Abstract
Objectives
To determine the concordance of dynamic contrast-enhanced (DCE) imaging findings with clinico-pathologic characteristics and their prognostic impact for predicting biochemical recurrence (BCR) in patients who underwent radical prostatectomy (RP) for prostate cancer.
Methods
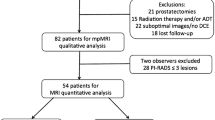
This retrospective study included patients who underwent MRI within 1 year after RP between November 2019 and October 2020. DCE findings and their concordance with the presence and location of positive surgical margin (PSM) were assessed using RP specimens. Kaplan–Meier and logistic regression analyses were used to evaluate the prognostic impact of DCE findings for BCR.
Results
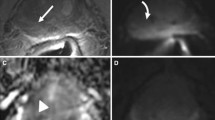
Among the 272 men (mean age ± standard deviation, 66.6 ± 7.4 years), focal nodular enhancement was more frequently observed in those with PSM compared to those with negative margin (85.4% versus 14.6%; p < 0.001). The sites of focal nodular enhancement were 72.9% (35/48) concordant with the PSM locations. Focal nodular enhancement was associated with a higher Gleason score, higher preoperative PSA (≥ 10 ng/mL), higher Gleason grade at the surgical margin, and non-limited margin involvement (p = 0.002, 0.006, 0.032, and 0.001, respectively). In patients without BCR at the time of MRI, focal nodular enhancement was associated with a shorter time to BCR (p < 0.001) and a significant factor predicting 1-year BCR in both univariate (odds ratio = 8.4 [95% CI: 2.5–28.3]; p = 0.001) and multivariate (odds ratio = 5.49 [1.56–19.3]; p = 0.008) analyses.
Conclusions
Focal nodular enhancement on post-prostatectomy MRI was associated with adverse clinico-pathologic characteristics of high risk for recurrence and can be a predictor for 1-year BCR in patients undergoing RP.
Key Points
• Focal nodular enhancement (PI-RR DCE score ≥ 4) was 72.9% (35/48) concordant with the site of positive resection margin by radiologic-histologic correlation.
• Focal nodular enhancement (PI-RR DCE score ≥ 4) was associated with higher Gleason score ( ≥ 8), preoperative PSA ( > 10 ng/mL), and Gleason grade 4 or 5 at the surgical margin and non-limited margin involvement (p ≤ 0.032).
• In patients without BCR at the time of MRI, focal nodular enhancement was a significant factor predicting 1-year BCR (odds ratio = 5.49; 95% CI: 1.56–19.3; p = 0.008).




Similar content being viewed by others
Abbreviations
- BCR:
-
Biochemical recurrence
- DCE:
-
Dynamic contrast enhancement
- EPE:
-
Extraprostatic extension
- GS:
-
Gleason score
- PI-RR:
-
Prostate Imaging for Recurrence Reporting
- PSA:
-
Prostate-specific antigen
- PSM:
-
Positive surgical margin
- RP:
-
Radical prostatectomy
- RT:
-
Radiation treatment
References
Freedland SJ, Humphreys EB, Mangold LA et al (2005) Risk of prostate cancer–specific mortality following biochemical recurrence after radical prostatectomy. JAMA 294:433–439
Roehl KA, Han M, Ramos CG, Antenor JAV, Catalona WJ (2004) Cancer progression and survival rates following anatomical radical retropubic prostatectomy in 3,478 consecutive patients: Long-term results. J Urol 172:910–914
Artibani W, Porcaro AB, De Marco V, Cerruto MA, Siracusano S (2018) Management of biochemical recurrence after primary curative treatment for prostate cancer: a review. Urol Int 100:251–262
Tilki D, Chen MH, Wu J et al (2021) Adjuvant versus early salvage radiation therapy for men at high risk for recurrence following radical prostatectomy for prostate cancer and the risk of death. J Clin Oncol 39:2284–2293
Hackman G, Taari K, Tammela TL et al (2019) Randomised trial of adjuvant radiotherapy following radical prostatectomy versus radical prostatectomy alone in prostate cancer patients with positive margins or extracapsular extension. Eur Urol 76:586–595
Pisansky TM, Thompson IM, Valicenti RK, D'Amico AV, Selvarajah S (2019) Adjuvant and salvage radiotherapy after prostatectomy: ASTRO/AUA guideline amendment 2018-2019. J Urol 202:533–538
Sooriakumaran P, Haendler L, Nyberg T et al (2012) Biochemical recurrence after robot-assisted radical prostatectomy in a European single-centre cohort with a minimum follow-up time of 5 years. Eur Urol 62:768–774
Van den Broeck T, van den Bergh RCN, Briers E et al (2020) Biochemical recurrence in prostate cancer: the European Association of Urology Prostate Cancer Guidelines panel recommendations. Eur Urol Focus 6:231–234
Van den Broeck T, van den Bergh RCN, Arfi N et al (2019) Prognostic value of biochemical recurrence following treatment with curative intent for prostate cancer: a systematic review. Eur Urol 75:967–987
Yossepowitch O, Briganti A, Eastham JA et al (2014) Positive surgical margins after radical prostatectomy: a systematic review and contemporary update. Eur Urol 65:303–313
Lee W, Lim B, Kyung YS, Kim C-S (2021) Impact of positive surgical margin on biochemical recurrence in localised prostate cancer. Prostate Int 9:151–156
Chapin BF, Nguyen JN, Achim MF et al (2018) Positive margin length and highest Gleason grade of tumor at the margin predict for biochemical recurrence after radical prostatectomy in patients with organ-confined prostate cancer. Prostate Cancer Prostatic Dis 21:221–227
