Abstract
Objectives
To evaluate the improvement of mammography interpretation for novice and experienced radiologists assisted by two commercial AI software.
Methods
We compared the performance of two AI software (AI-1 and AI-2) in two experienced and two novice readers for 200 mammographic examinations (80 cancer cases). Two reading sessions were conducted within 4 weeks. The readers rated the likelihood of malignancy (range, 1–7) and the percentage probability of malignancy (range, 0–100%), with and without AI assistance. Differences in AUROC, sensitivity, and specificity were analyzed.
Results
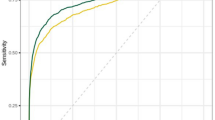
Mean AUROC increased in both novice (0.86 to 0.90 with AI-1 [p = 0.005]; 0.91 with AI-2 [p < 0.001]) and experienced readers (0.87 to 0.92 with AI-1 [p < 0.001]; 0.90 with AI-2 [p = 0.004]). Sensitivities increased from 81.3 to 88.8% with AI-1 (p = 0.027) and to 91.3% with AI-2 (p = 0.005) in novice readers, and from 81.9 to 90.6% with AI-1 (p = 0.001) and to 87.5% with AI-2 (p = 0.016) in experienced readers. Specificity did not decrease significantly in both novice (p > 0.999, both) and experienced readers (p > 0.999 with AI-1 and 0.282 with AI-2). There was no significant difference in the performance change depending on the type of AI software (p > 0.999).
Conclusion
Commercial AI software improved the diagnostic performance of both novice and experienced readers. The type of AI software used did not significantly impact performance changes. Further validation with a larger number of cases and readers is needed.
Clinical relevance statement
Commercial AI software effectively aided mammography interpretation irrespective of the experience level of human readers.
Key Points
• Mammography interpretation remains challenging and is subject to a wide range of interobserver variability.
• In this multi-reader study, two commercial AI software improved the sensitivity of mammography interpretation by both novice and experienced readers. The type of AI software used did not significantly impact performance changes.
• Commercial AI software may effectively support mammography interpretation irrespective of the experience level of human readers.




Similar content being viewed by others
Abbreviations
- AI:
-
Artificial intelligence
- AUROC:
-
Area under the receiver operating characteristics curve
- CAD:
-
Computer-aided detection
- CI:
-
Confidence interval
- FFDM:
-
Full-field digital mammography
- GEE:
-
Generalized estimating equation
- ICC:
-
Intraclass correlation coefficient
- LOM:
-
Likelihood of malignancy
- NPV:
-
Negative predictive value
- PPV:
-
Positive predictive value
- SD:
-
Standard deviation
References
World Health Organization (2015) IARC handbooks. Breast cancer screening, vol 15. International Agency for Research on Cancer, Lyon
Mook S, Van ’t Veer LJ, Rutgers EJ et al (2011) Independent prognostic value of screen detection in invasive breast cancer. J Natl Cancer Inst 103:585–597
Lehtimäki T, Lundin M, Linder N et al (2011) Long-term prognosis of breast cancer detected by mammography screening or other methods. Breast Cancer Res 13:R134
Siu AL (2016) Screening for breast cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann Intern Med 164:279–296
Cardoso F, Kyriakides S, Ohno S et al (2019) Early breast cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up†. Ann Oncol 30:1194–1220
Hamashima C, Hamashima CC, Hattori M et al (2016) The Japanese Guidelines for Breast Cancer Screening. Jpn J Clin Oncol 46:482–492
Hong S, Song SY, Park B et al (2020) Effect of digital mammography for breast cancer screening: a comparative study of more than 8 million Korean women. Radiology 294:247–255
Perry N, Broeders M, de Wolf C, Törnberg S, Holland R, von Karsa L (2008) European guidelines for quality assurance in breast cancer screening and diagnosis. Fourth edition–summary document. Ann Oncol 19:614–622
Lehman CD, Wellman RD, Buist DS, Kerlikowske K, Tosteson AN, Miglioretti DL (2015) Diagnostic accuracy of digital screening mammography with and without computer-aided detection. JAMA Intern Med 175:1828–1837
Cole EB, Zhang Z, Marques HS, Edward Hendrick R, Yaffe MJ, Pisano ED (2014) Impact of computer-aided detection systems on radiologist accuracy with digital mammography. AJR Am J Roentgenol 203:909–916
Rodriguez-Ruiz A, Krupinski E, Mordang JJ et al (2019) Detection of breast cancer with mammography: effect of an artificial intelligence support system. Radiology 290:305–314
Schaffter T, Buist DSM, Lee CI et al (2020) Evaluation of combined artificial intelligence and radiologist assessment to interpret screening mammograms. JAMA Netw Open 3:e200265
Kim HE, Kim HH, Han BK et al (2020) Changes in cancer detection and false-positive recall in mammography using artificial intelligence: a retrospective, multireader study. Lancet Digital Health 2:E138–E148
Lee JH, Kim KH, Lee EH et al (2022) Improving the performance of radiologists using artificial intelligence-based detection support software for mammography: a multi-reader study. Korean J Radiol 23:505–516
McKinney SM, Sieniek M, Godbole V et al (2020) International evaluation of an AI system for breast cancer screening. Nature 577:89–94
Rodriguez-Ruiz A, Lang K, Gubern-Merida A et al (2019) Stand-alone artificial intelligence for breast cancer detection in mammography: comparison with 101 radiologists. J Natl Cancer Inst 111:916–922
