Abstract
Sister chromatid cohesion is essential for chromosome segregation both in mitosis and meiosis. Cohesion between two chromatids is mediated by a protein complex called cohesin. The loading and unloading of the cohesin are tightly regulated during the cell cycle. In vertebrate cells, cohesin is released from chromosomes by two distinct pathways. The best characterized pathway occurs at the onset of anaphase, when the kleisin component of the cohesin is destroyed by a protease, separase. The cleavage of the cohesin by separase releases entrapped sister chromatids allowing anaphase to commence. In addition, prior to the metaphase–anaphase transition, most of cohesin is removed from chromosomes in a cleavage-independent manner. This cohesin release is referred to as the prophase pathway. In meiotic cells, sister chromatid cohesion is essential for the segregation of homologous chromosomes during meiosis I. Thus, it was assumed that the prophase pathway for cohesin removal from chromosome arms would be suppressed during meiosis to avoid errors in chromosome segregation. However, recent studies revealed the presence of a meiosis-specific prophase-like pathway for cleavage-independent removal of cohesin during late prophase I in different organisms. In budding yeast, the cleavage-independent removal of cohesin is mediated through meiosis-specific phosphorylation of cohesin subunits, Rec8, the meiosis-specific kleisin, and the yeast Wapl ortholog, Rad61/Wpl1. This pathway plays a role in chromosome morphogenesis during late prophase I, promoting chromosome compaction. In this review, we give an overview of the prophase pathway for cohesin dynamics during meiosis, which has a complex regulation leading to differentially localized populations of cohesin along meiotic chromosomes.



Similar content being viewed by others
References
Alavattam KG, Maezawa S, Sakashita A, Khoury H, Barski A, Kaplan N, Namekawa SH (2019) Attenuated chromatin compartmentalization in meiosis and its maturation in sperm development. Nat Struct Mol Biol. https://doi.org/10.1038/s41594-019-0189-y
Arguello-Miranda O et al (2017) Casein kinase 1 coordinates cohesin cleavage, gametogenesis, and exit from M phase in meiosis II. Dev Cell 40:37–52. https://doi.org/10.1016/j.devcel.2016.11.021
Argunhan B et al (2017) Fundamental cell cycle kinases collaborate to ensure timely destruction of the synaptonemal complex during meiosis. EMBO J 36:2488–2509. https://doi.org/10.15252/embj.201695895
Attner MA, Miller MP, Ee LS, Elkin SK, Amon A (2013) Polo kinase Cdc5 is a central regulator of meiosis I. Proc Natl Acad Sci USA 110:14278–14283. https://doi.org/10.1073/pnas.1311845110
Beckouet F et al (2016) Releasing activity disengages cohesin’s Smc3/Scc1 interface in a process blocked by acetylation. Mol Cell 61:563–574. https://doi.org/10.1016/j.molcel.2016.01.026
Brar GA, Kiburz BM, Zhang Y, Kim JE, White F, Amon A (2006) Rec8 phosphorylation and recombination promote the step-wise loss of cohesins in meiosis. Nature 441:532–536. https://doi.org/10.1038/nature04794
Brieno-Enriquez MA et al (2016) Cohesin removal along the chromosome arms during the first meiotic division depends on a NEK1-PP1gamma-WAPL axis in the mouse. Cell Rep 17:977–986. https://doi.org/10.1016/j.celrep.2016.09.059
