer. Front Oncol 12:974257. https://doi.org/10.3389/fonc.2022.974257
Ferro M, de Cobelli O, Musi G et al (2022) Radiomics in prostate cancer: an up-to-date review. Ther Adv Urol 14:17562872221109020. https://doi.org/10.1177/17562872221109020
Pirrone G, Matrone F, Chiovati P et al (2022) Predicting local failure after partial prostate re-irradiation using a dosiomic-based machine learning model. J Pers Med 12:1491. https://doi.org/10.3390/jpm12091491
Esteva A, Feng J, van der Wal D et al (2022) Prostate cancer therapy personalization via multi-modal deep learning on randomized phase III clinical trials. NPJ Digit Med 5:71. https://doi.org/10.1038/s41746-022-00613-w
Amin MB, Greene FL, Edge SB et al (2017) The Eighth Edition AJCC Cancer Staging Manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin 67:93–99. https://doi.org/10.3322/caac.21388
Turkbey B, Rosenkrantz AB, Haider MA et al (2019) Prostate Imaging Reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur Urol 76:340–351. https://doi.org/10.1016/j.eururo.2019.02.033
Scialpi M, Martorana E, Scialpi P et al (2021) MRI apparent diffusion coefficient (ADC): a biomarker for prostate cancer after radiation therapy. Turk J Urol 47:448–451. https://doi.org/10.5152/tud.2021.21274
Isaksson LJ, Repetto M, Summers PE et al (2023) High-performance prediction models for prostate cancer radiomics. Inform Med Unlocked 101161. https://doi.org/10.1016/j.imu.2023.101161
Ahdoot M, Wilbur AR, Reese SE et al (2020) MRI-targeted, systematic, and combined biopsy for prostate cancer diagnosis. N Engl J Med 382:917–928. https://doi.org/10.1056/NEJMoa1910038
Chen M, Zhang Q, Zhang C et al (2019) Combination of 68Ga-PSMA PET/CT and multiparametric MRI improves the detection of clinically significant prostate cancer: a lesion-by-lesion analysis. J Nucl Med 60:944–949. https://doi.org/10.2967/jnumed.118.221010
Rouvière O, Puech P, Renard-Penna R et al (2019) Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): a prospective, multicentre, paired diagnostic study. Lancet Oncol 20:100–109. https://doi.org/10.1016/S1470-2045(18)30569-2
Luzzago S, de Cobelli O, Mistretta FA et al (2021) MRI-targeted or systematic random biopsies for prostate cancer diagnosis in biopsy naïve patients: follow-up of a PRECISION trial-like retrospective cohort. Prostate Cancer Prostatic Dis 24:406–413. https://doi.org/10.1038/s41391-020-00290-4
Luzzago S, Colombo A, Mistretta FA et al (2023) Multiparametric MRI-based 5-year risk prediction model for biochemical recurrence of prostate cancer after radical prostatectomy. Radiology 309:e223349. https://doi.org/10.1148/radiol.223349
Drost F-JH, Osses D, Nieboer D et al (2020) Prostate magnetic resonance imaging, with or without magnetic resonance imaging-targeted biopsy, and systematic biopsy for detecting prostate cancer: a Cochrane systematic review and meta-analysis. Eur Urol 77:78–94. https://doi.org/10.1016/j.eururo.2019.06.023
Rehman A, El-Zaatari ZM, Han SH et al (2020) Seminal vesicle invasion combined with extraprostatic extension is associated with higher frequency of biochemical recurrence and lymph node metastasis than seminal vesicle invasion alone: Proposal for further pT3 prostate cancer subclassification. Ann Diagn Pathol 49:151611. https://doi.org/10.1016/j.anndiagpath.2020.151611
Kim W, Kim CK, Park JJ et al (2017) Evaluation of extracapsular extension in prostate cancer using qualitative and quantitative multiparametric MRI. J Magn Reson Imaging 45:1760–1770. https://doi.org/10.1002/jmri.25515
