Abstract
Objectives
To evaluate the clinical value of contrast-enhanced ultrasound (CEUS) with time-intensity curve (TIC) in distinguishing different types of hypovascular solid pancreatic lesions.
Methods
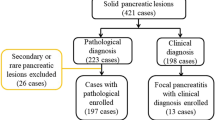
A total of 89 patients with 90 pancreatic lesions (all confirmed by surgery or biopsy pathology) that manifested hypoenhancement on contrast-enhanced ultrasound (CEUS) were included in this study. Six peak enhancement patterns were proposed for differentiating hypovascular pancreatic lesions. CEUS qualitative and TIC-based quantitative parameters were analyzed, and each lesion was scored based on the statistically significant qualitative parameters to evaluate the diagnostic ability of CEUS for hypovascular solid pancreatic lesions.
Results
Qualitative parameters such as peak enhancement pattern II/III/IV, penetrating vessels, centripetal enhancement, and early washout were reliable indicators of malignant lesions, and lesions scored based on these qualitative parameters, with a score ≥ 2, were highly suspected to be malignant lesions. Pattern I had an accuracy of 83.33% for predicting mass-forming pancreatitis (MFP), pattern V had an accuracy of 96.67% for predicting solid pseudopapillary tumors of the pancreas (SPTP), and pattern VI had an accuracy of 81.11% for predicting neuroendocrine tumors/carcinomas (NETs/NECs). For quantitative analysis, nodule/pancreatic parenchyma echo intensity reduction ratio was significantly greater in malignant lesions.
Conclusions
CEUS qualitative and TIC-based quantitative parameters have clinical value in distinguishing malignant from benign hypovascular pancreatic lesions.
Key Points
• Contrast-enhanced ultrasound helps clinicians assess patients with pancreatic lesions.
• Six peak enhancement patterns are proposed for differentiating pancreatic hypovascular lesions.
• Qualitative parameters such as peak enhancement pattern II/III/IV, penetrating vessels, centripetal enhancement, early washout, and quantitative parameter nodule/pancreatic parenchyma echo intensity reduction ratio were important characteristics to discriminate malignant from hypovascular benign lesions.






Similar content being viewed by others
Data Availability
The datasets analyzed in this study are available from the corresponding author on reasonable request.
Abbreviations
- ACC:
-
Acinic cell carcinoma
- ASCA:
-
Adenosquamous carcinoma
- Met:
-
Metastasis
- MFP:
-
Mass-forming pancreatitis
- NEC:
-
Neuroendocrine carcinoma
- NET:
-
Neuroendocrine tumor
- PDAC:
-
Pancreatic ductal adenocarcinoma
- SPTP:
-
Solid pseudopapillary tumors of the pancreas
References
Grossberg AJ, Chu LC, Deig CR et al (2020) Multidisciplinary standards of care and recent progress in pancreatic ductal adenocarcinoma. CA Cancer J Clin 70:375–403
Stathis A, Moore MJ (2010) Advanced pancreatic carcinoma: current treatment and future challenges. Nat Rev Clin Oncol 7:163–172
Fan Z, Li Y, Yan K et al (2013) Application of contrast-enhanced ultrasound in the diagnosis of solid pancreatic lesions–a comparison of conventional ultrasound and contrast-enhanced CT. Eur J Radiol 82:1385–1390
D’Onofrio M, Malagò R, Zamboni G et al (2005) Contrast-enhanced ultrasonography better identifies pancreatic tumor vascularization than helical CT. Pancreatology 5:398–402
Sidhu PS, Cantisani V, Dietrich CF et al (2018) The EFSUMB Guidelines and Recommendations for the Clinical Practice of Contrast-Enhanced Ultrasound (CEUS) in Non-Hepatic Applications: Update 2017 (Long Version). Ultraschall Med 39:e2–e44
Kersting S, Konopke R, Kersting F et al (2009) Quantitative perfusion analysis of transabdominal contrast-enhanced ultrasonography of pancreatic masses and carcinomas. Gastroenterology 137:1903–1911
Kersting S, Roth J, Bunk A (2011) Transabdominal contrast-enhanced ultrasonography of pancreatic cancer. Pancreatology 2:20–27
Takeshima K, Kumada T, Toyoda H et al (2005) Comparison of IV contrast-enhanced sonography and histopathology of pancreatic cancer. AJR Am J Roentgenol 185:1193–1200
Wang Y, Li G, Yan K et al (2022) Clinical value of contrast-enhanced ultrasound enhancement patterns for differentiating solid pancreatic lesions. Eur Radiol 32:2060–2069
Parmar D, Apte M (2021) Angiopoietin inhibitors: a review on targeting tumor angiogenesis. Eur J Pharmacol 899:174021
Kitano M, Kudo M, Maekawa K et al (2004) Dynamic imaging of pancreatic diseases by contrast enhanced coded phase inversion harmonic ultrasonography. Gut 53:854–859