Buonomo SB, Clyne RK, Fuchs J, Loidl J, Uhlmann F, Nasmyth K (2000) Disjunction of homologous chromosomes in meiosis I depends on proteolytic cleavage of the meiotic cohesin Rec8 by separin. Cell 103:387–398
Burkhardt S et al (2016) Chromosome cohesion established by Rec8-cohesin in fetal oocytes is maintained without detectable turnover in oocytes arrested for months in Mice. Curr Biol 26:678–685. https://doi.org/10.1016/j.cub.2015.12.073
Challa K, Lee MS, Shinohara M, Kim KP, Shinohara A (2016) Rad61/Wpl1 (Wapl), a cohesin regulator, controls chromosome compaction during meiosis. Nucleic Acids Res 44:3190–3203. https://doi.org/10.1093/nar/gkw034
Challa K, Fajish VG, Shinohara M, Klein F, Gasser SM, Shinohara A (2019) Meiosis-specific prophase-like pathway controls cleavage-independent release of cohesin by Wapl phosphorylation. PLoS Genet 15:e1007851. https://doi.org/10.1371/journal.pgen.1007851
Clift D, Bizzari F, Marston AL (2009) Shugoshin prevents cohesin cleavage by PP2A(Cdc55)-dependent inhibition of separase. Genes Dev 23:766–780. https://doi.org/10.1101/gad.507509
Crawley O et al (2016) Cohesin-interacting protein WAPL-1 regulates meiotic chromosome structure and cohesion by antagonizing specific cohesin complexes. Elife 5:e10851. https://doi.org/10.7554/eLife.10851
de Carvalho CE, Zaaijer S, Smolikov S, Gu Y, Schumacher JM, Colaiacovo MP (2008) LAB-1 antagonizes the Aurora B kinase in C. elegans. Genes Dev 22:2869–2885. https://doi.org/10.1101/gad.1691208
De K, Sterle L, Krueger L, Yang X, Makaroff CA (2014) Arabidopsis thaliana WAPL is essential for the prophase removal of cohesin during meiosis. PLoS Genet 10:e1004497. https://doi.org/10.1371/journal.pgen.1004497
Ferrandiz N et al (2018) Spatiotemporal regulation of Aurora B recruitment ensures release of cohesion during C. elegans oocyte meiosis. Nat Commun 9:834. https://doi.org/10.1038/s41467-018-03229-5
Gandhi R, Gillespie PJ, Hirano T (2006) Human Wapl is a cohesin-binding protein that promotes sister-chromatid resolution in mitotic prophase. Curr Biol 16:2406–2417. https://doi.org/10.1016/j.cub.2006.10.061
Gligoris TG et al (2014) Closing the cohesin ring: structure and function of its Smc3-kleisin interface. Science 346:963–967. https://doi.org/10.1126/science.1256917
Gruber S, Haering CH, Nasmyth K (2003) Chromosomal cohesin forms a ring. Cell 112:765–777
Haarhuis JH, Elbatsh AM, Rowland BD (2014) Cohesin and its regulation: on the logic of X-shaped chromosomes. Dev Cell 31:7–18. https://doi.org/10.1016/j.devcel.2014.09.010
Hara K et al (2014) Structure of cohesin subcomplex pinpoints direct shugoshin-Wapl antagonism in centromeric cohesion. Nat Struct Mol Biol 21:864–870. https://doi.org/10.1038/nsmb.2880
Hassler M, Shaltiel IA, Haering CH (2018) Towards a unified model of SMC complex function. Curr Biol 28:R1266–r1281. https://doi.org/10.1016/j.cub.2018.08.034
Herbert M, Kalleas D, Cooney D, Lamb M, Lister L (2015) Meiosis and maternal aging: insights from aneuploid oocytes and trisomy births. Cold Spring Harb Perspect Biol 7:a017970. https://doi.org/10.1101/cshperspect.a017970
Hong S, Joo JH, Yun H, Kim K (2019) The nature of meiotic chromosome dynamics and recombination in budding yeast. J Microbiol. https://doi.org/10.1007/s12275-019-8541-9
