Abstract
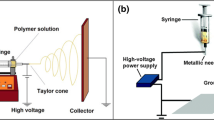
Electrospun nanofiber scaffolds mimic the structure of the natural extracellular matrix, making them promising for application in tissue engineering. Based on this, the biopolymers poly(ε-caprolactone) (PCL) and gelatin, with good mechanical and biological properties, respectively, were electrospun with acetic acid as the only solvent, without the crosslinking process in order to reduce the cytotoxic effects of commonly used solvents. This study evaluated the influence of scaffold thicknesses on the physicochemical properties. Scaffolds with 4 different thicknesses were produced by increasing the electrospinning time (A = 1 h, B = 1.5 h, C = 2 h and D = 3 h). Thicker scaffolds (B, C and D, 0.142 ± 0.029 to 0.306 ± 0.053 nm) were manipulated easily, presented higher mechanical strength (from 156.21 to 178.53%) and smaller diameter fibers (approximately 300.0 nm). On the other hand, the longer the processing time, the less uniform the fibers and more melting regions between fibers, which may hinder the use of the scaffold and should be considered. Only the thickest scaffold (D) showed higher wettability (contact angle 31.1° ± 3°). Intermediate thickness scaffolds showed less beads and fiber irregularities. Cell adhesion was good due to gelatin, though cell proliferation remained constant for the thicker scaffold indicating that longer processing time may not be advantageous for cell application. Results showed that intermediate thickness scaffolds of PCL/gelatin (0.142 ± 0.029 to 0.195 ± 0.034 nm) assure good physicochemical properties to be applied for different cell types.
Graphical abstract







Similar content being viewed by others
References
Hinderer S, Schesny M, Bayrak A, Ibold B, Hampel M, Walles T, Stock UA, Seifert M, Schenke-Layland K (2012) Engineering of fibrillar decorin matrices for a tissue-engineered trachea. Biomaterials 33:5259–5266. https://doi.org/10.1016/j.biomaterials.2012.03.075
Gautam S, Dinda AK, Mishra NC (2013) Fabrication and characterization of PCL/gelatin composite nanofibrous scaffold for tissue engineering applications by electrospinning method. Mater Sci Eng, C 33:1228–1235. https://doi.org/10.1016/j.msec.2012.12.015
Agarwal S, Wendorff JH, Greiner A (2009) Progress in the field of electrospinning for tissue engineering applications. Adv Mater 21:3343–3351. https://doi.org/10.1002/adma.200803092
Chandrasekaran AR, Venugopal J, Sundarrajan S, Ramakrishna S (2011) Fabrication of a nanofibrous scaffold with improved bioactivity for culture of human dermal fibroblasts for skin regeneration. Biomed Mater 6:015001. https://doi.org/10.1088/1748-6041/6/1/015001
Lian H, Meng Z (2017) Melt electrospinning vs. solution electrospinning: A comparative study of drug-loaded poly (ε-caprolactone) fibres. Mater Sci Eng C 74:117–123. https://doi.org/10.1016/j.msec.2017.02.024
Daglar O, Altinkok C, Acik G, Durmaz H (2020) Electrospinning of poly(1,4-cyclohexanedimethylene acetylene dicarboxylate): study on the morphology, wettability, thermal and biodegradation behaviors. Macromol Chem Phys. https://doi.org/10.1002/macp.202000310
Ratner BD, Hoffman AS, Schoen FJ, Lemons JE (2004) Biomaterials science: an introduction to materials in medicine, 2nd edn. Academic Press, San Diego. https://doi.org/10.1016/B978-0-08-087780-8.00148-0
Cipitria A, Skelton A, Dargaville TR, Dalton PD, Hutmacher DW (2011) Design, fabrication and characterization of PCL electrospun scaffolds—a review. J Mater Chem 93:1539–1550. https://doi.org/10.1039/c0jm04502k
