Abstract
To investigate the frequency, profile, and severity of COVID-19 breakthrough infections (BI) in patients with type I diabetes mellitus (T1DM) compared to healthy controls (HC) after vaccination. The second COVID-19 Vaccination in Autoimmune Diseases (COVAD-2) survey is a multinational cross-sectional electronic survey which has collected data on patients suffering from various autoimmune diseases including T1DM. We performed a subgroup analysis on this cohort to investigate COVID-19 BI characteristics in patients with T1DM. Logistic regression with propensity score matching analysis was performed. A total of 9595 individuals were included in the analysis, with 100 patients having T1DM. Among the fully vaccinated cohort, 16 (16%) T1DM patients had one BI and 2 (2%) had two BIs. No morbidities or deaths were reported, except for one patient who required hospitalization with oxygen without admission to intensive care. The frequency, clinical features, and severity of BIs were not significantly different between T1DM patients and HCs after adjustment for confounding factors. Our study did not show any statistically significant differences in the frequency, symptoms, duration, or critical care requirements between T1DM and HCs after COVID-19 vaccination. Further research is needed to identify factors associated with inadequate vaccine response in patients with BIs, especially in patients with autoimmune diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The ongoing COVID-19 pandemic has significantly impacted individuals living with systemic autoimmune diseases (SAIDs) worldwide. A weaker immune response to infection along with the use of long-term immunosuppressive drugs put them at the risk of adverse clinical outcomes and mortality [1, 2]. Individuals with SAIDs such as T1 diabetes (T1DM), particularly those with poor glycaemic control are more susceptible to developing infections due to associated cytokine dysregulation and premature immunosenescence (accelerated aging of the immune system) in hyperglycemia [3]. Additionally, they may be particularly predisposed due to hyperglycemia and consequent immune dysregulation [4]. These patients may also have multiple complications of their disease (e.g. cardiovascular disease, chronic kidney disease) [5]. Hence, this patient subgroup is more likely to develop severe COVID-19 infection and its complications.
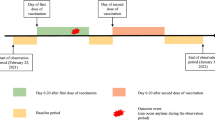
The development and widespread uptake of COVID-19 vaccines had been critical in containing and mitigating the impact of this virus. While current evidence suggests that the benefits of the vaccine outweigh its potential of vaccine-induced disease flares and other adverse outcomes [6, 7], the emergence of COVID-19 breakthrough infections (BI), defined as the detection of SARS-CoV-2 or antigen in the respiratory samples of an individual ≥ 14 days after complete vaccination, has raised concerns about the effectiveness of vaccines [8]. A diagnosis of systemic autoimmune diseases (SAIDs) is likely to render individuals at a higher risk of severe BI [9] contributing to vaccine hesitancy [10]. With limited evidence available on the safety of COVID-19 vaccines in T1DM patients, it is imperative to understand the prevalence and risk factors for BI in adults with T1DM.
This study aimed to investigate the frequency, profile, severity, and time to resolution of COVID-19 breakthrough infections (BI) in patients with type I diabetes mellitus (T1DM) compared to healthy controls (HC) after vaccination to elaborate the challenges and potential vulnerabilities faced by individuals with T1DM in the context of COVID-19 breakthrough infections, ultimately contributing to a more comprehensive understanding of vaccine efficacy and protection within this specific patient population.
Methodology
Study design
The second COVID-19 vaccination in autoimmune disease (COVAD-2), conducted in 2022, is a multinational, cross-sectional, patient-self-reported electronic survey [11]. This survey is a continuation of the first COVAD survey which was circulated in 2021 [9]. The first survey looked at short term vaccination-related adverse events, details of COVID-19 infection and health outcomes. The Checklist for Reporting Results of the Internet E-Surveys (CHERRIES) was adhered to when reporting the results [12]. The survey was administered to participants in over a 100 countries via > 150 centres. COVID-19 breakthrough infection (BI) data in patients with type 1 diabetes mellitus (T1D) and healthy controls (HC) was obtained.
