Abstract
Objectives
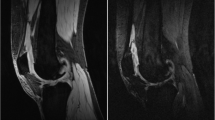
To determine whether synovitis graded by radiologists using hybrid quantitative double-echo in steady-state (qDESS) images can be utilized as a non-contrast approach to assess synovitis in the knee, compared against the reference standard of contrast-enhanced MRI (CE-MRI).
Methods
Twenty-two knees (11 subjects) with moderate to severe osteoarthritis (OA) were scanned using CE-MRI, qDESS with a high diffusion weighting (qDESSHigh), and qDESS with a low diffusion weighting (qDESSLow). Four radiologists graded the overall impression of synovitis, their diagnostic confidence, and regional grading of synovitis severity at four sites (suprapatellar pouch, intercondylar notch, and medial and lateral peripatellar recesses) in the knee using a 4-point scale. Agreement between CE-MRI and qDESS, inter-rater agreement, and intra-rater agreement were assessed using a linearly weighted Gwet’s AC2.
Results
Good agreement was seen between CE-MRI and both qDESSLow (AC2 = 0.74) and qDESSHigh (AC2 = 0.66) for the overall impression of synovitis, but both qDESS sequences tended to underestimate the severity of synovitis compared to CE-MRI. Good inter-rater agreement was seen for both qDESS sequences (AC2 = 0.74 for qDESSLow, AC2 = 0.64 for qDESSHigh), and good intra-rater agreement was seen for both sequences as well (qDESSLow AC2 = 0.78, qDESSHigh AC2 = 0.80). Diagnostic confidence was moderate to high for qDESSLow (mean = 2.36) and slightly less than moderate for qDESSHigh (mean = 1.86), compared to mostly high confidence for CE-MRI (mean = 2.73).
Conclusions
qDESS shows potential as an alternative MRI technique for assessing the severity of synovitis without the use of a gadolinium-based contrast agent.
Key Points
-
The use of the quantitative double-echo in steady-state (qDESS) sequence for synovitis assessment does not require the use of a gadolinium-based contrast agent.
-
Preliminary results found that low diffusion-weighted qDESS (qDESS Low ) shows good agreement to contrast-enhanced MRI for characterization of the severity of synovitis, with a relative bias towards underestimation of severity.
-
Preliminary results also found that qDESS Low shows good inter- and intra-rater agreement for the depiction of synovitis, particularly for readers experienced with the sequence.






Similar content being viewed by others
Abbreviations
- CE-MRI:
-
Contrast-enhanced MRI
- GBCA:
-
Gadolinium-based contrast agent
- OA:
-
Osteoarthritis
- qDESS:
-
Quantitative double-echo in steady-state
References
(2019) Osteoarthritis (OA) | Basics | Arthritis | CDC. https://www.cdc.gov/arthritis/basics/osteoarthritis.htm. Accessed 16 Nov 2019
(2015) Local treatment of inflammatory joint diseases: benefits and risks. Springer Berlin Heidelberg, New York
Roemer FW, Kwoh CK, Hannon MJ et al (2014) Can structural joint damage measured with MR imaging be used to predict knee replacement in the following year? Radiology 274:810–820. https://doi.org/10.1148/radiol.14140991
Roemer FW, Zhang Y, Niu J et al (2009) Tibiofemoral joint osteoarthritis: risk factors for MR-depicted fast cartilage loss over a 30-month period in the multicenter osteoarthritis study. Radiology 252:772–780. https://doi.org/10.1148/radiol.2523082197
Roemer FW, Kwoh CK, Hannon MJ et al (2015) What comes first? Multitissue involvement leading to radiographic osteoarthritis: magnetic resonance imaging–based trajectory analysis over four years in the osteoarthritis initiative. Arthritis Rheum 67:2085–2096. https://doi.org/10.1002/art.39176
Zhang Y, Nevitt M, Niu J et al (2011) Fluctuation of knee pain and changes in bone marrow lesions, effusions, and synovitis on magnetic resonance imaging. Arthritis Rheum 63:691–699. https://doi.org/10.1002/art.30148
Marckmann P, Skov L, Rossen K et al (2006) Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast-enhanced magnetic resonance imaging. J Am Soc Nephrol 17:2359–2362. https://doi.org/10.1681/ASN.2006060601
Rydahl C, Thomsen HS, Marckmann P (2008) High prevalence of nephrogenic systemic fibrosis in chronic renal failure patients exposed to gadodiamide, a gadolinium-containing magnetic resonance contrast agent. Invest Radiol 43:141–144. https://doi.org/10.1097/RLI.0b013e31815a3407
Sadowski EA, Bennett LK, Chan MR et al (2007) Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology 243:148–157. https://doi.org/10.1148/radiol.2431062144
Lawrence RC, Felson DT, Helmick CG et al (2008) Estimates of the prevalence of arthritis and other rheumatic conditions in the United States: part II. Arthritis Rheum 58:26–35. https://doi.org/10.1002/art.23176