Krishna S, Lim CS, McInnes MDF et al (2018) Evaluation of MRI for diagnosis of extraprostatic extension in prostate cancer. J Magn Reson Imaging 47:176–185. https://doi.org/10.1002/jmri.25729
Park KJ, Kim M-H, Kim JK (2020) Extraprostatic tumor extension: comparison of preoperative multiparametric MRI criteria and histopathologic correlation after radical prostatectomy. Radiology 296:87–95. https://doi.org/10.1148/radiol.2020192133
Bloch BN, Genega EM, Costa DN et al (2012) Prediction of prostate cancer extracapsular extension with high spatial resolution dynamic contrast-enhanced 3-T MRI. Eur Radiol 22:2201–2210. https://doi.org/10.1007/s00330-012-2475-5
Somford DM, Hamoen EH, Fütterer JJ et al (2013) The predictive value of endorectal 3 Tesla multiparametric magnetic resonance imaging for extraprostatic extension in patients with low, intermediate and high risk prostate cancer. J Urol 190:1728–1734. https://doi.org/10.1016/j.juro.2013.05.021
Matsuoka Y, Ishioka J, Tanaka H et al (2017) Impact of the Prostate Imaging Reporting and Data System, Version 2, on MRI diagnosis for extracapsular extension of prostate cancer. AJR Am J Roentgenol 209:W76–W84. https://doi.org/10.2214/AJR.16.17163
Caglic I, Kovac V, Barrett T (2019) Multiparametric MRI - local staging of prostate cancer and beyond. Radiol Oncol 53:159–170. https://doi.org/10.2478/raon-2019-0021
de Rooij M, Hamoen EHJ, Witjes JA et al (2016) Accuracy of magnetic resonance imaging for local staging of prostate cancer: a diagnostic meta-analysis. Eur Urol 70:233–245. https://doi.org/10.1016/j.eururo.2015.07.029
Briganti A, Blute ML, Eastham JH et al (2009) Pelvic lymph node dissection in prostate cancer. Eur Urol 55:1251–1265. https://doi.org/10.1016/j.eururo.2009.03.012
Michael J, Neuzil K, Altun E, Bjurlin MA (2022) Current opinion on the use of magnetic resonance imaging in staging prostate cancer: a narrative review. Cancer Manag Res 14:937–951. https://doi.org/10.2147/CMAR.S283299
Rajinikanth A, Manoharan M, Soloway CT et al (2008) Trends in Gleason score: concordance between biopsy and prostatectomy over 15 years. Urology 72:177–182. https://doi.org/10.1016/j.urology.2007.10.022
Kvåle R, Møller B, Wahlqvist R et al (2009) Concordance between Gleason scores of needle biopsies and radical prostatectomy specimens: a population-based study. BJU Int 103:1647–1654. https://doi.org/10.1111/j.1464-410X.2008.08255.x
Perera M, Mirchandani R, Papa N et al (2021) PSA-based machine learning model improves prostate cancer risk stratification in a screening population. World J Urol 39:1897–1902. https://doi.org/10.1007/s00345-020-03392-9
Zhong X, Cao R, Shakeri S et al (2019) Deep transfer learning-based prostate cancer classification using 3 Tesla multi-parametric MRI. Abdom Radiol (NY) 44:2030–2039. https://doi.org/10.1007/s00261-018-1824-5
Zhang B, Lian Z, Zhong L et al (2020) Machine-learning based MRI radiomics models for early detection of radiation-induced brain injury in nasopharyngeal carcinoma. BMC Cancer 20:502. https://doi.org/10.1186/s12885-020-06957-4
Filson CP, Natarajan S, Margolis DJA et al (2016) Prostate cancer detection with magnetic resonance-ultrasound fusion biopsy: the role of systematic and targeted biopsies. Cancer 122:884–892. https://doi.org/10.1002/cncr.29874
Park SY, Jung DC, Oh YT et al (2016) Prostate cancer: PI-RADS version 2 helps preoperatively predict clinically significant cancers. Radiology 280:108–116. https://doi.org/10.1148/radiol.16151133
Palisaar JR, Noldus J, Löppenberg B et al (2012) Comprehensive report on prostate cancer misclassification by 16 currently used low-risk and active surveillance criteria. BJU Int 110:E172-181. https://doi.org/10.1111/j.1464-410X.2012.10935.x