Data collection
After obtaining approval from worldwide experts, conducting a pilot test, and having the materials translated into 18 different languages, we hosted two extensive questionnaires (COVAD-1 AND COVAD-2) on the website surveymonkey.com. The survey was distributed by the COVAD study group internationally. Disease-specific information, such as the type of autoimmune rheumatic diseases (AIRD), duration, clinical symptoms, current medications, and patient-reported outcomes, such as PROMIS) physical and mental function and quality of life (QoL) scores, were also gathered in addition to patient demographics and comorbidities [13]. The specific type of COVID-19 vaccination administered to the patients included Pfizer-BioNTech, Oxford/Astra Zeneca, Johnson & Johnson (J&J), Moderna, Novavax, Covishield (Serum Institute of India), Covaxin (Bharat Biotech), Sputnik, Sinopharm, and Sinovac-CoronaVac, among others. We gathered information on breakthrough COVID infections in type 1 diabetics and healthy controls after vaccination. The previously published COVAD study protocol contain a full description of survey methodologies and questions [14].
Data extraction
Following data collection, data were extracted from COVAD-2 database on 28th September 2022. Respondents excluded from the analysis are participants with incomplete responses, those who had their vaccinations in the middle of 2020 (likely trial participants), unvaccinated respondents, and those who had received only a single dose of any COVID-19 vaccine. We performed a subgroup analysis which included patients who had T1D from the cohort.
Statistical analysis
Descriptive data are expressed as frequencies (percentages) and medians (inter-quartile ranges). Chi-squared (χ2) and Mann–Whitney U tests were used to compare groups for categorical and continuous variables, respectively. Propensity score matching analysis among BI in T1D and HCs was performed with 1:1 match and emphasis on exact matches. The factors matched were age, gender, ethnicity, presence of any comorbidity, and mental health disorders. Statistical analyses were performed using IBM SPSS version 28.
Ethical considerations
Ethical approval was obtained from the Institutional Ethics Committee of SGPGIMS, Lucknow, India, and all participants consented electronically.
Results
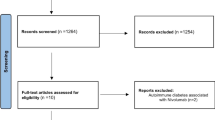
Of 10,783 respondents at the time of data analysis, 733 unvaccinated and 324 single-dose respondents, and 131 incomplete responses were excluded. Out of the total 9595 individuals included in the final analysis 3435 were HCs and 100 patients had the diagnosis of T1DM (Table S1 in the Supplementary File). Among the HCs, 467 (13.5%) reported experiencing a BI once and 124 (3.6%) reported experiencing a BI twice. Among the 100 T1DM patients (66% female, 46% Caucasian), 16 (16%) experienced one BI, and 2 (2%) experienced BI twice (Table S2 in the Supplementary File). The median time to symptom resolution was 10 days (2.5–72 days) in T1DM patients who received 2 doses of vaccine and 14 days (6 to 18 days) in those who received 3 doses. The most prevalent symptoms during the first BI in T1DM patients were cough (62.5%), fatigue (50%), fever (37.5%), and myalgia (31.25%). Only one patient required hospitalization for supplemental oxygen requirement, however, did not require intensive care.
Supplementary Table S3 presents the population characteristics of T1DM before and after PSM analysis. The results of PSM analysis indicated that individuals with T1DM had no statistically significant increase in the risk of experiencing one-time BI [T1DM vs HC OR 1.2 (0.7–2.0), p = 0.490] or two-time BI [T1DM vs HC OR 0.5 (0.1–2.2), p = 0.399] as compared to HCs (Table 1).
Discussion
In this study, we found no differences in the frequency, symptoms, duration, or critical care requirements between T1DM and HCs after COVID-19 vaccination. Individuals with SAIDs, including T1DM, may be at an increased risk of severe COVID-19 infection and associated morbidity and mortality when compared with healthy controls [15, 16]. This can be related to diabetes-associated hyperglycemia impairing the immune function and inducing immunosenescence [17, 18]. Moreover, hyperglycemia promotes viral replication via the production of mitochondrial reactive oxygen species and activation of hypoxia-inducible factor 1α [19], putting these patients at risk for BI even after full vaccination. A study by Basso et al. reported 2.41 times higher odds of contracting a BI in diabetics as compared to individuals without diabetes [20]. This contrasts with the reassuring findings of our study where there were no differences in the frequency of BIs between patients with T1DM and HCs. A recent study by Jia et al. reported similar humoral antibody responses to SARS-CoV-2 mRNA vaccines during 12 months of follow-up as well as similar rates of BI in T1DM patients and HCs [21].
