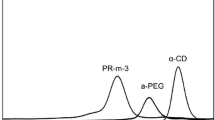
Abstract
An aqueous solution of chlorinated polyethylene glycol (Cl-PEG-Cl) and α-cyclodextrin (α-CD) was used to form inclusion complexes, which several α-CDs are threaded into the linear PEG chain and produced a so-called polypseudorotaxane (PPRx). By attaching 4-ethylaniline as the blocking agent to the chlorinated PEG chain ends, the complex will be stabilized, resulting in the formation of so-called polyrotaxane (PRx) (also known as molecular necklace). Finally, crosslinked αCD-based PRx with a tubular structure was formed via the condensation reactions between α-cyclodextrins and epichlorohydrin. Using concentrated NaOH solution, polymer chains were then removed from complexes, resulting in the so-called molecular tube (MT). This novel method led to the improvement in polyrotaxane conversion with less production steps and reduced materials overall costs. Resulting products in each section were characterized by 1H and 13C nuclear magnetic resonance spectroscopies (1H-NMR and 13C-NMR), from which complex inclusion ratio and length of the nanotubes were estimated. XRD was also used to confirm tubular structure of the inclusion complexes.
Graphical abstract









Similar content being viewed by others
Data availability
Data will be made available on the request.
Notes
(Benzotriazol-1-yloxy)tris(dimethylamino)-phosphonium hexafluorophosphate.
References
Ribeiro AC, Esteso MA (2023) Cyclodextrins as drug release modulators and toxic compound removal agents. Biomolecules 13:1056
Cai X, Liu H, Zhang G (2023) Control of the threading ratio of rings in a polypseudorotaxane: a computer simulation study. Polymer 268:125705
Harada A, Li J, Kamachi M (1993) Synthesis of a tubular polymer from threaded cyclodextrins. Nature 364:516–518
Kurlykin MP, Golovina MA, Tenkovtsev AV (2023) New type polyrotaxanes based on polyethyleneimine and α-cyclodextrin. Mendeleev Commu 33:360–361
Ceccato M, Lo Nostro P, Rossi C, Bonechi C, Donati A, Baglioni P (1997) Molecular dynamics of novel α-cyclodextrin adducts studied by 13C-NMR relaxation. J Phys Chem B 101:5094–5099
Harada A, Li J, Kamachi M (1993) Preparation and properties of inclusion complexes of polyethylene glycol with alpha.-cyclodextrin. Macromolecules 26:5698–5703
Harada A, Li J, Kamachi M (1994) Preparation and characterization of a polyrotaxane consisting of monodisperse poly (ethylene glycol) and alpha.-cyclodextrins. J Am Chem Soc 116:3192–3196
Harada A, Li J, Kamachi M (1992) The molecular necklace: a rotaxane containing many threaded α-cyclodextrins. Nature 356:325–327
Harada A, Li J, Nakamitsu T, Kamachi M (1993) Preparation and characterization of polyrotaxanes containing many threaded. alpha.-cyclodextrins. J Org Chem 58:7524–7528
Pillai VR, Mutter M, Bayer E, Gatfield I (1980) New, easily removable poly (ethylene glycol) supports for the liquid-phase method of peptide synthesis. J Org Chem 45:5364–5370
Chen HY, Shan YK, Qin YY, Zhou GX, Yuan ML (2013) Synthesis of poly (ethylene glycol) derivatives. Adv Mater Res 750:1674–1677
Zalipsky S (1995) Functionalized poly (ethylene glycols) for preparation of biologically relevant conjugates. Bioconjugate Chem 6:150–165
Fujita H, Ooya T, Yui N (1999) Thermally induced localization of cyclodextrins in a polyrotaxane consisting of β-cyclodextrins and poly (ethylene glycol)−poly(propylene glycol) triblock copolymer. Macromolecules 32:2534–2541
Tamura M, Ueno A (1998) Energy transfer in a rotaxane with a aphthalene-modified α-cyclodextrin threaded by dansyl-terminal poly (ethylene glycol). Chem Lett 27:369–370
Loethen S, Ooya T, Choi HS, Yui N, Thompson DH (2006) Synthesis, characterization, and pH-triggered dethreading of α-cyclodextrin-poly (ethylene glycol) polyrotaxanes bearing cleavable endcaps. Biomacromol 7:2501–2506
