Abstract
Objectives
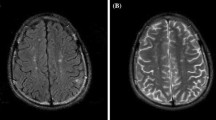
To evaluate whether quantitative susceptibility mapping (QSM) can be employed to detect abnormalities within normal-appearing basal ganglia on conventional MRI in patients with neuropsychiatric systemic lupus erythematosus (NPSLE).
Methods
For 33 SLE patients (13 NPSLE and 20 non-NPSLE patients) and 23 age/sex-matched controls, two radiologists independently measured the mean QSM and R2* values in various brain structures that appeared to be normal on conventional MR images. These values in each brain structure were compared among the two SLE groups and controls.
Results
Regarding the putamen, the NPSLE patients showed significantly higher QSM values than the non-NPSLE patients and controls (p < 0.05). For the lateral globus pallidus, both SLE groups showed significantly higher QSM values than the controls (p < 0.05). The R2* values were not significantly different between both SLE groups. The NPSLE patients showed a significant correlation between the mean QSM values in putamen and the disease duration (r = 0.63, p < 0.05). For the interobserver agreement, the QSM value was superior to the R2* value (0.690 vs. 0.446, Kendall W value).
Conclusions
QSM can be used to identify increased susceptibility of the basal ganglia appearing to be normal on conventional MR images in NPSLE patients.
Key Points
• QSM values in the putamen are significantly higher in NPSLE than non-NPSLE.
• NPSLE patients show correlation between QSM values in the putamen and disease duration.
• QSM is more sensitive than R2* mapping for detecting subtle changes.



Similar content being viewed by others
Abbreviations
- NPSLE:
-
Neuropsychiatric systemic lupus erythematosus
- SLE:
-
Systemic lupus erythematosus
- QSM:
-
Quantitative susceptibility mapping
References
O’Connor JF, Musher DM (1966) Central nervous system involvement in systemic lupus erythematosus: a study of 150 cases. Arch Neurol 14:157–164
Sibbitt WL Jr, Brooks WM, Kornfeld M, Hart BL, Bankhurst AD, Roldan CA (2010) Magnetic resonance imaging and brain histopathology in neuropsychiatric systemic lupus erythematosus. Semin Arthritis Rheum 40:32–52
Kim JH, Choi C-G, Choi S-J, Lee HK, Suh DC (2000) Primary antiphospholipid antibody syndrome: neuroradiologic findings in 11 patients. Korean J Radiol 1:5–10
Golstein M, Meyer O, Bourgeois P et al (1992) Neurological manifestations of systemic lupus erythematosus: role of antiphospholipid antibodies. Clin Exp Rheumatol 11:373–379
Zivadinov R, Shucard J, Hussein S et al (2013) Multimodal imaging in systemic lupus erythematosus patients with diffuse neuropsychiatric involvement. Lupus 22:675–683
Lee S, Park MC, Lee S, Park Y (2012) The efficacy of brain F-fluorodeoxyglucose positron emission tomography in neuropsychiatric lupus patients with normal brain magnetic resonance imaging findings. Lupus 21:1531–1537
Zhang X, Zhu Z, Zhang F, Shu H, Li F, Dong Y (2005) Diagnostic value of single-photon-emission computed tomography in severe central nervous system involvement of systemic lupus erythematosus: a case-control study. Arthritis Rheum 53:845–849
Popescu A, Kao AH (2011) Neuropsychiatric systemic lupus erythematosus. Curr Neuropharmacol 9:449
Mehta V, Pei W, Yang G et al (2013) Iron is a sensitive biomarker for inflammation in multiple sclerosis lesions. PLoS One 8, e57573
Hametner S, Wimmer I, Haider L, Pfeifenbring S, Brück W, Lassmann H (2013) Iron and neurodegeneration in the multiple sclerosis brain. Ann Neurol 74:848–861
Bagnato F, Hametner S, Yao B et al (2011) Tracking iron in multiple sclerosis: a combined imaging and histopathological study at 7 Tesla. Brain 134:3599–3612
Langkammer C, Liu T, Khalil M et al (2013) Quantitative susceptibility mapping in multiple sclerosis. Radiology 267:551–559
Walsh AJ, Lebel RM, Eissa A et al (2013) Multiple sclerosis: validation of MR imaging for quantification and detection of iron. Radiology 267:531–542
Chen W, Gauthier SA, Gupta A et al (2014) Quantitative susceptibility mapping of multiple sclerosis lesions at various ages. Radiology 271:183–192
Kell DB (2010) Towards a unifying, systems biology understanding of large-scale cellular death and destruction caused by poorly liganded iron: Parkinson’s, Huntington’s, Alzheimer’s, prions, bactericides, chemical toxicology and others as examples. Arch Toxicol 84:825–889
Haacke EM, Cheng NY, House MJ et al (2005) Imaging iron stores in the brain using magnetic resonance imaging. Magn Reson Imaging 23:1–25
Péran P, Cherubini A, Assogna F et al (2010) Magnetic resonance imaging markers of Parkinson’s disease nigrostriatal signature. Brain 133:3423–3433
Yao B, Li T-Q, Pv G, Shmueli K, de Zwart JA, Duyn JH (2009) Susceptibility contrast in high field MRI of human brain as a function of tissue iron content. Neuroimage 44:1259–1266