Our study reported only mild symptoms following BI in T1DM patients including cough, fatigue, fever, and myalgia with only one patient requiring hospitalization for oxygen supplementation. The PRO-VACS 2 study reported mild COVID-19 symptoms including cough, cold, sore throat, and fever in cases of BI in 24 fully immunized T1DM patients [22]. The above study also did not show any major effect of BI on glycaemic control in vaccinated patients. Though some studies suggest a glycemic deterioration in diabetic patients following a COVID-19 infection [23], there is limited evidence regarding the effect of BI on glycemic control in these patients. In addition, a meta-analysis reported small improvements in various outcomes of glycemic control in diabetic patients with no statistically significant effect on HBA1c [24].
Our study did not find significant differences in BI frequency, clinical features, or severity between T1DM patients and HCs, reaffirming comparable protection provided by vaccinations in both groups [9, 25]. With limited evidence of vaccine effectiveness in T1DM available in literature, this brief report attempts to fill this gap and may serve as a valuable resource for clinicians when addressing patient inquiries regarding the frequency, severity, and time to resolution of BIs following COVID-19 vaccination. In addition, it can serve as a foundation for researchers to expand on the limited literature available on the impact of BIs on the long-term glycemic control of these patients, and the occurrence of any severe, life-threatening symptoms that may result from BIs.
COVAD group leads a large collaborative initiative with patient voice on COVID-19 infection and vaccination with data collected from over hundred countries, encompassing patient responses from a wide range of geographical areas. This is a major strength of our study. However, our study also has some limitations that should be considered. The sample size of T1DM patients is relatively small, data on glycaemic control was not captured by the survey, and there is the possibility of inadvertent introduction of recall and selection bias in the online survey. This anonymized, self-reported patient survey addresses the difference in perspective between patients and physicians about symptom burden and vaccine side effects. Understanding the patient’s perspective is imperative in addressing vaccine hesitancy. Further studies with larger sample sizes and longitudinal design may provide more definitive conclusions.
In conclusion, the results of our study suggest that vaccination against COVID-19 is as effective and safe in T1DM patients as compared to healthy controls. Further research to understand the factors associated with inadequate vaccine response in individuals with BIs will contribute to a more comprehensive understanding of BIs in the context of T1DM and autoimmune diseases.
Data availability
The datasets generated and/or analysed during the current study are not publicly available but are available from the corresponding author upon reasonable request.
References
E.H. Tan, A.G. Sena, A. Prats-Uribe, S.C. You, W.U.R. Ahmed, K. Kostka, C. Reich, S.L. Duvall, K.E. Lynch, M.E. Matheny, T. Duarte-Salles, S.F. Bertolin, G. Hripcsak, K. Natarajan, T. Falconer, M. Spotnitz, A. Ostropolets, C. Blacketer, T.M. Alshammari, H. Alghoul, O. Alser, J.C.E. Lane, D.M. Dawoud, K. Shah, Y. Yang, L. Zhang, C. Areia, A. Golozar, M. Recalde, P. Casajust, J. Jonnagaddala, V. Subbian, D. Vizcaya, L.Y.H. Lai, F. Nyberg, D.R. Morales, J.D. Posada, N.H. Shah, M. Gong, A. Vivekanantham, A. Abend, E.P. Minty, M. Suchard, P. Rijnbeek, P.B. Ryan, D. Prieto-Alhambra (2021) COVID-19 in patients with autoimmune diseases: characteristics and outcomes in a multinational network of cohorts across three countries. Rheumatology (Oxford) 60: SI37–SI50