Coresh J, Astor BC, Greene T, Eknoyan G, Levey AS (2003) Prevalence of chronic kidney disease and decreased kidney function in the adult US population: third national health and nutrition examination survey. Am J Kidney Dis 41:1–12. https://doi.org/10.1053/ajkd.2003.50007
Errante Y, Cirimele V, Mallio CA, Di Lazzaro V, Zobel BB, Quattrocchi CC (2014) Progressive increase of T1 signal intensity of the dentate nucleus on unenhanced magnetic resonance images is associated with cumulative doses of intravenously administered gadodiamide in patients with normal renal function, suggesting dechelation. Invest Radiol 49:685–690. https://doi.org/10.1097/RLI.0000000000000072
Radbruch A, Weberling LD, Kieslich PJ et al (2015) Gadolinium retention in the dentate nucleus and globus pallidus is dependent on the class of contrast agent. Radiology 275:783–791. https://doi.org/10.1148/radiol.2015150337
Kanda T, Ishii K, Kawaguchi H, Kitajima K, Takenaka D (2014) High signal intensity in the dentate nucleus and globus pallidus on unenhanced T1-weighted MR images: relationship with increasing cumulative dose of a gadolinium-based contrast material. Radiology 270:834–841. https://doi.org/10.1148/radiol.13131669
McDonald RJ, McDonald JS, Kallmes DF et al (2015) Intracranial gadolinium deposition after contrast-enhanced MR imaging. Radiology 275:772–782. https://doi.org/10.1148/radiol.15150025
Darrah TH, Prutsman-Pfeiffer JJ, Poreda RJ, Campbell ME, Hauschka PV, Hannigan RE (2009) Incorporation of excess gadolinium into human bone from medical contrast agents. Metallomics 1:479–488. https://doi.org/10.1039/b905145g
Maximova N, Gregori M, Zennaro F, Sonzogni A, Simeone R, Zanon D (2016) Hepatic gadolinium deposition and reversibility after contrast agent-enhanced MR imaging of pediatric hematopoietic stem cell transplant recipients. Radiology 281:418–426. https://doi.org/10.1148/radiol.2016152846
Roberts DR, Lindhorst SM, Welsh CT et al (2016) High levels of gadolinium deposition in the skin of a patient with normal renal function. Invest Radiol 51:280–289. https://doi.org/10.1097/RLI.0000000000000266
McWalter EJ, Sveinsson B, Oei EH, et al (2014) Non-contrast diffusion-weighted MRI for detection of synovitis using DESS. In: Proceedings of the 22nd International Society for Magnetic Resonance in Medicine (ISMRM) Annual Meeting. Milan, Italy
Oei EH, McWalter EJ, Sveinsson B et al (2014) Non-contrast diffusion weighted imaging for the assessment of knee synovitis: a comparative study against contrast-enhanced MRI. Osteoarthritis Cartilage 22:S252. https://doi.org/10.1016/j.joca.2014.02.481
Fan AP, Fong G, Sveinsson B et al (2015) Automated, non-contrast MRI for detection of synovitis using diffusion-weighted dess. Osteoarthritis Cartilage 23:A240–A241. https://doi.org/10.1016/j.joca.2015.02.447
Bruder H, Fischer H, Graumann R, Deimling M (1988) A new steady-state imaging sequence for simultaneous acquisition of two MR images with clearly different contrasts. Magn Reson Med 7:35–42. https://doi.org/10.1002/mrm.1910070105
Chaudhari AS, Black MS, Eijgenraam S et al (2018) 5-minute knee MRI for simultaneous morphometry and T2 relaxometry of cartilage and meniscus and for semi-quantitative radiological assessment using double-Echo in steady-state at 3T. J Magn Reason Imaging 47:1328–1341. https://doi.org/10.1002/jmri.25883
Chaudhari AS, Sveinsson B, Moran CJ et al (2017) Imaging and T2 relaxometry of short-T2 connective tissues in the knee using ultrashort echo-time double-echo steady-state (UTEDESS). Magn Reson Med 78:2136–2148. https://doi.org/10.1002/mrm.26577
Burke CJ, Alizai H, Beltran L, Regatte RR (2019) MRI of Synovitis and joint fluid. J Magn Reason Imaging 49:1512–1527. https://doi.org/10.1002/jmri.26618
Son YN, Jin W, Jahng G-H et al (2018) Efficacy of double inversion recovery magnetic resonance imaging for the evaluation of the synovium in the femoro-patellar joint without contrast enhancement. Eur Radiol 28:459–467. https://doi.org/10.1007/s00330-017-5017-3
Yi J, Lee YH, Song H-T, Suh J-S (2019) Double-inversion recovery with synthetic magnetic resonance: a pilot study for assessing synovitis of the knee joint compared to contrast-enhanced magnetic resonance imaging. Eur Radiol 29:2573–2580. https://doi.org/10.1007/s00330-018-5800-9
Yoo HJ, Hong SH, Oh HY et al (2016) Diagnostic accuracy of a fluid-attenuated inversion-recovery sequence with fat suppression for assessment of peripatellar synovitis: preliminary results and comparison with contrast-enhanced MR imaging. Radiology 283:769–778. https://doi.org/10.1148/radiol.2016160155