Srigley JR (2006) Key issues in handling and reporting radical prostatectomy specimens. Arch Pathol Lab Med 130:303–317
Panebianco V, Villeirs G, Weinreb JC et al (2021) Prostate magnetic resonance imaging for local recurrence reporting (PI-RR): international consensus-based guidelines on multiparametric magnetic resonance imaging for prostate cancer recurrence after radiation therapy and radical prostatectomy. Eur Urol Oncol. https://doi.org/10.1016/j.euo.2021.01.003
Liauw SL, Pitroda SP, Eggener SE et al (2013) Evaluation of the prostate bed for local recurrence after radical prostatectomy using endorectal magnetic resonance imaging. Int J Radiat Oncol Biol Phys 85:378–384
Linder BJ, Kawashima A, Woodrum DA et al (2014) Early localisation of recurrent prostate cancer after prostatectomy by endorectal coil magnetic resonance imaging. Can J Urol 21:7283–7289
Couñago F, del Cerro E, Recio M et al (2015) Role of 3T multiparametric magnetic resonance imaging without endorectal coil in the detection of local recurrent prostate cancer after radical prostatectomy: the radiation oncology point of view. Scand J Urol 49:360–365
Vadi SK, Singh B, Basher RK et al (2017) 18F-Fluorocholine PET/CT complementing the role of dynamic contrast-enhanced MRI for providing comprehensive diagnostic workup in prostate cancer patients with suspected relapse following radical prostatectomy. Clin Nucl Med 42:e355–e361
Sooriakumaran P, Ploumidis A, Nyberg T et al (2015) The impact of length and location of positive margins in predicting biochemical recurrence after robot-assisted radical prostatectomy with a minimum follow-up of 5 years. BJU Int 115:106–113
Mottet N, Cornford P, van den Bergh RCN et al (2022) EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer. European Association of Urology Guidelines Office, Arnhem, The Netherlands
Mertan FV, Greer MD, Borofsky S et al (2016) Multiparametric magnetic resonance imaging of recurrent prostate cancer. Top Magn Reson Imaging 25:139–147
Kitajima K, Hartman RP, Froemming AT, Hagen CE, Takahashi N, Kawashima A (2015) Detection of local recurrence of prostate cancer after radical prostatectomy using endorectal coil MRI at 3 T: addition of DWI and dynamic contrast enhancement to T2-weighted MRI. AJR Am J Roentgenol 205:807–816
Patel P, Mathew MS, Trilisky I, Oto A (2018) Multiparametric MR imaging of the prostate after treatment of prostate cancer. Radiographics 38:437–449
Viers BR, Sukov WR, Gettman MT et al (2014) Primary Gleason grade 4 at the positive margin is associated with metastasis and death among patients with Gleason 7 prostate cancer undergoing radical prostatectomy. Eur Urol 66:1116–1124
Barchetti F, Panebianco V (2014) Multiparametric MRI for recurrent prostate cancer post radical prostatectomy and postradiation therapy. Biomed Res Int 2014:316272
Sandgren K, Westerlinck P, Jonsson JH et al (2019) Imaging for the detection of locoregional recurrences in biochemical progression after radical prostatectomy—a systematic review. Eur Urol Focus 5:550–560
Wu LM, Xu JR, Gu HY et al (2013) Role of magnetic resonance imaging in the detection of local prostate cancer recurrence after external beam radiotherapy and radical prostatectomy. Clin Oncol 25:252–264
Lawhn-Heath C, Salavati A, Behr SC et al (2021) Prostate-specific membrane antigen PET in prostate cancer. Radiology 299:248–260
Han S, Woo S, Kim YJ, Suh CH (2018) Impact of (68)Ga-PSMA PET on the management of patients with prostate cancer: a systematic review and meta-analysis. Eur Urol 74:179–190
Oh SW, Cheon GJ (2018) Prostate-specific membrane antigen PET imaging in prostate cancer: opportunities and challenges. Korean J Radiol 19:819–831
Achard V, Lamanna G, Denis A et al (2019) Recurrent prostate cancer after radical prostatectomy: restaging performance of 18F-choline hybrid PET/MRI. Med Oncol 36:67
Emmett L, van Leeuwen PJ, Nandurkar R et al (2017) Treatment outcomes from (68)Ga-PSMA PET/CT-informed salvage radiation treatment in men with rising PSA after radical prostatectomy: prognostic value of a negative PSMA PET. J Nucl Med 58:1972–1976
Heisen M, Fan X, Buurman J, van Riel NA, Karczmar GS, ter Haar Romeny BM (2010) The influence of temporal resolution in determining pharmacokinetic parameters from DCE-MRI data. Magn Reson Med 63:811–816
Calais J, Ceci F, Eiber M et al (2019) (18)F-fluciclovine PET-CT and (68)Ga-PSMA-11 PET-CT in patients with early biochemical recurrence after prostatectomy: a prospective, single-centre, single-arm, comparative imaging trial. Lancet Oncol 20:1286–1294
Funding
None
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Kye Jin Park, M.D., Ph.D.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
The requirement for written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at a single institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 24 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Park, M.Y., Park, K.J., Kim, MH. et al. Focal nodular enhancement on DCE MRI of the prostatectomy bed: radiologic-pathologic correlations and prognostic value. Eur Radiol 33, 2985–2994 (2023). https://doi.org/10.1007/s00330-022-09241-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-09241-0