Chong EJ, Phan TT, Lim IJ, Zhang YZ, Bay BH, Ramakrishna S, Lim CT (2007) Evaluation of electrospun PCL/gelatin nanofibrous scaffold for wound healing and layered dermal reconstitution. Acta Biomater 3:321–330. https://doi.org/10.1016/j.actbio.2007.01.002
Azimi B, Nourpanah P, Rabiee M, Arbab S (2014) Poly (ε-caprolactone) fiber: an overview. J Eng Fiber Fabr 9:74–90. https://doi.org/10.1177/155892501400900309
Xue J, He M, Liang Y, Crawford A, Coates P, Chen D, Shi R, Zhang L (2014) Fabrication and evaluation of electrospun PCL–gelatin micro-/nanofiber membranes for anti-infective GTR implants. J Mater Chem B 2:6867–6877. https://doi.org/10.1039/C4TB00737A
Zhou Z, Zhou Y, Chen Y, Nie H, Wang Y, Li F, Zheng Y (2011) Bilayer porous scaffold based on poly-(ε-caprolactone) nanofibrous membrane and gelatin sponge for favoring cell proliferation. Appl Surf Sci 258:1670–1676. https://doi.org/10.1016/j.apsusc.2011.09.120
Merk M, Chirikian O, Adlhart C (2021) 3D PCL/gelatin/genipin nanofiber sponge as scaffold for regenerative medicine. Materials. https://doi.org/10.3390/ma14082006
Ghomi ER, Lakshminarayanan R, Chellappan V, Verma NK, Chinnappan A, Neisiany RE, Amuthavalli K, Poh ZS, Wong BHS, Dubey N, Narayan R, Ramakrishna S (2023) Electrospun aligned PCL/gelatin scaffolds mimicking the skin ECM for effective antimicrobial wound dressings. Adv Fiber Mater 5:235–251. https://doi.org/10.1007/s42765-022-00216-w
Lashkari M, Rahmani M, Yousefpoor Y, Ahmadi-Zeidabadi M, Faridi-Majidi R, Ameri Z, Salary M, Azizi S, Shahabi A, Rahi A, Mirzaei-Parsa MJ (2023) Cell-based wound dressing: bilayered PCL/gelatin nanofibers-alginate/collagen hydrogel scaffold loaded with mesenchymal stem cells. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2023.124099
Kim YH, Kim DH, Hwang J, Kim HS, Lim GY, Ryoo ZY, Choi SU, Lee S (2013) The inclusion of fetal bovine serum in gelatin/PCL electrospun scaffolds reduces short-term osmotic stress in HEK 293 cells caused by scaffold components. J Appl Polym Sci 129:3273–3281. https://doi.org/10.1002/app.39052
Heydarkhan-Hagvall S, Schenke-Layland K, Dhanasopon AP, Rofail F, Smith H, Wu BM, Shemin R, Beygui RE, MacLellan WR (2008) Three-dimensional electrospun ECM-based hybrid scaffolds for cardiovascular tissue engineering. Biomaterials 29:2907–2914. https://doi.org/10.1016/j.biomaterials.2008.03.034
Zhang Y, Ouyang H, Chwee TL, Ramakrishna S, Huang ZM (2005) Electrospinning of gelatin fibers and gelatin/PCL composite fibrous scaffolds. J Biomed Mater Res B Appl Biomater 72:156–165. https://doi.org/10.1002/jbm.b.30128
Reddy N, Reddy R, Jiang Q (2015) Crosslinking biopolymers for biomedical applications. Trends Biotechnol 33:362–369. https://doi.org/10.1016/j.tibtech.2015.03.008
Rajzer I, Rom M, Menaszek E, Pasierb P (2015) Conductive PANI patterns on electrospun PCL/gelatin scaffolds modified with bioactive particles for bone tissue engineering. Mater Lett 138:60–63. https://doi.org/10.1016/j.matlet.2014.09.077
Ferreira JL, Gomes S, Henriques C, Borges JP, Silva JC (2014) Electrospinning polycaprolactone dissolved in glacial acetic acid: fiber production, nonwoven characterization, and in vitro evaluation. J Appl Polym Sci 41068:37–39. https://doi.org/10.1002/app.41068
Erencia M, Cano F, Tornero JA, Fernandes MM, Tzanov T, Macanás J, Carrillo F (2015) Electrospinning of gelatin fibers using solutions with low acetic acid concentration: effect of solvent composition on both diameter of electrospun fibers and cytotoxicity. J Appl Polym Sci 132:1–11. https://doi.org/10.1002/app.42115