Wang S, Lou M, Liu T, Cui D, Chen X, Wang Y (2013) Hematoma volume measurement in gradient echo MRI using quantitative susceptibility mapping. Stroke 44:2315–2317
Yablonskiy DA, Haacke EM (1994) Theory of NMR signal behavior in magnetically inhomogeneous tissues: the static dephasing regime. Magn Reson Med 32:749–763
de Rochefort L, Liu T, Kressler B et al (2010) Quantitative susceptibility map reconstruction from MR phase data using bayesian regularization: validation and application to brain imaging. Magn Reson Med 63:194–206
Liu T, Wisnieff C, Lou M, Chen W, Spincemaille P, Wang Y (2013) Nonlinear formulation of the magnetic field to source relationship for robust quantitative susceptibility mapping. Magn Reson Med 69:467–476
Liu J, Liu T, de Rochefort L et al (2012) Morphology enabled dipole inversion for quantitative susceptibility mapping using structural consistency between the magnitude image and the susceptibility map. Neuroimage 59:2560–2568
Bilgic B, Pfefferbaum A, Rohlfing T, Sullivan EV, Adalsteinsson E (2012) MRI estimates of brain iron concentration in normal aging using quantitative susceptibility mapping. Neuroimage 59:2625–2635
Ide S, Kakeda S, Ueda I et al (2014) Internal structures of the globus pallidus in patients with Parkinson’s disease: evaluation with quantitative susceptibility mapping (QSM). Eur Radiol 25:1–9
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40:1725–1725
Ainiala H, Hietaharju A, Loukkola J et al (2001) Validity of the new American College of Rheumatology criteria for neuropsychiatric lupus syndromes: a population-based evaluation. Arthritis Rheum 45:419–423
West SG, Emlen W, Wener MH, Kotzin BL (1995) Neuropsychiatric lupus erythematosus: a 10-year prospective study on the value of diagnostic tests. Am J Med 99:153–163
Fazekas F, Kleinert R, Offenbacher H et al (1991) The morphologic correlate of incidental punctate white matter hyperintensities on MR images. Am J Neuroradiol 12:915–921
Bombardier C, Gladman D, Urowitz M, Caron D, Chang CH (1992) Derivation of the SLEDAI: a disease activity index for lupus patients. The Committee on Prognosis Studies in SLE. Arthritis Rheum 35:630–640
Liu T, Khalidov I, de Rochefort L et al (2011) A novel background field removal method for MRI using projection onto dipole fields (PDF). NMR Biomed 24:1129–1136
Jin L, Wang J, Zhao L et al (2011) Decreased serum ceruloplasmin levels characteristically aggravate nigral iron deposition in Parkinson’s disease. Brain 134:50–58
Lim MK, Suh CH, Kim HJ et al (2000) Systemic lupus erythematosus: brain MR imaging and single-voxel hydrogen 1 MR spectroscopy. Radiology 217:43–49
Jung RE, Caprihan A, Chavez RS et al (2010) Diffusion tensor imaging in neuropsychiatric systemic lupus erythematosus. BMC Neurol 10:65
Roldan CA, Shively BK, Crawford MH (1996) An echocardiographic study of valvular heart disease associated with systemic lupus erythematosus. N Engl J Med 335:1424–1430
Moore PM, Lisak RP (1996) Systemic lupus erythematosus: immunopathogenesis of neurologic dysfunction. Springer Semin Immunoneurol 17:43–60
Campbell A, Smith MA, Sayre LM, Bondy SC, Perry G (2001) Mechanisms by which metals promote events connected to neurodegenerative diseases. Brain Res Bull 55:125–132
Hallgren B, Sourander P (1958) The effect of age on the non-haemin iron in the human brain. J Neurochem 3:41–51
Takanashi J-I, Barkovich AJ, Dillon WP, Sherr EH, Hart KA, Packman S (2003) T1 hyperintensity in the pulvinar: key imaging feature for diagnosis of Fabry disease. Am J Neuroradiol 24:916–921
Langkammer C, Krebs N, Goessler W et al (2010) Quantitative MR imaging of brain iron: a postmortem validation study. Radiology 257:455–462
Pauling L, Coryell CD (1936) The magnetic properties and structure of hemoglobin, oxyhemoglobin and carbonmonoxyhemoglobin. Proc Natl Acad Sci U S A 22:210
Acknowledgements
The scientific guarantor of this publication is Yukunori Korogi M.D. The authors of this manuscript declare relationships with the following companies: Y. Tanaka has received consulting fees, speaking fees and/or honoraria from Abbvie, Chugai, Astellas, Takeda, Santen, Mitsubishi-Tanabe, Pfizer, Janssen, Eisai, Daiichi-Sankyo, UCB, GlaxoSmithKline, Bristol-Myers and has received research grants from Mitsubishi-Tanabe, Chugai, MSD, Astellas, Novartis. The other authors state that this work has not received any funding. No complex statistical methods were necessary for this paper. Institutional review board approval was obtained. Written informed consent was waived by the institutional review board. Methodology: retrospective case-control study performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ogasawara, A., Kakeda, S., Watanabe, K. et al. Quantitative susceptibility mapping in patients with systemic lupus erythematosus: detection of abnormalities in normal-appearing basal ganglia. Eur Radiol 26, 1056–1063 (2016). https://doi.org/10.1007/s00330-015-3929-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-3929-3