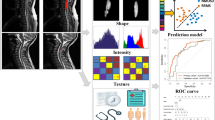
Abstract
Objective
To develop a discrimination pipeline concerning both radiomics and spatial distribution features of brain lesions for discrimination of multiple sclerosis (MS), aquaporin-4-IgG-seropositive neuromyelitis optica spectrum disorder (NMOSD), and myelin-oligodendrocyte-glycoprotein-IgG-associated disorder (MOGAD).
Methods
Hyperintensity T2 lesions were delineated in 212 brain MRI scans of MS (n = 63), NMOSD (n = 87), and MOGAD (n = 45) patients. To avoid the effect of fixed training/test dataset sampling when developing machine learning models, patients were allocated into 4 sub-groups for cross-validation. For each scan, 351 radiomics and 27 spatial distribution features were extracted. Three models, i.e., multi-lesion radiomics, spatial distribution, and joint models, were constructed using random forest and logistic regression algorithms for differentiating: MS from the others (MS models) and MOGAD from NMOSD (MOG-NMO models), respectively. Then, the joint models were combined with demographic characteristics (i.e., age and sex) to create MS and MOG-NMO discriminators, respectively, based on which a three-disease discrimination pipeline was generated and compared with radiologists.
Results
For classification of both MS-others and MOG-NMO, the joint models performed better than radiomics or spatial distribution model solely. The MS discriminator achieved AUC = 0.909 ± 0.027 and bias-corrected C-index = 0.909 ± 0.027, and the MOG-NMO discriminator achieved AUC = 0.880 ± 0.064 and bias-corrected C-index = 0.883 ± 0.068. The three-disease discrimination pipeline differentiated MS, NMOSD, and MOGAD patients with 75.0% accuracy, prominently outperforming the three radiologists (47.6%, 56.6%, and 66.0%).
Conclusions
The proposed pipeline integrating multi-lesion radiomics and spatial distribution features could effectively differentiate MS, NMOSD, and MOGAD.
Clinical relevance statement
The discrimination pipeline merging both radiomics and spatial distribution features of brain lesions may facilitate the differential diagnoses of multiple sclerosis, neuromyelitis optica spectrum disorder, and myelin-oligodendrocyte-glycoprotein-IgG-associated disorder.
Key Points
• Our study introduces an approach by combining radiomics and spatial distribution models.
• The joint model exhibited superior performance in distinguishing multiple sclerosis from aquaporin-4-IgG-seropositive neuromyelitis optica spectrum disorder and myelin-oligodendrocyte-glycoprotein-IgG-associated disorder as well as discriminating the latter two diseases.
• The three-disease discrimination pipeline showcased remarkable accuracy, surpassing the performance of experienced radiologists, highlighting its potential as a valuable diagnostic tool.





Similar content being viewed by others
Abbreviations
- AQP4-IgG:
-
Aquaporin-4-immunoglobulin-G
- IBSI:
-
Imaging biomarker standardization initiative
- MOGAD:
-
Myelin-oligodendrocyte-glycoprotein-IgG-associated disorder
- MS:
-
Multiple sclerosis
- NMOSD:
-
Neuromyelitis optica spectrum disorder
- RIL:
-
The Radiomics Index for Lesion
- RIS:
-
The Radiomics Index for Scan
- SDI:
-
The Spatial Distribution Index
References
Chawla S, Kister I, Wuerfel J et al (2016) Iron and non-iron-related characteristics of multiple sclerosis and neuromyelitis optica lesions at 7T MRI. AJNR Am J Neuroradiol 37:1223–1230
Sinnecker T, Schumacher S, Mueller K et al (2016) MRI phase changes in multiple sclerosis vs neuromyelitis optica lesions at 7T. Neurol Neuroimmunol Neuroinflamm 3:e259
Wegner C (2013) Recent insights into the pathology of multiple sclerosis and neuromyelitis optica. Clin Neurol Neurosurg 115:S38–S41
