Abstract
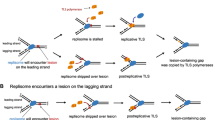
We use genetic assays to suggest that transcription-coupled repair or new origin formation in Escherichia coli involves removal of RNAP to create an RNA primer for DNA synthesis. Transcription factor DksA was shown to play a role in numerous reactions involving RNA polymerase. Some, but not all, of the activities of DksA at promoters or during transcription elongation require (p)ppGpp. In addition to its role during transcription, DksA is also involved in maintaining genome integrity. Cells lacking DksA are sensitive to multiple DNA damaging agents including UV light, ionizing radiation, mitomycin C, and nalidixic acid. Here, we focus on two recent studies addressing the importance of DksA in the repair of double-strand breaks (DSBs), one by Sivaramakrishnan et al. (Nature 550:214–218, 2017) and one originating in our laboratory, Myka et al. (Mol Microbiol 111:1382–1397. https://doi.org/10.1111/mmi.14227, 2019). It appears that depending on the type and possibly location of DNA damage, DksA can play either a passive or an active role in DSB repair. The passive role relies on exclusion of anti-backtracking factors from the RNAP secondary channel. The exact mechanism of active DksA-mediated DNA repair is unknown. However, DksA was proposed to destabilize transcription complexes, thus clearing the way for recombination and DNA repair. Based on the requirement for DksA, both in repair of DSBs and the R-loop-dependent formation of new origins of DNA replication, we propose that DksA may allow for removal of RNAP without unwinding of the RNA:DNA hybrid, which can then be extended by a DNA polymerase. This mechanism obviates the need for RNAP backtracking to repair damaged DNA.
Similar content being viewed by others
References
Adebali O, Sancar A, Selby CP (2017) Mfd translocase is necessary and sufficient for transcription-coupled repair in Escherichia coli. J Biol Chem 292:18386–18391. https://doi.org/10.1074/jbc.C117.818807
Aparicio T, Baer R, Gottesman M, Gautier J (2016) MRN, CtIP, and BRCA1 mediate repair of topoisomerase II-DNA adducts. J Cell Biol 212:399–408. https://doi.org/10.1083/jcb.201504005
Baharoglu Z, Lestini R, Duigou S, Michel B (2010) RNA polymerase mutations that facilitate replication progression in the rep uvrD recF mutant lacking two accessory replicative helicases. Mol Microbiol 77:324–336. https://doi.org/10.1111/j.1365-2958.2010.07208.x
Borukhov S, Sagitov V, Goldfarb A (1993) Transcript cleavage factors from E. coli. Cell 72:459–466
Epshtein V et al (2014) UvrD facilitates DNA repair by pulling RNA polymerase backwards. Nature 505:372–377. https://doi.org/10.1038/nature12928
Freudenreich CH (2018) R-loops: targets for nuclease cleavage and repeat instability. Curr Genet 64:789–794. https://doi.org/10.1007/s00294-018-0806-z
Furman R, Sevostyanova A, Artsimovitch I (2012) Transcription initiation factor DksA has diverse effects on RNA chain elongation. Nucleic Acids Res 40:3392–3402. https://doi.org/10.1093/nar/gkr1273
Furman R, Tsodikov OV, Wolf YI, Artsimovitch I (2013) An insertion in the catalytic trigger loop gates the secondary channel of RNA polymerase. J Mol Biol 425:82–93. https://doi.org/10.1016/j.jmb.2012.11.008
Gaca AO, Colomer-Winter C, Lemos JA (2015) Many means to a common end: the intricacies of (p)ppGpp metabolism and its control of bacterial homeostasis. J Bacteriol 197:1146–1156. https://doi.org/10.1128/JB.02577-14
Gourse RL, Chen AY, Gopalkrishnan S, Sanchez-Vazquez P, Myers A, Ross W (2018) Transcriptional responses to ppGpp and DksA. Annu Rev Microbiol 72:163–184. https://doi.org/10.1146/annurev-micro-090817-062444
Hauryliuk V, Atkinson GC, Murakami KS, Tenson T, Gerdes K (2015) Recent functional insights into the role of (p)ppGpp in bacterial physiology. Nat Rev Microbiol 13:298–309. https://doi.org/10.1038/nrmicro3448
Kamarthapu V, Epshtein V, Benjamin B, Proshkin S, Mironov A, Cashel M, Nudler E (2016) ppGpp couples transcription to DNA repair in E. coli. Science 352:993–996. https://doi.org/10.1126/science.aad6945
Kogoma T (1997) Stable DNA replication: interplay between DNA replication, homologous recombination, and transcription. Microbiol Mol Biol Rev 61:212–238
Kogoma T, Maldonado RR (1997) DNA polymerase I in constitutive stable DNA replication in Escherichia coli. J Bacteriol 179:2109–2115
Maduike NZ, Tehranchi AK, Wang JD, Kreuzer KN (2014) Replication of the Escherichia coli chromosome in RNase HI-deficient cells: multiple initiation regions and fork dynamics. Mol Microbiol 91:39–56. https://doi.org/10.1111/mmi.12440