Wang Y, Yan K, Fan Z, Sun L, Wu W, Yang W (2016) Contrast-enhanced ultrasonography of pancreatic carcinoma: correlation with pathologic findings. Ultrasound Med Biol 42:891–898
Imaoka H, Shimizu Y, Mizuno N et al (2014) Ring-enhancement pattern on contrast-enhanced CT predicts adenosquamous carcinoma of the pancreas: a matched case-control study. Pancreatology 14:221–226
Cros J, Raffenne J, Couvelard A, Poté N (2018) Tumor heterogeneity in pancreatic adenocarcinoma. Pathobiology 85:64–71
Kosmahl M, Pauser U, Anlauf M, Klöppel G (2005) Pancreatic ductal adenocarcinomas with cystic features: neither rare nor uniform. Mod Pathol 18:1157–1164
Yoon S, Byun J, Kim K et al (2010) Pancreatic ductal adenocarcinoma with intratumoral cystic lesions on MRI: correlation with histopathological findings. Br J Radiol 83:318–326
Canzonieri V, Berretta M, Buonadonna A et al (2003) Solid pseudopapillary tumour of the pancreas. Lancet Oncol 4:255–256
Kosmahl M, Pauser U, Peters K et al (2004) Cystic neoplasms of the pancreas and tumor-like lesions with cystic features: a review of 418 cases and a classification proposal. Virchows Arch 445:168–178
Liu BA, Li ZM, Su ZS, She XL (2010) Pathological differential diagnosis of solid-pseudopapillary neoplasm and endocrine tumors of the pancreas. World J Gastroenterol 16:1025–1030
Choi JY, Kim MJ, Kim JH et al (2006) Solid pseudopapillary tumor of the pancreas: typical and atypical manifestations. AJR Am J Roentgenol 187:W178–W186
Cantisani V, Mortele KJ, Levy A et al (2003) MR imaging features of solid pseudopapillary tumor of the pancreas in adult and pediatric patients. AJR Am J Roentgenol 181:395–401
Anil G, Zhang J, Al Hamar NE, Nga ME (2017) Solid pseudopapillary neoplasm of the pancreas: CT imaging features and radiologic-pathologic correlation. Diagn Interv Radiol 23:94–99
Xu M, Li XJ, Zhang XE et al (2019) Application of contrast-enhanced ultrasound in the diagnosis of solid pseudopapillary tumors of the pancreas: imaging findings compared with contrast-enhanced computed tomography. J Ultrasound Med 38:3247–3255
Kawamoto S, Johnson PT, Shi C et al (2013) Pancreatic neuroendocrine tumor with cystlike changes: evaluation with MDCT. AJR Am J Roentgenol 200:283–290
Goh BK, Ooi LL, Tan YM et al (2006) Clinico-pathological features of cystic pancreatic endocrine neoplasms and a comparison with their solid counterparts. Eur J Surg Oncol 32:553–556
Karmazanovsky G, Belousova E, Schima W, Glotov A, Kalinin D, Kriger A (2019) Nonhypervascular pancreatic neuroendocrine tumors: spectrum of MDCT imaging findings and differentiation from pancreatic ductal adenocarcinoma. Eur J Radiol 110:66–73
Hyodo R, Suzuki K, Ogawa H, Komada T, Naganawa S (2015) Pancreatic neuroendocrine tumors containing areas of iso- or hypoattenuation in dynamic contrast-enhanced computed tomography: Spectrum of imaging findings and pathological grading. Eur J Radiol 84:2103–2109
Jupp J, Fine D, Johnson CD (2010) The epidemiology and socioeconomic impact of chronic pancreatitis. Best Pract Res Clin Gastroenterol 24:219–231
Choi SY, Kim SH, Kang TW, Song KD, Park HJ, Choi YH (2016) Differentiating mass-forming autoimmune pancreatitis from pancreatic ductal adenocarcinoma on the basis of contrast-enhanced MRI and DWI findings. AJR Am J Roentgenol 206:291–300
D’Onofrio M, Zamboni G, Tognolini A et al (2006) Mass-forming pancreatitis: value of contrast-enhanced ultrasonography. World J Gastroenterol 12:4181–4184
D’Onofrio M, Gallotti A, Principe F, Mucelli RP (2010) Contrast-enhanced ultrasound of the pancreas. World J Radiol 2:97–102
Jia H, Li J, Huang W, Lin G (2021) Multimodel magnetic resonance imaging of mass-forming autoimmune pancreatitis: differential diagnosis with pancreatic ductal adenocarcinoma. BMC Med Imaging 21:149
Matsubara H, Itoh A, Kawashima H et al (2011) Dynamic quantitative evaluation of contrast-enhanced endoscopic ultrasonography in the diagnosis of pancreatic diseases. Pancreas 40:1073–1079
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Fang Nie.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., Nie, F., Dong, T. et al. Role of contrast-enhanced ultrasound with time-intensity curve analysis for differentiating hypovascular solid pancreatic lesions. Eur Radiol 33, 4885–4894 (2023). https://doi.org/10.1007/s00330-023-09393-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-023-09393-7