Rotstein D, Montalban X (2019) Reaching an evidence-based prognosis for personalized treatment of multiple sclerosis. Nat Rev Neurol 15:287–300
Palace J, Leite MI, Nairne A, Vincent A (2010) Interferon beta treatment in neuromyelitis optica. Arch Neurol 67:1016–1017
Sechi E, Cacciaguerra L, Chen JJ et al (2022) Myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD): a review of clinical and MRI features, diagnosis, and management. Front Neurol 13:885218
Reindl M, Waters P (2019) Myelin oligodendrocyte glycoprotein antibodies in neurological disease. Nat Rev Neurol 15:89–102
Jarius S, Paul F, Aktas O et al (2018) MOG encephalomyelitis: international recommendations on diagnosis and antibody testing. J Neuroinflammation 15:134
Matthews L, Marasco R, Jenkinson M et al (2013) Distinction of seropositive NMO spectrum disorder and MS brain lesion distribution. Neurology 80:1330–1337
Juryńczyk M, Tackley G, Kong Y et al (2017) Brain lesion distribution criteria distinguish MS from AQP4-antibody NMOSD and MOG-antibody disease. J Neurol Neurosurg Psychiatry 88:132–136
Hyun J-W, Huh S-Y, Shin H-J et al (2019) Evaluation of brain lesion distribution criteria at disease onset in differentiating MS from NMOSD and MOG-IgG-associated encephalomyelitis. Mult Scler 25:585–590
Juryńczyk M, Geraldes R, Probert F et al (2017) Distinct brain imaging characteristics of autoantibody-mediated CNS conditions and multiple sclerosis. Brain 140:617–627
Cacciaguerra L, Meani A, Mesaros S et al (2019) Brain and cord imaging features in neuromyelitis optica spectrum disorders. Ann Neurol 85:371–384
Cai M-T, Zheng Y, Shen C-H et al (2021) Evaluation of brain and spinal cord lesion distribution criteria at disease onset in distinguishing NMOSD from MS and MOG antibody-associated disorder. Mult Scler 27:871–882
Patel J, Pires A, Derman A et al (2022) Development and validation of a simple and practical method for differentiating MS from other neuroinflammatory disorders based on lesion distribution on brain MRI. J Clin Neurosci 101:32–36
Cortese R, Prados Carrasco F, Tur C et al (2023) Differentiating multiple sclerosis from AQP4-neuromyelitis optica spectrum disorder and MOG-antibody disease with imaging. Neurology 100:e308–e323
Filippi M, Preziosa P, Banwell BL et al (2019) Assessment of lesions on magnetic resonance imaging in multiple sclerosis: practical guidelines. Brain 142:1858–1875
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762
Ma X, Zhang L, Huang D et al (2019) Quantitative radiomic biomarkers for discrimination between neuromyelitis optica spectrum disorder and multiple sclerosis. J Magn Reson Imaging 49:1113–1121
Luo X, Piao S, Li H et al (2022) Multi-lesion radiomics model for discrimination of relapsing-remitting multiple sclerosis and neuropsychiatric systemic lupus erythematosus. Eur Radiol 32:5700–5710
Yang L, Li H, Xia W et al (2020) Quantitative brain lesion distribution may distinguish MOG-ab and AQP4-ab neuromyelitis optica spectrum disorders. Eur Radiol 30:1470–1479
Thompson AJ, Banwell BL, Barkhof F et al (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17:162–173
Wingerchuk DM, Banwell B, Bennett JL et al (2015) International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 85:177–189
Ashrafinia S (2019) Quantitative nuclear medicine imaging using advanced image reconstruction and radiomics. Johns Hopkins University, Baltimore, US. Available via https://jscholarship.library.jhu.edu/handle/1774.2/61551. Accessed 20 Feb 2022
Zwanenburg A, Vallieres M, Abdalah MA et al (2020) The Image Biomarker Standardization Initiative: standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 295:328–338
Zhang Y, Moore GW, Laule C et al (2013) Pathological correlates of magnetic resonance imaging texture heterogeneity in multiple sclerosis. Ann Neurol 74:91–99