Salim M, Wåhlin E, Dembrower K et al (2020) External evaluation of 3 commercial artificial intelligence algorithms for independent assessment of screening mammograms. JAMA Oncol 6:1581–1588
Rawashdeh MA, Lee WB, Bourne RM et al (2013) Markers of good performance in mammography depend on number of annual readings. Radiology 269:61–67
Miglioretti DL, Gard CC, Carney PA et al (2009) When radiologists perform best: the learning curve in screening mammogram interpretation. Radiology 253:632–640
Elmore JG, Jackson SL, Abraham L et al (2009) Variability in interpretive performance at screening mammography and radiologists’ characteristics associated with accuracy. Radiology 253:641–651
Sohns C, Angic B, Sossalla S, Konietschke F, Obenauer S (2010) Computer-assisted diagnosis in full-field digital mammography–results in dependence of readers experiences. Breast J 16:490–497
Hupse R, Samulski M, Lobbes MB et al (2013) Computer-aided detection of masses at mammography: interactive decision support versus prompts. Radiology 266:123–129
Choi JS, Han BK, Ko EY, Kim GR, Ko ES, Park KW (2019) Comparison of synthetic and digital mammography with digital breast tomosynthesis or alone for the detection and classification of microcalcifications. Eur Radiol 29:319–329
Obuchowski NA, Bullen JA (2019) Statistical considerations for testing an AI algorithm used for prescreening lung CT images. Contemp Clin Trials Commun 16:100434
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Oppong BA, Dash C, O’Neill S et al (2018) Breast density in multiethnic women presenting for screening mammography. Breast J 24:334–338
Freer PE (2015) Mammographic breast density: impact on breast cancer risk and implications for screening. Radiographics 35:302–315
Conant EF, Barlow WE, Herschorn SD et al (2019) Association of digital breast tomosynthesis vs digital mammography with cancer detection and recall rates by age and breast density. JAMA Oncol 5:635–642
Phi XA, Tagliafico A, Houssami N, Greuter MJW, de Bock GH (2018) Digital breast tomosynthesis for breast cancer screening and diagnosis in women with dense breasts - a systematic review and meta-analysis. BMC Cancer 18:380
Weigel S, Heindel W, Heidrich J, Hense HW, Heidinger O (2017) Digital mammography screening: sensitivity of the programme dependent on breast density. Eur Radiol 27:2744–2751
Cheung YC, Lin YC, Wan YL et al (2014) Diagnostic performance of dual-energy contrast-enhanced subtracted mammography in dense breasts compared to mammography alone: interobserver blind-reading analysis. Eur Radiol 24:2394–2403
Sardanelli F, Cozzi A, Trimboli RM, Schiaffino S (2020) Gadolinium retention and breast MRI screening: more harm than good? AJR Am J Roentgenol 214:324–327
Sechopoulos I (2013) A review of breast tomosynthesis. Part I. The image acquisition process. Med Phys 40:014301
Kim EK, Kim HE, Han K et al (2018) Applying Data-driven imaging biomarker in mammography for breast cancer screening: preliminary study. Sci Rep 8:2762
Kim HJ, Kim HH, Kim KH et al (2022) Mammographically occult breast cancers detected with AI-based diagnosis supporting software: clinical and histopathologic characteristics. Insights Imaging 13:57
Partyka L, Lourenco AP, Mainiero MB (2014) Detection of mammographically occult architectural distortion on digital breast tomosynthesis screening: initial clinical experience. AJR Am J Roentgenol 203:216–222
Yi A, Chang JM, Shin SU et al (2019) Detection of noncalcified breast cancer in patients with extremely dense breasts using digital breast tomosynthesis compared with full-field digital mammography. Br J Radiol 92:20180101
Cho KR, Seo BK, Woo OH et al (2016) Breast cancer detection in a screening population: comparison of digital mammography, computer-aided detection applied to digital mammography and breast ultrasound. J Breast Cancer 19:316–323
Murakami R, Kumita S, Tani H et al (2013) Detection of breast cancer with a computer-aided detection applied to full-field digital mammography. J Digit Imaging 26:768–773
Sadaf A, Crystal P, Scaranelo A, Helbich T (2011) Performance of computer-aided detection applied to full-field digital mammography in detection of breast cancers. Eur J Radiol 77:457–461
Acknowledgements
The authors thank Min-Ju Kim from the Department of Clinical Epidemiology and Biostatistics at Asan Medical Center for providing statistical consultation.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Woo Jung Choi.
Conflict of interest
All authors declare no competing interests.
Statistics and biometry
Min-Ju Kim from the Department of Clinical Epidemiology and Biostatistics at Asan Medical Center kindly provided statistical advice for this manuscript.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained (Asan Medical Center, approval no. 2020–0281).
Study subjects or cohorts overlap
No study subjects or cohorts have been previously reported.
Methodology
• retrospective
• observational study
• performed at one institution
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The work originated at Asan Medical Center, University of Ulsan College of Medicine, 88, Olympic-ro, 43-gil, Songpa-gu, Seoul 05505, South Korea.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, H.J., Choi, W.J., Gwon, H.Y. et al. Improving mammography interpretation for both novice and experienced readers: a comparative study of two commercial artificial intelligence software. Eur Radiol (2023). https://doi.org/10.1007/s00330-023-10422-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00330-023-10422-8