Abstract
Objective
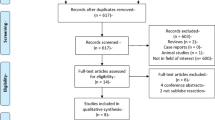
Multiple cohort studies have compared surgical resection with CT-guided percutaneous ablation for patients with stage 1 non-small cell lung cancer (NSCLC); however, the results have been heterogeneous. This systematic review and meta-analysis aims to compare surgery with ablation for stage 1 NSCLC.
Method
A search of five databases was performed from inception to 5 July 2020. Studies were included if overall survival (OS), cancer-specific survival (CSS), and/or disease-free survival (DFS) were compared between patients treated with surgical resection versus ablation (radiofrequency ablation (RFA) or microwave ablation (MWA)) for stage 1 NSCLC. Pooled odds ratios (OR) were calculated.
Results
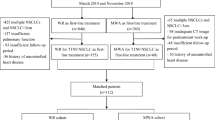
A total of eight studies were included (total 792 patients: 460 resection and 332 ablation). There were no significant differences in 1- to 5-year OS or CSS between surgery versus ablation. There were significantly better 1- and 2-year DFS for surgery over ablation (OR 2.22, 95% CI 1.14–4.34; OR 2.60, 95% CI 1.21–5.57 respectively), but not 3- to 5-year DFS. Subgroup analysis demonstrated no significant OS difference between lobectomy and MWA, but there were significantly better 1- and 2-year OS with sublobar resection (wedge resection or segmentectomy) versus RFA (OR 2.85, 95% CI 1.33–6.10; OR 4.54, 95% CI 2.51–8.21, respectively). In the two studies which only included patients with stage 1A NSCLC, pooled outcomes demonstrated no significant differences in 1- to 3-year OS or DFS between surgery versus ablation.
Conclusion
Surgical resection of stage 1 NSCLC remains the optimal choice. However, for non-surgical patients with stage 1A, ablation offers promising DFS, CSS, and OS. Future prospective randomized controlled trials are warranted.
Key Points
• Surgical resection of stage 1 NSCLC remains the optimal choice.
• In patients with stage 1A NSCLC who are not surgical candidates, CT-guided microwave or radiofrequency ablation may be an alternative which offers promising disease-free survival and overall survival.




Similar content being viewed by others
Abbreviations
- CI:
-
Confidence interval
- CSS:
-
Cancer-specific survival
- CT:
-
Computed tomography
- DFS:
-
Disease-free survival
- MWA:
-
Microwave ablation
- NSCLC:
-
Non-small cell lung cancer
- ORs:
-
Odds ratios
- OS:
-
Overall survival
- PET:
-
Positron emission tomography
- RFA:
-
Radiofrequency ablation
References
Torre LA, Bray F, Siegel RL et al (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108
Postmus P, Kerr KM, Oudkerk M et al (2017) Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 28(suppl_4):iv1–iv21
Keenan RJ, Landreneau RJ, Maley RH Jr et al (2004) Segmental resection spares pulmonary function in patients with stage I lung cancer. Ann Thorac Surg 78(1):228–233
Berry MF, Hanna J, Tong BC et al (2009) Risk factors for morbidity after lobectomy for lung cancer in elderly patients. Ann Thorac Surg 88(4):1093–1099
Yang J, Xia Y, Yang Y et al (2014) Risk factors for major adverse events of video-assisted thoracic surgery lobectomy for lung cancer. Int J Med Sci 11(9):863
Donington J, Ferguson M, Mazzone P et al (2012) American College of Chest Physicians and Society of Thoracic Surgeons consensus statement for evaluation and management for high-risk patients with stage I non-small cell lung cancer. Chest 142(6):1620–1635
Safi S, Rauch G, op den Winkel J et al (2015) Sublobar resection, radiofrequency ablation or radiotherapy in stage I non-small cell lung cancer. Respiration 89(6):550–557
Wang Y, Liu B, Cao P et al (2018) Comparison between computed tomography-guided percutaneous microwave ablation and thoracoscopic lobectomy for stage I non-small cell lung cancer. Thorac Cancer 9(11):1376–1382
Yao W, Lu M, Fan W et al (2018) Comparison between microwave ablation and lobectomy for stage I non-small cell lung cancer: a propensity score analysis. Int J Hyperthermia 34(8):1329–1336
Kim SR, Han HJ, Park SJ et al (2012) Comparison between surgery and radiofrequency ablation for stage I non-small cell lung cancer. Eur J Radiol 81(2):395–399
Subramanian M, McMurry T, Meyers B et al (2018) Long-term results for clinical stage IA lung cancer: comparing lobectomy and sublobar resection. Ann Thorac Surg 106(2):375–381
Sienel W, Dango S, Kirschbaum A et al (2008) Sublobar resections in stage IA non-small cell lung cancer: segmentectomies result in significantly better cancer-related survival than wedge resections. Eur J Cardiothorac Surg 33(4):728–734
Ginsberg RJ, Rubinstein LV, L.C.S. Group (1995) Randomized trial of lobectomy versus limited resection for T1 N0 non-small cell lung cancer. Ann Thorac Surg 60(3):615–623
Solazzo SA, Liu Z, Lobo SM et al (2005) Radiofrequency ablation: importance of background tissue electrical conductivity—an agar phantom and computer modeling study. Radiology 236(2):495–502
Brace CL, Hinshaw JL, Laeseke PF, Sampson LA, Lee FT (2009) Pulmonary thermal ablation: comparison of radiofrequency and microwave devices by using gross pathologic and CT findings in a swine model. Radiology 251(3):705–711
Vogl TJ, Eckert R, Naguib NN, Beeres M, Gruber-Rouh T, Nour-Eldin NA (2016) Thermal ablation of colorectal lung metastases: retrospective comparison among laser-induced thermotherapy, radiofrequency ablation, and microwave ablation. AJR Am J Roentgenol 207(6):1340–1349
Moher D, Shamser L, Clarke M et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4(1):1
NHMRC (2000) How to use the evidence: assessment and application of scientific evidence. Canberra, National Health and Medical Research Council.
