Abstract
Objectives
To evaluate quantification of iodine uptake in metastatic and non-metastatic lymph nodes (LNs) by dual-energy CT (DECT) and to assess if the distribution of iodine within LNs at DECT correlates with the pathological structure.
Methods
Ninety LNs from 37 patients (23 with lung and 14 with gynaecological malignancies) were retrospectively selected. Information of LNs sent for statistical analysis included Hounsfield units (HU) at different energy levels; decomposition material densities fat–iodine, iodine–fat, iodine–water, water–iodine. Statistical analysis included evaluation of interobserver variability, material decomposition densities and spatial HU distribution within LNs.
Results
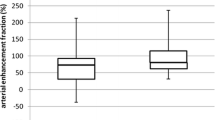
Interobserver agreement was excellent. There was a significant difference in iodine–fat and iodine–water decompositions comparing metastatic and non-metastatic LNs (p < 0.001); fat–iodine and water–iodine did not show significant differences. HU distribution showed a significant gradient from centre to periphery within non-metastatic LNs that was significant up to 20–30% from the centre, whereas metastatic LNs showed a more homogeneous distribution of HU, with no significant gradient.
Conclusions
DECT demonstrated a lower iodine uptake in metastatic compared to non-metastatic LNs. Moreover, the internal iodine distribution showed an evident gradient of iodine distribution from centre to periphery in non-metastatic LNs, and a more homogeneous distribution within metastatic LNs, which corresponded to the pathological structure.
Key points
• This study demonstrated a lower iodine uptake in metastatic than non-metastatic LNs.
• Internal distribution of HU was different between metastatic and non-metastatic lymph nodes.
• The intranodal iodine distribution disclosed a remarkable correlation with the histological LN structure.






Similar content being viewed by others
Abbreviations
- DECT:
-
Dual-energy computed tomography
- HU:
-
Hounsfield units
- keV:
-
Kilo electron volt
- kV:
-
Kilovolt
- LN(s):
-
Lymph node(s)
- LS:
-
Least-squares
- SE:
-
Standard error
References
Johnson TR (2012) Dual-energy CT: general principles. AJR Am J Roentgenol 199:S3–S8
Agrawal MD, Pinho DF, Kulkarni NM, Han PF, Guimaraes AR, Sahani DV (2014) Oncological application of dual-energy CT in the abdomen. RadioGraphics 34:589–612
Marin D, Nelson RC, Samei E et al (2009) Hypervascular liver tumors: low tube voltage, high tube current multidetector CT during late hepatic arterial phase for detection—initial clinical experience. Radiology 251:771–779
Patel BN, Thomas JV, Lockhart ME, Berland LL, Morgan DE (2013) Single-source dual-energy spectral multidetector CT of pancreatic adenocarcinoma: optimization of energy level viewing significantly increases lesion contrast. Clin Radiol 68:148–154
Song KD, Kim CK, Park BK, Kim B (2011) Utility of iodine overlay technique and virtual unenhanced images for the characterization of renal masses by dual-energy CT. AJR Am J Roentgenol 197:W1076–W1082
Ho LM, Marin D, Neville AM et al (2012) Characterization of adrenal nodules with dual-energy CT: can virtual unenhanced attenuation values replace true unenhanced attenuation values? AJR Am J Roentgenol 198:840–845
Elmore SA (2006) Histopathology of the lymph nodes. Toxicol Pathol 34:425–454
Tawfik AM, Razek AA, Kerl JM, Nour-Eldim NE, Bauer R, Vogl TJ (2014) Comparison of dual-energy CT-derived iodine content and iodine overlay of normal, inflammatory and metastatic squamous cell carcinoma cervical lymph nodes. Eur Radiol 24:574–580
Liu H, Yan F, Pan Z et al (2015) Evaluation of dual energy spectral CT in differentiating metastatic from non-metastatic lymph nodes in rectal cancer: Initial experience. Eur Radiol 84:228–234
Yang L, Luo D, Li L, Zhao Y, Lin M, Guo W, Zhou C (2016) Differentiation of malignant cervical lymphadenopathy by dual-energy CT: a preliminary analysis. Sci Rep 6:31020
Raudenbush SW, Bryk AS (2002) Hierarchical linear models: applications and data analysis methods, 2nd edn. Sage, London
McCollough CH, Leng S, Yu L, Fletcher JG (2015) Dual- and multi-energy CT: principles, technical approaches, and clinical applications. Radiology 276:637–653
Kato T, Uehara K, Ishigaki S et al (2015) Clinical significance of dual-energy CT-derived iodine quantification in the diagnosis of metastatic LN in colorectal cancer. Eur J Surg Oncol 41:1464–1470
Willard-Mack CL (2006) Normal structure, function and histology of lymph nodes. Toxicol Pathol 34:409–424
Hussaini SAB, Andola SK, Mahanta A (2015) Study of metastasis in lymphnode biopsies with special reference to immunohistochemistry (IHC) in metastatic breast carcinoma. J Clin Diagn Res 9:EC13–EC16
Nathanson SD (2003) Insights into the mechanisms of lymph node metastasis. Cancer 98:413–423
Pastushenko I, Van den Eynden GG, Vicente-Arregui S, Prieto-Torres L, Alvarez-Alegret R, Querol I et al (2016) Increased angiogenesis and lymphangiogenesis in metastatic sentinel lymph nodes is associated with nonsentinel lymph node involvement and distant metastasis in patients with melanoma. Am J Dermatopathol 38:338–346
Jeong HS, Jones D, Liao S, Wattson DA, Cui CH, Duda DG et al (2015) Investigation of the lack of angiogenesis in the formation of lymph node metastases. J Natl Cancer Inst. doi:10.1093/jnci/djv155
Acknowledgements
The English text has been revised by Anne Prudence Collins (Editor and Translator Medical & Scientific Publications).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Stefania Rizzo, MD, Istituto Europeo di Oncologia.
Conflict of interest
The authors of this manuscript declare relationships with the following companies: Alberto Mauro is an employee of GE healthcare, who helped the authors in setting the CT protocol, and did not alter in any way the data collected for this article.
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Funding
The authors state that this work has not received any funding.
Statistics and biometry
One of the authors (Davide Radice) has significant statistical expertise.
Informed consent
Written informed consent was obtained from all patients in this study.
Ethical approval
Institutional review board approval was obtained.
Methodology
• retrospective
• observational
• performed at one institution
Electronic supplementary material
ESM 1
(DOCX 5672 kb)
Rights and permissions
About this article
Cite this article
Rizzo, S., Radice, D., Femia, M. et al. Metastatic and non-metastatic lymph nodes: quantification and different distribution of iodine uptake assessed by dual-energy CT. Eur Radiol 28, 760–769 (2018). https://doi.org/10.1007/s00330-017-5015-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5015-5