Meddows TR, Savory AP, Grove JI, Moore T, Lloyd RG (2005) RecN protein and transcription factor DksA combine to promote faithful recombinational repair of DNA double-strand breaks. Mol Microbiol 57:97–110. https://doi.org/10.1111/j.1365-2958.2005.04677.x
Mikolaskova B, Jurcik M, Cipakova I, Kretova M, Chovanec M, Cipak L (2018) Maintenance of genome stability: the unifying role of interconnections between the DNA damage response and RNA-processing pathways. Curr Genet 64:971–983. https://doi.org/10.1007/s00294-018-0819-7
Molodtsov V, Sineva E, Zhang L, Huang X, Cashel M, Ades SE, Murakami KS (2018) Allosteric effector ppGpp potentiates the inhibition of transcript initiation by DksA. Mol Cell 69(828–839):e825. https://doi.org/10.1016/j.molcel.2018.01.035
Myka KK, Kusters K, Washburn R, Gottesman ME (2019) DksA-RNA polymerase interactions support new origin formation and DNA repair in Escherichia coli. Mol Microbiol 111:1382–1397. https://doi.org/10.1111/mmi.14227
Nakayama H (1975) Phleomycin-induced DNA lesions and their repair in Escherichia coli K12. Basic Life Sci 5B:483–485
Ogawa T, Pickett GG, Kogoma T, Kornberg A (1984) RNase H confers specificity in the dnaA-dependent initiation of replication at the unique origin of the Escherichia coli chromosome in vivo and in vitro. Proc Natl Acad Sci USA 81:1040–1044
Okazaki R, Arisawa M, Sugino A (1971) Slow joining of newly replicated DNA chains in DNA polymerase I-deficient Escherichia coli mutants. Proc Natl Acad Sci USA 68:2954–2957
Orlova M, Newlands J, Das A, Goldfarb A, Borukhov S (1995) Intrinsic transcript cleavage activity of RNA polymerase. Proc Natl Acad Sci USA 92:4596–4600
Parshin A, Shiver AL, Lee J, Ozerova M, Schneidman-Duhovny D, Gross CA, Borukhov S (2015) DksA regulates RNA polymerase in Escherichia coli through a network of interactions in the secondary channel that includes Sequence Insertion 1. Proc Natl Acad Sci USA 112:E6862–E6871. https://doi.org/10.1073/pnas.1521365112
Perederina A, Svetlov V, Vassylyeva MN, Tahirov TH, Yokoyama S, Artsimovitch I, Vassylyev DG (2004) Regulation through the secondary channel-structural framework for ppGpp-DksA synergism during transcription. Cell 118:297–309. https://doi.org/10.1016/j.cell.2004.06.030
Pomerantz RT, O’Donnell M (2008) The replisome uses mRNA as a primer after colliding with RNA polymerase. Nature 456:762–766. https://doi.org/10.1038/nature07527
Roghanian M, Zenkin N, Yuzenkova Y (2015) Bacterial global regulators DksA/ppGpp increase fidelity of transcription. Nucleic Acids Res 43:1529–1536. https://doi.org/10.1093/nar/gkv003
Ross W, Vrentas CE, Sanchez-Vazquez P, Gaal T, Gourse RL (2013) The magic spot: a ppGpp binding site on E. coli RNA polymerase responsible for regulation of transcription initiation. Mol Cell 50:420–429. https://doi.org/10.1016/j.molcel.2013.03.021
Ross W, Sanchez-Vazquez P, Chen AY, Lee JH, Burgos HL, Gourse RL (2016) ppGpp binding to a site at the RNAP-DksA interface accounts for its dramatic effects on transcription initiation during the stringent response. Mol Cell 62:811–823. https://doi.org/10.1016/j.molcel.2016.04.029
Rutherford ST, Villers CL, Lee JH, Ross W, Gourse RL (2009) Allosteric control of Escherichia coli rRNA promoter complexes by DksA. Genes Dev 23:236–248. https://doi.org/10.1101/gad.1745409
Satory D, Gordon AJ, Wang M, Halliday JA, Golding I, Herman C (2015) DksA involvement in transcription fidelity buffers stochastic epigenetic change. Nucleic Acids Res 43:10190–10199. https://doi.org/10.1093/nar/gkv839
Sivaramakrishnan P et al (2017) The transcription fidelity factor GreA impedes DNA break repair. Nature 550:214–218. https://doi.org/10.1038/nature23907
Sleigh MJ (1976) The mechanism of DNA breakage by phleomycin in vitro. Nucleic Acids Res 3:891–901
Tehranchi AK et al (2010) The transcription factor DksA prevents conflicts between DNA replication and transcription machinery. Cell 141:595–605. https://doi.org/10.1016/j.cell.2010.03.036
Vinella D, Potrykus K, Murphy H, Cashel M (2012) Effects on growth by changes of the balance between GreA, GreB, and DksA suggest mutual competition and functional redundancy in Escherichia coli. J Bacteriol 194:261–273. https://doi.org/10.1128/JB.06238-11
Zhang Y, Mooney RA, Grass JA, Sivaramakrishnan P, Herman C, Landick R, Wang JD (2014) DksA guards elongating RNA polymerase against ribosome-stalling-induced arrest. Mol Cell 53:766–778. https://doi.org/10.1016/j.molcel.2014.02.005
Acknowledgements
Research in our laboratory is supported by NIH Grant no. GM37219 awarded to MEG.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Myka, K.K., Gottesman, M.E. DksA and DNA double-strand break repair. Curr Genet 65, 1297–1300 (2019). https://doi.org/10.1007/s00294-019-00983-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-019-00983-x