Raiker R, DeYoung C, Pakhchanian H, Ahmed S, Kavadichanda C, Gupta L, Kardeş S (2021) Outcomes of COVID-19 in patients with rheumatoid arthritis: a multicenter research network study in the United States, Semin. Arthritis Rheum 51:1057–1066. https://doi.org/10.1016/J.SEMARTHRIT.2021.08.010
Boddu SK, Aurangabadkar G, Kuchay MS (2020) New onset diabetes, type 1 diabetes and COVID-19. Diabetes Metab Syndr 14:2211–2217. https://doi.org/10.1016/J.DSX.2020.11.012
Zóka A, Muzes G, Somogyi A, Varga T, Szémán B, Al-Aissa Z, Hadarits O, Firneisz G (2013) Altered immune regulation in type 1 diabetes. Clin Dev Immunol 2013:1–7. https://doi.org/10.1155/2013/254874
Melendez-Ramirez LY, Richards RJ, Cefalu WT (2010) Complications of type 1 diabetes. Endocrinol Metab Clin N Am 39:625–640. https://doi.org/10.1016/J.ECL.2010.05.009
Medeiros-Ribeiro AC, Aikawa NE, Saad CGS, Yuki EFN, Pedrosa T, Fusco SRG, Rojo PT, Pereira RMR, Shinjo SK, Andrade DCO, Sampaio-Barros PD, Ribeiro CT, Deveza GBH, Martins VAO, Silva CA, Lopes MH, Duarte AJS, Antonangelo L, Sabino EC, Kallas EG, Pasoto SG, Bonfa E (2021) Immunogenicity and safety of the CoronaVac inactivated vaccine in patients with autoimmune rheumatic diseases: a phase 4 trial. Nat Med 27:1744–1751. https://doi.org/10.1038/S41591-021-01469-5
Sattui SE, Liew JW, Kennedy K, Sirotich E, Putman M, Moni TT, Akpabio A, Alpízar-Rodríguez D, Berenbaum F, Bulina I, Conway R, Singh AD, Duff E, Durrant KL, Gheita TA, Hill CL, Howard RA, Hoyer BF, Hsieh E, El Kibbi L, Kilian A, Kim AH, Liew DFL, Lo C, Miller B, Mingolla S, Nudel M, Palmerlee CA, Singh JA, Singh N, Ugarte-Gil MF, Wallace J, Young KJ, Bhana S, Costello W, Grainger R, MacHado PM, Robinson PC, Sufka P, Wallace ZS, Yazdany J, Harrison C, Larché M, Levine M, Foster G, Thabane L, Rider LG, Hausmann JS, Simard JF, Sparks JA (2021) Early experience of COVID-19 vaccination in adults with systemic rheumatic diseases: results from the COVID-19 Global Rheumatology Alliance Vaccine Survey. RMD Open. https://doi.org/10.1136/RMDOPEN-2021-001814
Birhane M, Bressler S, Chang G, Clark T, Dorough L, Fischer M, Watkins LF, Goldstein JM, Kugeler K, Langley G, Lecy K, Martin S, Medalla F, Mitruka K, Nolen L, Sadigh K, Spratling R, Thompson G, Trujillo A (2021) COVID-19 vaccine breakthrough infections reported to CDC—United States, January 1–April 30, 2021. MMWR Morb Mortal Wkly Rep 70:792–793. https://doi.org/10.15585/MMWR.MM7021E3
Hoff LS, Ravichandran N, Shinjo SK, Day J, Sen P, Junior JG, Lilleker JB, Joshi M, Agarwal V, Kardes S, Kim M, Milchert M, Makol A, Gheita T, Salim B, Velikova T, Gracia-Ramos AE, Parodis I, O’Callaghan AS, Nikiphorou E, Tan AL, Chatterjee T, Cavagna L, Saavedra MA, Ziade N, Knitza J, Kuwana M, Nune A, Distler O, Cansu DÜ, Traboco L, Wibowo SAK, Tehozol EAZ, Serrano JR, De La Torre IG, Wincup C, Pauling JD, Chinoy H, Agarwal V, Aggarwal R, Gupta L (2023) COVID-19 severity and vaccine breakthrough infections in idiopathic inflammatory myopathies, other systemic autoimmune and inflammatory diseases, and healthy controls: a multicenter cross-sectional study from the COVID-19 Vaccination in Autoimmune Diseases (COVAD) survey. Rheumatol Int 43:47–58. https://doi.org/10.1007/S00296-022-05229-7