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560
Alexander ES, Machan JT, Ng T, Breen LD, DiPetrillo TA, Dupuy DE (2013) Cost and effectiveness of radiofrequency ablation versus limited surgical resection for stage I non-small-cell lung cancer in elderly patients: is less more? J Vasc Interv Radiol 24(4):476–482
Ambrogi MC, Fanucchi O, Dini P (2015) Wedge resection and radiofrequency ablation for stage I nonsmall cell lung cancer. Eur Respir J 45(4):1089–1097
Kwan SW, Mortell KE, Talenfeld AD, Brunner MC (2014) Thermal ablation matches sublobar resection outcomes in older patients with early-stage non-small cell lung cancer. J Vasc Interv Radiol 25(1):1–9.e1
Zemlyak A, Moore WH, Bilfinger TV (2010) Comparison of survival after sublobar resections and ablative therapies for stage I non-small cell lung cancer. J Am Coll Surg 211(1):68–72
Yasufuku K, Nakajima T, Motoori K et al (2006) Comparison of endobronchial ultrasound, positron emission tomography, and CT for lymph node staging of lung cancer. Chest 130(3):710–718
Cackler S, Abbas G (2009) RFA is an effective alternative to lobectomy for lung cancer. JAAPA 22(1):25–28
Vogl TJ, Naguib NNN, Gruber-Fouh T et al (2011) Microwave ablation therapy: clinical utility in treatment of pulmonary metastases. Radiology 261(2):643–651
Ito N, Nakatsuka S, Inoue M et al (2012) Computed tomographic appearance of lung tumors treated with percutaneous cryoablation. J Vasc Interv Radiol 23(8):1043–1052
Deandreis D, Leboulleux S, Dromain C et al (2011) Role of FDG PET/CT and chest CT in the follow-up of lung lesions treated with radiofrequency ablation. Radiology 258(1):270–276
Detterbeck FC, Boffa DJ, Kim AW, Tanoue LT (2017) The eighth edition lung cancer stage classification. Chest 151(1):193–203
Lanuti M, Sharma A, Willers H, Digumarthy SR, Mathisen DJ, Shepart JA (2012) Radiofrequency ablation for stage I non-small cell lung cancer: management of locoregional recurrence. Ann Thorac Surg 93(3):921–927 discussion 927-88
Kashima M, Yamakado K, Takaki H et al (2011) Complications after 1000 lung radiofrequency ablation sessions in 420 patients: a single center’s experiences. AJR Am J Roentgenol 197(4):W576–W580
Cao C, Wang D, Chung C et al (2019) A systematic review and meta-analysis of stereotactic body radiation therapy versus surgery for patients with non–small cell lung cancer. J Thorac Cardiovasc Surg 157(1):362–373. e8
Uhlig J, Ludwig JM, Goldberg SB, Chiang A, Blasberg JD, Kim HS (2018) Survival rates after thermal ablation versus stereotactic radiation therapy for stage 1 non-small cell lung cancer: a National Cancer Database study. Radiology 289(3):862–870
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr Ya Ruth Huo.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was not required for this study because no human patients were directly involved in this systematic review and meta-analysis.
Ethical approval
Institutional Review Board approval was not required because this was a systematic review and meta-analysis.
Study subjects or cohorts overlap
Some study subjects or cohorts have been previously reported in published papers. This systematic review and meta-analysis pools the studies together.
Methodology
• Systematic review and meta-analysis
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Level of evidence: Level 1
Michael Vinchill Chan and Ya Ruth Huo are Equal first co-authors
Supplementary Information
ESM 1
(DOCX 17 kb)
LOS, length of stay; NR, not reported
Rights and permissions
About this article
Cite this article
Chan, M.V., Huo, Y.R., Cao, C. et al. Survival outcomes for surgical resection versus CT-guided percutaneous ablation for stage I non-small cell lung cancer (NSCLC): a systematic review and meta-analysis. Eur Radiol 31, 5421–5433 (2021). https://doi.org/10.1007/s00330-020-07634-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07634-7