Isaksson L, Pepa M, Summers P et al (2022) Comparison of automated segmentation techniques for magnetic resonance images of the prostate. BMC Med Imaging 23(1):32. https://doi.org/10.21203/rs.3.rs-1850296/v1
Isaksson LJ, Summers P, Bhalerao A et al (2022) Quality assurance for automatically generated contours with additional deep learning. Insights Imaging 13:137. https://doi.org/10.1186/s13244-022-01276-7
Saha A, Twilt JJ, Bosma JS et al (2022) Artificial intelligence and radiologists at prostate cancer detection in MRI: the PI-CAI challenge (study protocol). https://doi.org/10.5281/zenodo.6522364
Tolkach Y, Kristiansen G (2018) The heterogeneity of prostate cancer: a practical approach. Pathobiology 85:108–116. https://doi.org/10.1159/000477852
Jafari-Khouzani K, Paynabar K, Hajighasemi F, Rosen B (2019) Effect of region of interest size on the repeatability of quantitative brain imaging biomarkers. IEEE Trans Biomed Eng 66:864–872. https://doi.org/10.1109/TBME.2018.2860928
Jensen LJ, Kim D, Elgeti T et al (2021) Stability of radiomic features across different region of interest sizes-a CT and MR phantom study. Tomography 7:238–252. https://doi.org/10.3390/tomography7020022
Acknowledgements
JLI is a Ph.D. student within the European School of Molecular Medicine (SEMM) in Milan, Italy. MGV was supported by a research fellowship from the Associazione Italiana per la Ricerca sul Cancro (AIRC) entitled “Radioablation ± hormonotherapy for prostate cancer oligorecurrences (RADIOSA trial): potential of imaging and biology” registered at ClinicalTrials.gov NCT03940235, approved by the Ethics Committee of IRCCS Istituto Europeo di Oncologia and Centro Cardiologico Monzino (IEO-997). GC was partially supported by a research fellowship from Accuray Inc. IEO, the European Institute of Oncology, is partially supported by the Italian Ministry of Health (with “Ricerca Corrente” and “5x1000” funds).
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Giulia Marvaso.
Conflict of interest
Division of Radiotherapy IEO received research funding from AIRC (Italian Association for Cancer Research), Fondazione IEO-CCM (Istituto Europeo di Oncologia-Centro Cardiologico Monzino) all outside the current project. BAJF received speakers fees from Roche, Bayer, Janssen, Carl Zeiss, Ipsen, Accuray, Astellas, Elekta, and IBA Astra Zeneca (all outside the current project). The remaining authors declare no conflicts of interest that are relevant to the content of this article.
Statistics and biometry
One of the authors (Sara Gandini) has significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
The study complies with the principles of the Declaration of Helsinki and was approved by the Ethical Committee of IEO-European Institute of Oncology IRCSS and Cardiologic Centre Monzino IRCCS (Notification No. UID 2438).
Study subjects or cohorts overlap
None.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Giulia Marvaso and Lars Johannes Isaksson are co-first authors.
Giuseppe Petralia and Barbara Alicja Jereczek-Fossa are co-last authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marvaso, G., Isaksson, L.J., Zaffaroni, M. et al. Can we predict pathology without surgery? Weighing the added value of multiparametric MRI and whole prostate radiomics in integrative machine learning models. Eur Radiol (2024). https://doi.org/10.1007/s00330-024-10699-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00330-024-10699-3