Ooya T, Ichi T, Furubayashi T, Katoh M, Yui N (2007) Cationic hydrogels of PEG crosslinked by a hydrolyzable polyrotaxane for cartilage regeneration. React Funct Polym 67:1408–1417
Arunachalam M, Gibson HW (2014) Recent developments in polypseudorotaxanes and polyrotaxanes. Prog Polym Sci 39:1043–1073
Kobayashi Y (2021) Precise synthesis of polyrotaxane and preparation of supramolecular materials based on its mobility. Polym J 53:505–513
Zhao T, Beckham HW (2003) Direct synthesis of cyclodextrin-rotaxanated poly (ethylene glycol) s and their self-diffusion behavior in dilute solution. Macromolecules 36:9859–9865
Teuchert C, Michel C, Hausen F, Park DY, Beckham HW, Wenz G (2013) Cylindrical polymer brushes by atom transfer radical polymerization from cyclodextrin–PEG polyrotaxanes: synthesis and mechanical stability. Macromolecules 46:2–7
Araki J, Zhao C, Ito K (2005) Efficient production of polyrotaxanes from α-cyclodextrin and poly (ethylene glycol). Macromolecules 38:7524–7527
Tait CN, Davies DM (2002) Polypseudorotaxanes from scratch. Langmuir 18:2453–2454
Harada A (1999) Design and construction of supramolecular architectures consisting of cyclodextrins and polymers. In: Canelas DA, DeSimone JM et al. (eds) Metal complex catalysts supercritical fluid polymerization supramolecular architecture. Springer, New York, pp141–191
Harris JM (1985) Laboratory synthesis of polyethylene glycol derivatives. J Macromol Sci Rev Macromol Chem Phys 25:325–373
Huang L, Tonelli AE (1998) Polymer inclusion compounds. J Macromol Sci C 38:781–837
Peet J, Rusa CC, Hunt MA, Tonelli AE, Balik CM (2005) Solid-state complexation of poly (ethylene glycol) with α-cyclodextrin. Macromolecules 38:537–541
Zalipsky S, Gilon C, Zilkha A (1983) Attachment of drugs to polyethylene glycols. Eur Polym J 19:1177–1183
Bayer E, Zheng H, Geckeler K (1982) Functionalization of soluble polymers. Polym Bull 8:585–591
Tonegawa A, Tamura A, Zhang S, Yui N (2020) Hydrophobicity of acyl groups in α-cyclodextrin-threaded polyrotaxanes dominates the formation and stability of self-assembled nanoparticles. Polymer 200:122537
Wang J, Gao P, Jiang L, Ye L, Zhang AY, Feng ZG (2012) Poly (n-butyl methacrylate) end-capped polyrotaxanes via ATRP initiated with α-cyclodextrin and Pluronic 17R4 based inclusion complexes. Polymer 53:2864–2872
Guo CG, Wang L, Li YK, Wang CQ (2013) Suprarmolecular hydrogels driven by the dual host–guest interactions between α-cyclodextrin and ferrocene-modified poly (ethylene glycol) with low-molecular-weight. React Funct Polym 73:805–812
El-Aouni N, Hsissou R, El Azzaoui J, El Bouchti M, Elharfi A (2020) Synthesis, rheological and thermal studies of epoxy polymer and its composite. Chem Data Collect 30:100584
Acknowledgements
The authors would like to acknowledge the Tarbiat Modares University (Grant number # 81586) for the financial support.
Author information
Authors and Affiliations
Contributions
NP: Experimental design; Carrying out measurements; Formal analysis; Investigation; Methodology; Validation; Writing—original draft; MA: Conceptualization; Data curation; Funding acquisition; Investigation; Project administration; Supervision; Writing—review & editing. MAS: Conceptualization, Project administration; Supervision; Writing—review & editing.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that there is no conflicts of interest.
Ethical approval
There is no experiment involving human tissue in the present work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pakzad, N., Abdollahi, M. & Semsarzadeh, M.A. Synthesis and characterization of polyrotaxane-based molecular nanotubes prepared from α-cyclodextrin/chlorinated polyethylene glycol complex end capped with 4-ethylaniline. Polym. Bull. 81, 4661–4676 (2024). https://doi.org/10.1007/s00289-023-04936-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-023-04936-w