Ishiguro KI (2019) The cohesin complex in mammalian meiosis. Genes Cells 24:6–30. https://doi.org/10.1111/gtc.12652
Ishiguro T, Tanaka K, Sakuno T, Watanabe Y (2010) Shugoshin-PP2A counteracts casein-kinase-1-dependent cleavage of Rec8 by separase. Nat Cell Biol 12:500–506. https://doi.org/10.1038/ncb2052
Ishiguro K, Kim J, Fujiyama-Nakamura S, Kato S, Watanabe Y (2011) A new meiosis-specific cohesin complex implicated in the cohesin code for homologous pairing. EMBO Rep 12:267–275. https://doi.org/10.1038/embor.2011.2
Jin H, Guacci V, Yu HG (2009) Pds5 is required for homologue pairing and inhibits synapsis of sister chromatids during yeast meiosis. J Cell Biol 186:713–725. https://doi.org/10.1083/jcb.200810107
Kakui Y, Uhlmann F (2018) SMC complexes orchestrate the mitotic chromatin interaction landscape. Curr Genet 64:335–339. https://doi.org/10.1007/s00294-017-0755-y
Katis VL et al (2010) Rec8 phosphorylation by casein kinase 1 and Cdc7-Dbf4 kinase regulates cohesin cleavage by separase during meiosis. Dev Cell 18:397–409
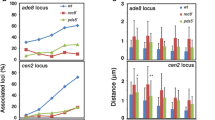
Kiburz BM et al (2005) The core centromere and Sgo1 establish a 50-kb cohesin-protected domain around centromeres during meiosis I. Genes Dev 19:3017–3030. https://doi.org/10.1101/gad.1373005
Kitajima TS, Sakuno T, Ishiguro K, Iemura S, Natsume T, Kawashima SA, Watanabe Y (2006) Shugoshin collaborates with protein phosphatase 2A to protect cohesin. Nature 441:46–52. https://doi.org/10.1038/nature04663
Kleckner N (2006) Chiasma formation: chromatin/axis interplay and the role(s) of the synaptonemal complex. Chromosoma 115:175–194
Klein F, Laroche T, Cardenas ME, Hofmann JF, Schweizer D, Gasser SM (1992) Localization of RAP1 and topoisomerase II in nuclei and meiotic chromosomes of yeast. J Cell Biol 117:935–948
Klein F, Mahr P, Galova M, Buonomo SB, Michaelis C, Nairz K, Nasmyth K (1999) A central role for cohesins in sister chromatid cohesion, formation of axial elements, and recombination during yeast meiosis. Cell 98:91–103. https://doi.org/10.1016/S0092-8674(00)80609-1
Kueng S, Hegemann B, Peters BH, Lipp JJ, Schleiffer A, Mechtler K, Peters JM (2006) Wapl controls the dynamic association of cohesin with chromatin. Cell 127(5):955–967. https://doi.org/10.1016/j.cell.2006.09.040
Lee BH, Amon A (2003) Role of Polo-like kinase CDC5 in programming meiosis I chromosome segregation. Science 300:482–486. https://doi.org/10.1126/science.1081846
Lee J, Hirano T (2011) RAD21L, a novel cohesin subunit implicated in linking homologous chromosomes in mammalian meiosis. J Cell Biol 192:263–276. https://doi.org/10.1083/jcb.201008005
Lin SJ, O’Connell MJ (2017) DNA Topoisomerase II modulates acetyl-regulation of cohesin-mediated chromosome dynamics. Curr Genet 63:923–930. https://doi.org/10.1007/s00294-017-0691-x
Litwin I, Wysocki R (2018) New insights into cohesin loading. Curr Genet 64:53–61. https://doi.org/10.1007/s00294-017-0723-6
Lopez-Serra L, Lengronne A, Borges V, Kelly G, Uhlmann F (2013) Budding yeast Wapl controls sister chromatid cohesion maintenance and chromosome condensation. Curr Biol 23:64–69. https://doi.org/10.1016/j.cub.2012.11.030
Mainiero S, Pawlowski WP (2014) Meiotic chromosome structure and function in plants. Cytogenet Genome Res 143:6–17. https://doi.org/10.1159/000365260