Pok SW, Wallace KN, Madihally SV (2010) In vitro characterization of polycaprolactone matrices generated in aqueous media. Acta Biomater 6:1061–1068. https://doi.org/10.1016/j.actbio.2009.08.002
Beachley V, Wen X (2009) Effect of electrospinning parameters on the nanofiber diameter and length. Mater Sci Eng C 29:663–668. https://doi.org/10.1016/j.msec.2008.10.037
Gomes SR, Rodrigues G, Martins GG, Roberto MA, Mafra M, Henriques CMR, Silva JC (2015) In vitro and in vivo evaluation of electrospun nanofibers of PCL, chitosan and gelatin: a comparative study. Mater Sci Eng C 46:348–358. https://doi.org/10.1016/j.msec.2014.10.051
Meng ZX, Zheng W, Li L, Zheng YF (2010) Fabrication and characterization of three-dimensional nanofiber membrane of PCL-MWCNTs by electrospinning. Mater Sci Eng C 30:1014–1021. https://doi.org/10.1016/j.msec.2010.05.003
Ku SH, Park CB (2010) Human endothelial cell growth on mussel-inspired nanofiber scaffold for vascular tissue engineering. Biomaterials 31:9431–9437. https://doi.org/10.1016/j.biomaterials.2010.08.071
Sill TJ, von Recum HA (2008) Electrospinning: applications in drug delivery and tissue engineering. Biomaterials 29:1989–2006. https://doi.org/10.1016/j.biomaterials.2008.01.011
Chiono V, Ciardelli G, Vozzi G, Cortez J, Barbani N, Gentile P, Giusti P (2008) Enzymatically-modified melt-extruded guides for peripheral nerve repair. Eng Life Sci 8:226–237. https://doi.org/10.1002/elsc.200700069
Ghasemi-Mobarakeh L, Prabhakaran MP, Morshed M, Nasr-Esfahani MH, Ramakrishna S (2008) Electrospun poly(ε-caprolactone)/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials 29:4532–4539. https://doi.org/10.1016/j.biomaterials.2008.08.007
Lee J, Tae G, Kim YH, Park IS, Kim SH, Kim SH (2008) The effect of gelatin incorporation into electrospun poly(l-lactide-co-ε-caprolactone) fibers on mechanical properties and cytocompatibility. Biomaterials 29:1872–1879. https://doi.org/10.1016/j.biomaterials.2007.12.029
Njobuenwu DO, Oboho EO, Gumus RH (2007) Determination of contact angle from contact area of liquid droplet spreading on solid substrate. Leonardo Electron J Pract Technol 6:29–38
Ryan BJ, Poduska KM (2008) Roughness effects on contact angle measurements. Am J Phys 76:1074. https://doi.org/10.1119/1.2952446
Stalder AF, Kulik G, Sage D, Barbieri L, Hoffmann P (2006) A snake-based approach to accurate determination of both contact points and contact angles. Colloids Surf A Physicochem Eng Asp 286:92–103. https://doi.org/10.1016/j.colsurfa.2006.03.008
Acknowledgements
This work was supported by Coordenação de Aperfeiçoamento Pessoal de Nível Superior (CAPES). The authors also thank Escola Politécnica da Universidade de São Paulo (POLI-USP) and Instituto de Pesquisas Tecnológicas do Estado de São Paulo (IPT). The authors thank James Hesson for English language editing.
Author information
Authors and Affiliations
Contributions
VTK contributed to conceptualization, methodology, validation, formal analysis, investigation, data curation, writing—original draft, and visualization. MHAZ contributed to conceptualization, methodology, resources, project administration, writing—review and editing, and supervision. SHW helped in conceptualization, methodology, resources, project administration, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kimura, V.T., Zanin, M.H.A. & Wang, S.H. Influence of thickness on the properties of electrospun PCL/gelatin nanofiber scaffolds. Polym. Bull. (2024). https://doi.org/10.1007/s00289-024-05159-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00289-024-05159-3