Kogan F, Levine E, Chaudhari AS et al (2018) Simultaneous bilateral-knee MR imaging. Magn Reson Med 80:529–537. https://doi.org/10.1002/mrm.27045
Sveinsson B, Chaudhari A, Gold G, Hargreaves B (2017) A simple analytic method for estimating T2 in the knee from DESS. Magn Reson Imaging 38:63–70. https://doi.org/10.1016/j.mri.2016.12.018
Weigel M (2015) Extended phase graphs: dephasing, RF pulses, and echoes - pure and simple. J Magn Reson Imaging 41:266–295. https://doi.org/10.1002/jmri.24619
Thoenen J, MacKay JW, Chaudhari A, et al (2020) Ideal weighting parameter for non-contrast detection of synovitis using diffusion-weighted DESS. In: proceedings of 28th annual meeting of ISMRM. Paris, France
Gold GE, Suh B, Sawyer-Glover A, Beaulieu C (2004) Musculoskeletal MRI at 3.0 T: initial clinical experience. AJR Am J Roentgenol 183:1479–1486. https://doi.org/10.2214/ajr.183.5.1831479
Bieri O (2012) Superbalanced steady state free precession. Magn Reson Med 67:1346–1354. https://doi.org/10.1002/mrm.23122
Rhodes LA, Grainger AJ, Keenan A-M, Thomas C, Emery P, Conaghan PG (2005) The validation of simple scoring methods for evaluating compartment-specific synovitis detected by MRI in knee osteoarthritis. Rheumatology 44:1569–1573. https://doi.org/10.1093/rheumatology/kei094
Guermazi A, Roemer FW, Hayashi D et al (2011) Assessment of synovitis with contrast-enhanced MRI using a whole-joint semiquantitative scoring system in people with, or at high risk of, knee osteoarthritis: the MOST study. Ann Rheum Dis 70:805–811. https://doi.org/10.1136/ard.2010.139618
Hunter DJ, Guermazi A, Lo GH et al (2011) Evolution of semi-quantitative whole joint assessment of knee OA: MOAKS (MRI Osteoarthritis Knee Score). Osteoarthritis Cartilage 19:990–1002. https://doi.org/10.1016/j.joca.2011.05.004
Gwet KL (2008) Computing inter-rater reliability and its variance in the presence of high agreement. Br J Math Stat Psychol 61:29–48. https://doi.org/10.1348/000711006X126600
Chaudhari AS, Stevens KJ, Sveinsson B et al (2019) Combined 5-minute double-echo in steady-state with separated echoes and 2-minute proton-density-weighted 2D FSE sequence for comprehensive whole-joint knee MRI assessment. J Magn Reson Imaging 49:e183–e194. https://doi.org/10.1002/jmri.26582
Chaudhari AS, Grissom MJ, Fang Z et al (2020) Diagnostic accuracy of quantitative multi-contrast 5-minute knee MRI using prospective artificial intelligence image quality enhancement. AJR Am J Roentgenol. https://doi.org/10.2214/AJR.20.24172
Chaudhari AS, Fang Z, Kogan F et al (2018) Super-resolution musculoskeletal MRI using deep learning. Magn Reson Med 80:2139–2154. https://doi.org/10.1002/mrm.27178
Chaudhari AS, Stevens KJ, Wood JP et al (2020) Utility of deep learning super-resolution in the context of osteoarthritis MRI biomarkers. J Magn Reason Imaging 51:768–779. https://doi.org/10.1002/jmri.26872
Jahng G-H, Jin W, Yang DM, Ryu KN (2011) Optimization of a double inversion recovery sequence for noninvasive synovium imaging of joint effusion in the knee. Med Phys 38:2579–2585. https://doi.org/10.1118/1.3581060
Treutlein C, Bäuerle T, Nagel AM et al (2020) Comprehensive assessment of knee joint synovitis at 7 T MRI using contrast-enhanced and non-enhanced sequences. BMC Musculoskelet Disord 21:116. https://doi.org/10.1186/s12891-020-3122-y
Funding
This study has received grant support from GE Healthcare and NIH grants R01EB002524, R01AR077604, K24AR062068, R00EB022634, and an OARSI Collaborative Research Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Feliks Kogan.
Conflict of interest
The authors of this manuscript declare relationships with the following companies: Drs. Gold, MacKay, Hargreaves, Kogan, Chaudhari, and Stevens receive research funding from GE Healthcare, Drs. Gold, Hargreaves, Kogan, and Chaudhari receive research funding from Phillips Healthcare, and Dr. MacKay receives research funding from GlaxoSmithKline. Dr. MacKay is now an employee of AstraZeneca.
Statistics and biometry
Dr. Jarrett Rosenberg kindly provided statistical advice for this manuscript.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• experimental
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 320 kb)
Rights and permissions
About this article
Cite this article
Thoenen, J., Stevens, K.J., Turmezei, T.D. et al. Non-contrast MRI of synovitis in the knee using quantitative DESS. Eur Radiol 31, 9369–9379 (2021). https://doi.org/10.1007/s00330-021-08025-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08025-2