Marston AL (2014) Chromosome segregation in budding yeast: sister chromatid cohesion and related mechanisms. Genetics 196:31–63. https://doi.org/10.1534/genetics.112.145144
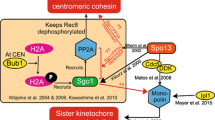
Mehta G, Anbalagan GK, Bharati AP, Gadre P, Ghosh SK (2018) An interplay between Shugoshin and Spo13 for centromeric cohesin protection and sister kinetochore mono-orientation during meiosis I in Saccharomyces cerevisiae. Curr Genet 64:1141–1152. https://doi.org/10.1007/s00294-018-0832-x
Morales C, Losada A (2018) Establishing and dissolving cohesion during the vertebrate cell cycle. Curr Opin Cell Biol 52:51–57. https://doi.org/10.1016/j.ceb.2018.01.010
Muller H et al (2018) Characterizing meiotic chromosomes’ structure and pairing using a designer sequence optimized for Hi-C. Mol Syst Biol 14:e8293. https://doi.org/10.15252/msb.20188293
Nagaoka SI, Hassold TJ, Hunt PA (2012) Human aneuploidy: mechanisms and new insights into an age-old problem Nature reviews. Genetics 13:493–504. https://doi.org/10.1038/nrg3245
Nishiyama T (2018) Cohesion and cohesin-dependent chromatin organization. Curr Opin Cell Biol 58:8–14. https://doi.org/10.1016/j.ceb.2018.11.006
Nishiyama T et al (2010) Sororin mediates sister chromatid cohesion by antagonizing Wapl. Cell 143:737–749. https://doi.org/10.1016/j.cell.2010.10.031
Nishiyama T, Sykora MM, Huis in ‘t Veld PJ, Mechtler K, Peters JM (2013) Aurora B and Cdk1 mediate Wapl activation and release of acetylated cohesin from chromosomes by phosphorylating Sororin. Proc Natl Acad Sci USA 110:13404–13409. https://doi.org/10.1073/pnas.1305020110
Padmore R, Cao L, Kleckner N (1991) Temporal comparison of recombination and synaptonemal complex formation during meiosis in S. cerevisiae. Cell 66:1239–1256
Patel L et al (2019) Dynamic reorganization of the genome shapes the recombination landscape in meiotic prophase. Nat Struct Mol Biol. https://doi.org/10.1038/s41594-019-0187-0
Pezzi N et al (2000) STAG3, a novel gene encoding a protein involved in meiotic chromosome pairing and location of STAG3-related genes flanking the Williams-Beuren syndrome deletion. FASEB J 14:581–592
Prieto I, Suja JA, Pezzi N, Kremer L, Martinez AC, Rufas JS, Barbero JL (2001) Mammalian STAG3 is a cohesin specific to sister chromatid arms in meiosis I. Nat Cell Biol 3:761–766. https://doi.org/10.1038/35087082
Rankin S (2005) Sororin, the cell cycle and sister chromatid cohesion. Cell Cycle 4:1039–1042. https://doi.org/10.4161/cc.4.8.1926
Revenkova E et al (2004) Cohesin SMC1 beta is required for meiotic chromosome dynamics, sister chromatid cohesion and DNA recombination. Nat Cell Biol 6:555–562. https://doi.org/10.1038/ncb1135
Riedel CG et al (2006) Protein phosphatase 2A protects centromeric sister chromatid cohesion during meiosis I. Nature 441:53–61. https://doi.org/10.1038/nature04664
Rogers E, Bishop JD, Waddle JA, Schumacher JM, Lin R (2002) The aurora kinase AIR-2 functions in the release of chromosome cohesion in Caenorhabditis elegans meiosis. J Cell Biol 157:219–229. https://doi.org/10.1083/jcb.200110045
Severson AF, Meyer BJ (2014) Divergent kleisin subunits of cohesin specify mechanisms to tether and release meiotic chromosomes. Elife 3:e03467. https://doi.org/10.7554/eLife.03467