Sinnecker T, Dörr J, Pfueller CF et al (2012) Distinct lesion morphology at 7-T MRI differentiates neuromyelitis optica from multiple sclerosis. Neurology 79:708–714
Sinnecker T, Clarke MA, Meier D et al (2019) Evaluation of the central vein sign as a diagnostic imaging biomarker in multiple sclerosis. JAMA Neurol 76:1446–1456
Jeong IH, Choi JY, Kim S-H et al (2016) Comparison of myelin water fraction values in periventricular white matter lesions between multiple sclerosis and neuromyelitis optica spectrum disorder. Mult Scler 22:1616–1620
Ito S, Mori M, Makino T, Hayakawa S, Kuwabara S (2009) “Cloud-like enhancement” is a magnetic resonance imaging abnormality specific to neuromyelitis optica. Ann Neurol 66:425–428
Kaneko K, Sato DK, Nakashima I et al (2018) CSF cytokine profile in MOG-IgG+ neurological disease is similar to AQP4-IgG+ NMOSD but distinct from MS: a cross-sectional study and potential therapeutic implications. J Neurol Neurosurg Psychiatry 89:927–936
Chen X, Zeng C, Luo T et al (2012) Iron deposition of the deep grey matter in patients with multiple sclerosis and neuromyelitis optica: a control quantitative study by 3D-enhanced susceptibility-weighted angiography (ESWAN). Eur J Radiol 81:e633–e639
Cooray GK, Sundgren M, Brismar T (2020) Mechanism of visual network dysfunction in relapsing-remitting multiple sclerosis and its relation to cognition. Clin Neurophysiol 131:361–367
Höftberger R, Guo Y, Flanagan EP et al (2020) The pathology of central nervous system inflammatory demyelinating disease accompanying myelin oligodendrocyte glycoprotein autoantibody. Acta Neuropathol 139:875–892
Amiry-Moghaddam M, Ottersen OP (2003) The molecular basis of water transport in the brain. Nat Rev Neurosci 4:991–1001
Hinson SR, Pittock SJ, Lucchinetti CF et al (2007) Pathogenic potential of IgG binding to water channel extracellular domain in neuromyelitis optica. Neurology 69:2221–2231
Takai Y, Misu T, Suzuki H et al (2021) Staging of astrocytopathy and complement activation in neuromyelitis optica spectrum disorders. Brain 144:2401–2415
Brunner C, Lassmann H, Waehneldt TV, Matthieu J-M, Linington C (1989) Differential ultrastructural localization of myelin basic protein, myelin/oligodendroglial glycoprotein, and 2′,3′-cyclic nucleotide 3′-phosphodiesterase in the CNS of adult rats. J Neurochem 52:296–304
Fujimori J, Takai Y, Nakashima I et al (2017) Bilateral frontal cortex encephalitis and paraparesis in a patient with anti-MOG antibodies. J Neurol Neurosurg Psychiatry 88:534–536
Funding
This work has received funding from the National Natural Science Foundation of China (82102132, 81771296, 82171341, 82102016, 82202122, 8237071280), the Science and Technology Commission of Shanghai Municipality (20S31904300, 22TS1400900, 23S31904100, 22ZR1409500), and the Greater Bay Area Institute of Precision Medicine (Guangzhou) (KCH2310094).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Daoying Geng.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Approval was obtained from Huashan Hospital Institutional Review Board.
Study subjects or cohorts overlap
Some study subjects or cohorts have been previously reported published in two studies: https://doi.org/10.1007/s00330-019-06506-z, and https://doi.org/10.1007/s00330-022-08653-2.
Methodology
• retrospective
• cross sectional study
• performed at one institution
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, X., Li, H., Xia, W. et al. Joint radiomics and spatial distribution model for MRI-based discrimination of multiple sclerosis, neuromyelitis optica spectrum disorder, and myelin-oligodendrocyte-glycoprotein-IgG-associated disorder. Eur Radiol (2023). https://doi.org/10.1007/s00330-023-10529-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00330-023-10529-y