Gaur P, Agrawat H, Shukla A (2021) COVID-19 vaccine hesitancy in patients with systemic autoimmune rheumatic disease: an interview-based survey. Rheumatol Int 41:1601–1605. https://doi.org/10.1007/S00296-021-04938-9/FIGURES/1
Fazal ZZ, Sen P, Joshi M, Ravichandran N, Lilleker JB, Agarwal V, Kardes S, Kim M, Day J, Makol A, Milchert M, Gheita T, Salim B, Velikova T, Gracia-Ramos AE, Parodis I, Nikiphorou E, Tan AL, Chatterjee T, Cavagna L, Saavedra MA, Shinjo SK, Ziade N, Selva-O’Callaghan A, Nune A, Knitza J, Kuwana M, Gutiérrez CET, Caballero-Uribe CV, Dey D, Distler O, Chinoy H, Agarwal V, Aggarwal R, Gupta L, Barman B, Singh YP, Ranjan R, Jain A, Pandya SC, Pilania RK, Sharma A, Manesh Manoj M, Gupta V, Kavadichanda CG, Patro PS, Ajmani S, Phatak S, Goswami RP, Chowdhury AC, Mathew AJ, Shenoy P, Asranna A, Bommakanti KT, Shukla A, Pandey AKR, Gaur PS, Mamadapur M, Ghodke A, Chandwar K, Jagtap K, Cansu DÜ, Yildirim R, Patel A, Pauling JD, Wincup C, Giannini M, Maurier F, Campagne J, Meyer A, Del Papa N, Sambataro G, Fabiola A, Govoni M, Parisi S, Bocci EB, Sebastiani GD, Fusaro E, Sebastiani M, Quartuccio L, Franceschini F, Sainaghi PP, Orsolini G, De Angelis R, Danielli MG, Venerito V, Grignaschi S, Giollo A, Traboco LS, Shaharir SS, Wibowo SAK, Tehozol EAZ, Serrano JR, De La Torre IG, Colunga-Pedraza IJ, Colunga-Pedraza IJ, Merayo-Chalico J, Loarce-Martos J, Prieto-González S, Gil-Vila A, Aranega R, Hoff LS, Nakashima R, Sato S, Kimura N, Kaneko Y, Tomaras S, Proft FN, Holzer MT, Gromova MA, Aharonov O, Nagy-Vincze M, Griger Z, Hmamouchi I, El Bouchti PI, Baba Z, Ima-Edomwonyi U, Dedeke I, Airenakho E, Madu NH, Yerima A, Olaosebikan H, Chibuzo OC, Becky A, Koussougbo OD, Palalane E, Langguth D, Limaye V, Needham M, Srivastav N, Hudson M, Landon-Cardinal O, Zuleta WGR, Arbeláez Á, Cajas J, Silva JAP, Fonseca JE, Zimba O, Bohdana D, So H, Ugarte-Gil MF, Chinchay L, Bernaola JP, Pimentel V, Tanveer Hasan ATM, Saha S, Vaidya B, Fathi HM, Mohammed RHA, Chen YM, Harifi G, El Kibbi L, Halabi HM, Akawatcharangura P, Katchamart W, Fuentes-Silva Y, Cabriza K, Losanto J, Colaman N, Cachafeiro-Vilar A, Bautista GG, Ho EJG, González RA, Nunez LS, Cristian Vergara M, Báez JT, Alonzo H, Pastelin CBS, Salinas RG, Obiols AQ, Chávez N, Ordóñez AB, Argueta S, Quijivix D, Llerena GAR, Sierra-Zorita R, Arrieta D, Hidalgo ER, Saenz R, Idania Escalante M, Morales R, Calapaqui W, Quezada I, Arredondo G (2022) COVAD survey 2 long-term outcomes: unmet need and protocol. Rheumatol Int 42:2151–2158. https://doi.org/10.1007/S00296-022-05157-6
Eysenbach G (2004) Improving the quality of Web surveys: the Checklist for Reporting Results of Internet E-Surveys (CHERRIES). J Med Internet Res. https://doi.org/10.2196/JMIR.6.3.E34
PROMIS (n.d.) https://www.healthmeasures.net/explore-measurement-systems/promis. Accessed 5 Sept 2023
Sen P, Gupta L, Lilleker JB, Aggarwal V, Kardes S, Milchert M, Gheita T, Salim B, Velikova T, Gracia-Ramos AE, Parodis I, O’Callaghan AS, Nikiphorou E, Tan AL, Cavagna L, Saavedra MA, Shinjo SK, Ziade N, Knitza J, Kuwana M, Cagnotto G, Nune A, Distler O, Chinoy H, Aggarwal V, Aggarwal R, Barman B, Singh YP, Ranjan R, Jain A, Pandya SC, Malaviya AN, Pilania RK, Sharma A, Manoj MM, Gupta V, Kavadichanda CG, Patro PS, Ajmani S, Phatak S, Goswami RP, Chowdhury AC, Mathew AJ, Shenoy P, Asranna A, Bommakanti KT, Shukla A, Naveen R, Cansu DÜ, Pauling JD, Wincup C, Chatterjee