Shinohara M, Gasior SL, Bishop DK, Shinohara A (2000) Tid1/Rdh54 promotes colocalization of Rad51 and Dmc1 during meiotic recombination. Proc Natl Acad Sci USA 97:10814–10819
Tachibana-Konwalski K, Godwin J, van der Weyden L, Champion L, Kudo NR, Adams DJ, Nasmyth K (2010) Rec8-containing cohesin maintains bivalents without turnover during the growing phase of mouse oocytes. Genes Dev 24:2505–2516. https://doi.org/10.1101/gad.605910
Tachibana-Konwalski K, Godwin J, Borsos M, Rattani A, Adams DJ, Nasmyth K (2013) Spindle assembly checkpoint of oocytes depends on a kinetochore structure determined by cohesin in meiosis I. Curr Biol 23:2534–2539. https://doi.org/10.1016/j.cub.2013.10.052
Tedeschi A et al (2013) Wapl is an essential regulator of chromatin structure and chromosome segregation. Nature 501:564–568. https://doi.org/10.1038/nature12471
van Ruiten MS, Rowland BD (2018) SMC complexes: universal DNA looping machines with distinct regulators. Trends Genet 34:477–487. https://doi.org/10.1016/j.tig.2018.03.003
Verni F, Gandhi R, Goldberg ML, Gatti M (2000) Genetic and molecular analysis of wings apart-like (wapl), a gene controlling heterochromatin organization in Drosophila melanogaster. Genetics 154:1693–1710
Villa-Hernandez S, Bermejo R (2018) Cohesin dynamic association to chromatin and interfacing with replication forks in genome integrity maintenance. Curr Genet 64:1005–1013. https://doi.org/10.1007/s00294-018-0824-x
Watanabe Y, Nurse P (1999) Cohesin Rec8 is required for reductional chromosome segregation at meiosis. Nature 400:461–464. https://doi.org/10.1038/22774
Wolf PG, Cuba Ramos A, Kenzel J, Neumann B, Stemmann O (2018) Studying meiotic cohesin in somatic cells reveals that Rec8-containing cohesin requires Stag3 to function and is regulated by Wapl and sororin. J Cell Sci. https://doi.org/10.1242/jcs.212100
Yamada T, Tahara E, Kanke M, Kuwata K, Nishiyama T (2017) Drosophila Dalmatian combines sororin and shugoshin roles in establishment and protection of cohesion. EMBO J 36:1513–1527. https://doi.org/10.15252/embj.201695607
Yu HG, Koshland D (2005) Chromosome morphogenesis: condensin-dependent cohesin removal during meiosis. Cell 123:397–407. https://doi.org/10.1016/j.cell.2005.09.014
Zhu Z, Mori S, Oshiumi H, Matsuzaki K, Shinohara M, Shinohara A (2011) Cyclin-dependent kinase promotes formation of the synaptonemal complex in yeast meiosis. Genes Cells 15:1036–1050
Zickler D, Kleckner N (1999) Meiotic chromosomes: integrating structure and function. Annu Rev Genet 33:603–754
Acknowledgements
We are grateful for Drs. Susan Gasser and Franz Klein for constructive comments on the paper. We also thank Dr. K. Kim for strains. This work was supported by JSPS KAKENHI Grant Numbers; 22125001, 22125002, 15H05973 and 16H04742 to A.S.
Author information
Authors and Affiliations
Contributions
KC and AS wrote an initial draft and AS revised. KC, MS, and AS edited the manuscript.
Corresponding author
Additional information
Communicated by M. Kupiec.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Challa, K., Shinohara, M. & Shinohara, A. Meiotic prophase-like pathway for cleavage-independent removal of cohesin for chromosome morphogenesis. Curr Genet 65, 817–827 (2019). https://doi.org/10.1007/s00294-019-00959-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-019-00959-x