T, Kim M, Giannini M, Del Papa N, Sambataro G, Fabiola A, Govoni M, Parisi S, Bocci EB, Sebastiani GD, Fusaro E, Sebastiani M, Quartuccio L, Franceschini F, Sainaghi PP, Orsolini G, De Angelis R, Danielli MG, Traboco LS, Wibowo SAK, Serrano JR, De La Torre IG, Tehozol EAZ, Loarce-Martos J, Prieto-González S, Gil-Vila A, Aranega R, Nakashima R, Sato S, Kimura N, Kaneko Y, Tomaras S, Gromova MA, Aharonov O (2022) COVID-19 vaccination in autoimmune disease (COVAD) survey protocol. Rheumatol Int 42:23–29. https://doi.org/10.1007/S00296-021-05046-4
Xu C, Yi Z, Cai R, Chen R, Thong BYH, Mu R (2021) Clinical outcomes of COVID-19 in patients with rheumatic diseases: a systematic review and meta-analysis of global data. Autoimmun Rev. https://doi.org/10.1016/J.AUTREV.2021.102778
Conway R, Grimshaw AA, Konig MF, Putman M, Duarte-García A, Tseng LY, Cabrera DM, Chock YPE, Degirmenci HB, Duff E, Egeli BH, Graef ER, Gupta A, Harkins P, Hoyer BF, Jayatilleke A, Jin S, Kasia C, Khilnani A, Kilian A, Kim AHJ, Lin CMA, Low C, Proulx L, Sattui SE, Singh N, Sparks JA, Tam H, Ugarte-Gil MF, Ung N, Wang K, Wise LM, Yang Z, Young KJ, Liew JW, Grainger R, Wallace ZS, Hsieh E (2022) SARS-CoV-2 infection and COVID-19 outcomes in rheumatic diseases: a systematic literature review and meta-analysis. Arthritis Rheumatol 74:766–775. https://doi.org/10.1002/ART.42030/ABSTRACT
Berbudi A, Rahmadika N, Tjahjadi AI, Ruslami R (2020) Type 2 diabetes and its impact on the immune system. Curr Diabetes Rev 16:442–449. https://doi.org/10.2174/1573399815666191024085838
Stefan N (2022) Metabolic disorders, COVID-19 and vaccine-breakthrough infections. Nat Rev Endocrinol 18:75–76. https://doi.org/10.1038/S41574-021-00608-9
Lim S, Bae JH, Kwon HS, Nauck MA (2020) COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nat Rev Endocrinol 171:11–30. https://doi.org/10.1038/s41574-020-00435-4
Basso P, Negro C, Cegolon L, Larese Filon F (2022) Risk of vaccine breakthrough SARS-CoV-2 infection and associated factors in healthcare workers of Trieste Teaching Hospitals (North-Eastern Italy). Viruses. https://doi.org/10.3390/V14020336
Jia X, Dong F, Pyle L, Michels AW, Yu L, Rewers M (2023) Similar time course of humoral response to SARS-CoV-2 mRNA vaccines in people with and without type 1 diabetes. Diabetes Technol Ther 25:492–496. https://doi.org/10.1089/DIA.2023.0016
Dicembrini I, Scoccimarro D, Del Vescovo GG, Marinelli C, Zago E, Poggi CD, Pala L, Mannucci E (2022) Patient-reported outcomes of COVID-19 vaccine breakthrough infection-associated changes in glucose control in subjects with type 1 diabetes (PRO-VACS 2 study). Diabetes Care 46:e8–e9. https://doi.org/10.2337/DC22-0868
Tanji Y, Sawada S, Watanabe T, Mita T, Kobayashi Y, Murakami T, Metoki H, Akai H (2021) Impact of COVID-19 pandemic on glycemic control among outpatients with type 2 diabetes in Japan: a hospital-based survey from a country without lockdown. Diabetes Res Clin Pract 176:108840. https://doi.org/10.1016/J.DIABRES.2021.108840
O’Mahoney LL, Highton PJ, Kudlek L, Morgan J, Lynch R, Schofield E, Sreejith N, Kapur A, Otunla A, Kerneis S, James O, Rees K, Curtis F, Khunti K, Hartmann-Boyce J (2022) The impact of the COVID-19 pandemic on glycaemic control in people with diabetes: a systematic review and meta-analysis. Diabetes Obes Metab 24:1850–1860. https://doi.org/10.1111/DOM.14771
Cook C, Patel NJ, D’Silva KM, Hsu TYT, DiIorio M, Prisco L, Martin LW, Vanni K, Zaccardelli A, Todd D, Sparks JA, Wallace ZS (2022) Clinical characteristics and outcomes of COVID-19 breakthrough infections among vaccinated patients with systemic autoimmune rheumatic diseases. Ann Rheum Dis 81:289–291. https://doi.org/10.1136/ANNRHEUMDIS-2021-221326
Acknowledgements
The authors are grateful to all respondents for completing the questionnaire. The authors also thank the Myositis Association, Myositis India, Myositis UK, Myositis Support and Understanding, the Myositis Global Network, Deutsche Gesellschaft für Muskelkranke e.V. (DGM), Dutch and Swedish Myositis patient support groups, Cure JM, Cure IBM, Sjögren’s India Foundation, Patients Engage, Scleroderma India, Lupus UK, Lupus Sweden, Emirates Arthritis Foundation, EULAR PARE, ArLAR research group, AAAA patient group, Myositis Association of Australia, APLAR myositis special interest group, Thai Rheumatism association, PANLAR, AFLAR NRAS, Anti-Synthetase Syndrome support group, and various other patient support groups and organizations for their contribution to the dissemination of this survey. Finally, the authors wish to thank all members of the COVAD study group for their invaluable role in the data collection. COVAD Study Group Authors: Parikshit Sen, Jessica Day, Elena Nikiphorou, Nelly Ziade, Marcin Milchert, Lorenzo Cavagna, Yi Ming Chen, Ashima Makol, Vishwesh Agarwal, Aarat Patel, John D Pauling, Chris Wincup, Erick Adrian Zamora Tehozol, Jorge Rojas Serrano, Ignacia García-De La Torre, Iris J. Colunga-Pedraza, Javier Merayo-Chalico, Okwara Celestine Chibuzo, Wanruchada Katchamart, Phonpen Akawatcharangura Goo, Russka Shumnalieva, Leonardo Santos Hoff, Lina El Kibbi, Hussein Halabi, Binit Vaidya, Syahrul Sazliyana Shaharir, A T M Tanveer Hasan, Dzifa Dey, Carlos Enrique Toro Gutiérrez, James B. Lilleker, Babur Salim, Tamer Gheita, Oliver Distler, Miguel A Saavedra, Sinan Kardes, Laura Andreoli, Daniele Lini, Karen Schreiber, Melinda Nagy Vince, Yogesh Preet Singh, Rajiv Ranjan, Avinash Jain, Sapan C Pandya, Rakesh Kumar Pilania, Aman Sharma, Manesh Manoj M, Vikas Gupta, Chengappa G Kavadichanda, Pradeepta Sekhar Patro, Sajal Ajmani, Sanat Phatak, Rudra Prosad Goswami, Abhra Chandra Chowdhury, Ashish Jacob Mathew, Padnamabha Shenoy, Ajay Asranna, Keerthi Talari Bommakanti, Anuj Shukla, Arunkumar R Pande, Kunal Chandwar, Akanksha Ghodke, Zoha Zahid Fazal, Döndü Üsküdar Cansu, Reşit Yıldırım, Armen Yuri Gasparyan, Nicoletta Del Papa, Gianluca Sambataro, Atzeni Fabiola, Marcello Govoni, Simone Parisi, Elena Bartoloni Bocci, Gian Domenico Sebastiani, Enrico Fusaro, Marco Sebastiani, Luca Quartuccio, Franco Franceschini, Pier Paolo Sainaghi, Giovanni Orsolini, Rossella De Angelis, Maria Giovanna Danielli, Vincenzo Venerito, Silvia Grignaschi, Alessandro Giollo, Alessia Alluno, Florenzo Ioannone, Marco Fornaro, Lisa S Traboco, Suryo Anggoro Kusumo Wibowo, Jesús Loarce-Martos, Sergio Prieto-González, Raquel Aranega Gonzalez, Akira Yoshida, Ran Nakashima, Shinji Sato, Naoki Kimura, Yuko Kaneko, Takahisa Gono, Stylianos Tomaras, Fabian Nikolai Proft, Marie-Therese Holzer, Margarita Aleksandrovna Gromova, Or Aharonov, Zoltán Griger, Ihsane Hmamouchi, Imane El bouchti, Zineb Baba, Margherita Giannini, François Maurier, Julien Campagne, Alain Meyer, Daman Langguth, Vidya Limaye, Merrilee Needham, Nilesh Srivastav, Marie Hudson, Océane Landon-Cardinal, Wilmer Gerardo Rojas Zuleta, Álvaro Arbeláez, Javier Cajas, José António Pereira Silva, João Eurico Fonseca, Olena Zimba, Doskaliuk Bohdana, Uyi Ima-Edomwonyi, Ibukunoluwa Dedeke, Emorinken Airenakho, Nwankwo Henry Madu, Abubakar Yerima, Hakeem Olaosebikan, Becky A., Oruma Devi Koussougbo, Elisa Palalane, Ho So, Manuel Francisco Ugarte-Gil, Lyn Chinchay, José Proaño Bernaola, Victorio Pimentel, Hanan Mohammed Fathi, Reem Hamdy A Mohammed, Ghita Harifi, Yurilís Fuentes-Silva, Karoll Cabriza, Jonathan Losanto, Nelly Colaman, Antonio Cachafeiro-Vilar, Generoso Guerra Bautista, Enrique Julio Giraldo Ho, Raúl González, Lilith Stange Nunez, Cristian Vergara M, Jossiell Then Báez, Hugo Alonzo, Carlos Benito Santiago Pastelin, Rodrigo García Salinas, Alejandro Quiñónez Obiols, Nilmo Chávez, Andrea Bran Ordóñez, Gil Alberto Reyes Llerena, Radames Sierra-Zorita, Dina Arrieta, Eduardo Romero Hidalgo, Ricardo Saenz, Idania Escalante M, Wendy Calapaqui, Ivonne Quezada, Gabriela Arredondo
Author information
Authors and Affiliations
Consortia
Contributions
Conceptualization: LG, VA, TC. Data curation: all authors. Formal analysis: NR; Interpretation of data: All authors; Funding acquisition: N/A. Investigation: LG, VA, TC. Methodology: LG, VA; Software: LG. Validation: VA and RA; Visualization: RA, VA, and LG. Writing-original draft: SP, TC. Writing- review and editing: all authors; Accountability for integrity/accuracy of paper: All authors; Formal approval for publication: all authors.
Corresponding author
Ethics declarations
Conflicts of interest
ALT has received honoraria for advisory boards and speaking for Abbvie, Gilead, Janssen, Lilly, Novartis, Pfizer, and UCB. IP has received research funding and/or honoraria from Amgen, AstraZeneca, Aurinia Pharmaceuticals, Elli Lilly and Company, Gilead Sciences, GlaxoSmithKline, Janssen Pharmaceuticals, Novartis and F. Hoffmann-La Roche AG. MK has received speaker honoraria/participated in advisory boards for Abbvie, Asahi-Kasei, Astellas, AstraZeneca, Boehringer-Ingelheim, Chugai, Corbus, Eisai, GSK, Horizon, Kissei, BML, Mochida, Nippon Shinyaku, Ono Pharmaceuticals, Tanabe-Mitsubishi. RA has a consultancy relationship with and/or has received research funding from the following companies: Bristol Myers-Squibb, Pfizer, Genentech, Octapharma, CSL Behring, Mallinckrodt, AstraZeneca, Corbus, Kezar, Abbvie, Janssen, Kyverna Alexion, Argenx, Q32, EMD-Serono, Boehringer Ingelheim, Roivant, Merck, Galapagos, Actigraph, Scipher, Horizon Therepeutics, Teva, Beigene, ANI Pharmaceuticals, Biogen, Nuvig, Capella Bioscience, and CabalettaBio. TV has received speaker honoraria from Pfizer and AstraZeneca, non-related to the current manuscript. Rest of the authors have no conflict of interest relevant to this manuscript.
Ethical approval
Ethical approval was obtained from the Institutional Ethics Committee of the Sanjay Gandhi Postgraduate Institute of Medical Sciences, Raebareli Road, Lucknow, 226014 (IEC Code: 2021-143-IP-EXP-39).
Disclaimer
No part of this manuscript has been copied or published elsewhere either in whole or in part.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The complete list of authors part of the COVAD Study Group as well as their affiliations are provided in the supplement.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Panchawagh, S., Ravichandran, N., Barman, B. et al. COVID-19 breakthrough infections in type 1 diabetes mellitus: a cross-sectional study by the COVID-19 Vaccination in Autoimmune Diseases (COVAD) Group. Rheumatol Int 44, 73–80 (2024). https://doi.org/10.1007/s00296-023-05496-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-023-05496-y